Quality prescribing for chronic pain: guide for improvement 2026-2029
Chronic pain, defined as persistent pain lasting beyond three months, can significantly impact quality of life, affecting physical and mental wellbeing. This guide supports clinicians and people living with chronic pain, using a person-centred approach to pain management.
9. Benefits and limitations of treatment with medication
9.1 Introduction
Jennifer’s account of living with and managing chronic pain
“I have been suffering from chronic pain since 2014. I had an operation on my back about ten years ago, as I had benign tumours on my hips and neck. Five years ago, I had a spinal leakage, and then had sepsis and meningitis and was in and out of hospital many times. I was in chronic pain all throughout my illness, which includes fibromyalgia, and was on constant painkillers. For almost three years I was taking high dose opioids.
It feels like I actually lost three years of my life as I was spending up to 19 hours a day in bed. I lost interest in everything and just couldn’t do anything. I was in constant pain, even with the pain medicines, and I wasn’t able to go out.
I was referred to the Pain Team and they greatly helped me to manage and reduce my medication. I feel now that I am getting part of my life back. I am still in pain, but I was also in pain when I was on the high doses of opioids. If I have a bad day now, I will rest but when I feel a bit better, I try to get dressed and go out.
I take pride in my appearance again. I now feel better in myself as I was very low in spirit, when I was very ill and on the maximum pain medicines. I feel as though I am part of my old self again and look forward to making things better.
I am still not free from chronic pain, but I feel very grateful for the help of the doctor and Pain Team to help me feel as well as I can possibly be. It took a lot of determination to reach this point, but I feel a lot better now that I have reduced my medication. I am less tired and have more energy. I feel like a new person now.”
How our understanding of medicine use in chronic pain is changing
The use of analgesics in chronic pain is changing, as more evidence is available:
- about longer term risks and harms
- what groups of medicines can be used for different indications/conditions
- the benefits of non-pharmacological treatments
A summary of treatment options and indications is included alongside this guide as a supporting document (based on that of NHS Fife).
Numbers Needed to Treat (NNT)
This guide intends to be a practical tool to help prescribers decide when it is appropriate to initiate and continue medicines for people with chronic pain. In some circumstances it may be appropriate to discontinue treatments. Presentation of the number needed to treat (NNT) for a range of medicines is a tool that prescribers may use to aid discussions with individuals
The NNT is defined as the expected number of people who need to receive the intervention (be it a medicine, treatment, change) rather than the comparator intervention for one person to either incur or avoid an event in each time frame. An NNT of 10 can be interpreted that one additional (or less) person will incur an event for every 10 participants receiving the experimental intervention rather than control over a given time frame. This is referred to as the NNT to benefit.
For more information on NNTs please see Appendix I.
9.2 Important benefits and risks of treatment
General
- Numbers needed to treat (NNTs) in the management of chronic pain may appear relatively low, e.g. NNT for gabapentin for managing fibromyalgia is 5.4. However, this means that for every one person with reduced pain intensity, there are four to five people who receive no benefit, with increased risk of adverse effects. Ineffective medication should be safely stopped
- Clinicians should have honest conversations with people at the onset of prescribing about the limitation of medication in the management of chronic pain
- Opioids, gabapentinoids and antidepressants are associated with a risk of dependency. [42],[43] Individuals should be made aware of these risks on initiation and supported to withdraw these medicines as appropriate
- Clinicians should be aware of the possibility that some individuals may present with ‘drug seeking behaviour’, due to the potential for addiction and withdrawal reactions from gabapentinoids, opioids and antidepressants. MHRA has strengthened advice on the dependency and addiction risk of these medicines
- There are potentially fatal interactions between medicines that cause central nervous system (CNS) depression, particularly opioids, gabapentinoids and benzodiazepines
Medication effectiveness
- Total eradication of pain is unlikely, and evidence reviews frequently consider an intervention effective if it reduces pain by 30%5 to 50% [44]
- There is a high placebo effect, highlighting that biopsychosocial effects are contributory factors in pain management. For example, a Cochrane review showed pregabalin 300mg daily reduced pain intensity by 30% in 47% of patients, but the placebo was similarly effective in 42% of patients44
- It is important to not only assess the impact of medication on pain levels, but also consider their effect on physical function, mental health and emotional factors (e.g. mood, anxiety, sleep, social and sexual function)
9.3 Paracetamol
Indications and effectiveness
Efficacy is clear in acute situations with an NNT of 3.8 for 50% pain relief with a single 1000mg dose. It is less clear in chronic pain depending on the condition.
- Clinicians should be aware that paracetamol does not significantly improve pain or function in osteoarthritis in short term (three to six months) or intermediate term (more than six months)46
- Current guidance notes paracetamol should not be used as sole therapy for chronic backpain17,18, [45] sciatica (lumbar radiculopathy)18,45 or osteoarthritis [46] but may be considered in conjunction with NSAIDs
- Evidence showed showed that it has no additional benefit in improving osteoarthritis pain, quality of life or physical function compared with placebo.[47] However, it is recognised that some people cannot use NSAIDS. Therefore it was noted there were some circumstances where it may be used as a sole agent
- There are unlikely to be new large-scale trials with paracetamol. It is widely available over the counter (OTC) and as such individuals can be dismissive of its benefits before trialling. Pragmatically, for longer term pain conditions it may be worth trialling and assessing impact on pain relief and/or function. There are no strong recommendations for regular or ‘as required’ dosing and this should be assessed at individual level of benefit. Similarly, whilst there is no specific evidence to support, it may help minimise opioid load and could be prescribed alongside opioids either in combination or as individual products
- Good Practice Points:
- In persistent pain a two-to-four-week trial of regular paracetamol dosing three to four times daily can be used to assess efficacy
- Individuals can determine whether regular use or when required use suits their pain management most
- Regular use may support reduced use of opioids
Risks and harms
- Paracetamol is generally safe in appropriate doses although adverse reactions such as gastrointestinal (GI) bleeds and decreased renal function have been identified, linked to possible prostacyclin mode of action.[48] More research is needed
- Standard dosing of a healthy average weight adult in the UK is 500-1000mg every four to six hours (4 grams per 24 hours)
- Dosing should be reduced in people with low body weight (>33kg to <50kg) and in those over 50 kg with risk factors for hepatotoxicity
- Risk factors include:
- excess alcohol consumption
- concomitant medicines that induce liver enzymes (e.g. carbamazepine, St John’s Wort)
- those likely to have reduced glutathione levels (eating disorders/cystic fibrosis)
- the frail and/or elderly - some clinicians prefer to limit dose per administration in the frail elderly over 75 years to 500mg as a single dose and a maximum of 2g daily. Check your local guidance
Individuals should be aware that other medicinal products may contain paracetamol and lead to inadvertent excess dosing e.g. cough and cold preparations. The total daily dose of paracetamol across all products should not exceed 4g.
| Our recommendations | Strength of recommendation |
|---|---|
| There is no evidence to support the use of paracetamol in osteoarthritis, chronic back pain or sciatica (lumbar radiculopathy)17,18,45,47 | Strong |
| Paracetamol could be considered in osteoarthritis with an NSAID, recognising that some people cannot use NSAIDs and therefore sole use could be considered47 | Conditional |
| A two-to-four-week trial of regular paracetamol could be considered in chronic pain to assess efficacy | Good practice point |
| Paracetamol dosing should be reduced in people with low body weight (>33kg to <50kg) and in those over 50 kg with risk factors for hepatotoxicity[49] | Strong |
9.4 Non-steroidal anti-inflammatory drugs (NSAIDs)
Indications and effectiveness
Oral NSAIDs
Generally:
- 60% of people will respond to any NSAID. Others will respond to one NSAID more than another
- Analgesic effect starts with the first dose and full analgesic effect occurs after one week, whereas anti-inflammatory effect is evident after three weeks
- If there is no response within the expected timeframe, there should be a change of NSAID [50]
- NNTs vary from 2.4 (ibuprofen 400mg) to 3.0 (naproxen 550mg) to obtain 50% pain relief in acute pain, based on dental abscess pain,[51] over four to six hours
- Fibromyalgia:[52] NSAIDs cannot be considered as useful in the management of fibromyalgia due to very low-quality evidence
- NSAIDs (non-selective and selective) should be considered for short term (less than six months) use in those with osteoarthritis and inflammatory arthritis46
- Osteoarthritis: NSAIDs should be considered in the management of pain in individuals with hip or knee osteoarthritis with paracetamol (1,000-4,000mg/day) in addition to non-pharmacological treatments47, [53]
- Low back pain and sciatica (lumbar radiculopathy):
- SIGN 2025 notes that evidence for the use of NSAIDs for chronic lower back pain is inconsistent and the certainty of evidence was insufficient and therefore could not make a recommendation for or against use of NSAIDs in chronic low back pain46
- There is low certainty of evidence that NSAIDs may provide small effect on chronic low back pain[54]
- NICE states there is insufficient evidence for and against the use of NSAIDs in sciatica (lumbar radiculopathy), but there is increased risk of harm45
- Neuropathic pain (including post herpetic neuralgia): NSAIDs should not be used
Topical NSAIDs
- There was no difference in efficacy in a direct comparison of a topical NSAID with an oral NSAID [55]
- Topical NSAIDs could be considered in chronic musculoskeletal conditions, particularly in individuals who cannot tolerate oral NSAIDs
- Osteoarthritis:
- Offer a topical NSAID to people with knee osteoarthritis46,53 and hand osteoarthritis55 although evidence is limited to six to 12 weeks follow up.
- o Consider a topical NSAID for people with osteoarthritis that affects other joints47
Risks and harms of oral NSAIDs
- Gastrointestinal (GI):
- There are risks associated with long-term use
- These effects are systemic rather than local. Therefore, use of enteric-coated preparations are generally not beneficial50
- GI adverse effects are variable between different NSAIDs:
- highest risk: piroxicam, ketoprofen
- intermediate risk: indomethacin, diclofenac, naproxen
- lowest risk: ibuprofen50
- COX-2 inhibitors (e.g. etoricoxib, celecoxib) have lower risk of upper GI events, but these are negated if low dose aspirin is taken
- If NSAIDs are required in those at risk (e.g. history of peptic ulcer disease or gastrointestinal bleeding, aged over 65 years, dual antiplatelet, with concurrent corticosteroids, combination with selective serotonin reuptake inhibitors (SSRI)) GI protection should be considered
- Renal impairment: NSAIDs should be avoided as the risk of renal failure is highest in those with existing renal impairment.[56] If necessary, use with caution for the shortest time period and at the lowest effective dose. Note that the risk is higher when individuals are prescribed any three of metformin, NSAIDs, ACE inhibitors/ angiotensin-II receptor blocker (ARB), or diuretics (triple whammy).
- Bleeding
- high-risk combinations – gastrointestinal bleeding concomitant use of NSAIDs and SSRIs
- high-risk combinations - Use of combination antiplatelets/anticoagulants
Cardiovascular Risk
Individuals treated with COX-2 inhibitors may be at an increased risk of cardiovascular issues, such as angina pectoris, myocardial infarction, cerebral infarction, embolism and thrombosis. COX-2 inhibitors are estimated to be associated with approximately three additional thrombotic events per 1000 individuals per year in the general population.
Diclofenac is associated with cardiovascular risks that are higher than the other non-selective NSAIDs and are similar to the selective COX-2 inhibitors.
Naproxen and low-dose ibuprofen (less than 2400mg daily) are considered to have the most favourable cardiovascular safety profiles of all non-selective NSAIDs.
- Do not prescribe COX-2 inhibitors, diclofenac, aceclofenac, or high-dose ibuprofen (more than 2400mg daily) to individuals with:
- ischaemic heart disease
- congestive heart failure (New York Heart Association [NYHA] classification II–IV)
- cerebrovascular disease
- peripheral arterial disease
- inflammatory bowel disease (COX-2 inhibitors only)
- Do not prescribe etoricoxib or high-dose ibuprofen to individuals with uncontrolled hypertension (persistently above 140/90 mmHg).
- Prescribe NSAIDs with caution to people with:
- cardiac impairment or heart failure
- impaired renal function
- cerebrovascular disease
- hypertension — NSAIDs may impair renal function
- ischaemic heart disease
- peripheral arterial disease
- risk factors for cardiovascular events — for example, hypertension, hyperlipidaemia, diabetes mellitus, and smoking
- Elderly/frailty: avoid use of NSAIDs wherever possible due to increased risks of adverse effects56
- NSAIDs are cautioned or contra-indicated in people with asthma, depending on their previous history of NSAID use, as they may worsen asthma symptoms[57]
Risks and harms of topical NSAIDs
- Local adverse effects (mostly mild skin reactions) were more common with topical NSAIDs compared with placebo or oral NSAIDs. There was no increase in serious adverse events
- GI adverse effects with topical NSAIDs did not differ from placebo but were less frequent than with oral NSAIDs55
- Topical NSAIDs can be used in renal impairment, although deterioration of renal function has been reported after topical use
| Our recommendations | Strength of recommendation |
|---|---|
| NSAIDs (non-selective and selective) should be considered for less than six months use in people with osteoarthritis and inflammatory arthritis46 | Conditional |
| Assessment of risk of gastrointestinal bleeding and cardiovascular issues should be undertaken before initiating NSAIDs (both non-selective and selective)50 | Strong |
| Consider offering a topical NSAID to those with knee and hand osteoarthritis (and other joints affected by osteoarthritis)55 | Conditional |
9.5 Opioids
Indications and effectiveness
Summary:
- Opioids should not be considered routinely for people with chronic non-malignant pain. In carefully selected individuals, when other therapies have been fully explored, opioids can be considered for short-term treatment (up to three months), if it is considered that the potential benefits outweigh the risks of serious harms such as addiction, overdose and death37, 53, [58], [59], [60], [61], [62], [63], [64]
- The benefits of opioids in chronic pain are limited to lower doses for short to medium-term treatment (less than three months)
- There is some evidence for intermittent use of weaker opioids e.g. codeine or dihydrocodeine on a when required basis, especially if a particular activity/scenario causes a short term increase in pain47
Generally:
- Prescribers should consider the risks and harms against the benefits of taking opioids61
- Individual review is recommended within four weeks of starting opioid treatment or sooner if required [65]
- Individuals should be informed that if pain remains severe despite opioid treatment, this indicates that the opioid is not working and should be stopped, even if no other treatment is available [66]
- The frequency of review once the opioid regimen is established depends on individual factors, but should be at least, annually65 to identify possible harms and consider the effectiveness of treatment
- All people on an opioid should have a review of their treatment, prioritising individuals prescribed more than 50mg morphine equivalent dose (MED), who should be reviewed at least annually to consider ongoing effectiveness and potential harms, due to lack of evidence to support long-term use.65, [67], [68] See Appendix C for dose equivalence table
- Higher doses of opioids have a greater risk of serious harm with no additional benefit:
- 50-100mg MED, has a four times higher risk than less than 50mg MED
- Greater than 100mg MED has a nine times higher risk
- Serious harms include inappropriate opioid use, overdose and death, increased risk for falls and major trauma, and increased likelihood of road accidents65
- Due to tolerance developing, the analgesic effect of opioids lessens over time and individuals require higher doses to maintain the same level of pain relief[69],65,[70]
- If a change from one opioid to another is required, the relative potency should be accounted for and dose equivalence charts used (see Appendix C). Particular caution is required with fentanyl due to its high potency
- If the decision has been agreed to prescribe an opioid for an individual with chronic pain it is good practice to agree an individualised treatment plan, which sets realistic goals, highlights potential side effects and risks and sets criteria for stopping the opioid. For more details, see Appendix C
- Where possible, the usefulness of opioids should be explored by prescribing a short trial (one-to-two-week supply) of immediate release morphine tablets or liquid and reviewing to assess the response
Morphine:
- Individuals should be considered as opioid non-responsive if 20mg of immediate-release morphine does not reduce pain intensity
- As there is no evidence of difference in pain control between opioids, morphine is the preferred cost-effective option when a stronger opioid is required18
- As more evidence is available, the accepted upper dosage limits are being reviewed and reduced. Current recommendations indicate the upper dosage levels should be less than 50mg to minimise longer term risks and adverse effects, and previous upper limits of 90mg to 120mg should be reviewed
- It is good practice to not prescribe more than 90mg/day MED without seeking advice from prescriber specialising in pain
- It is good practice to use extreme caution when prescribing more than 120mg/day MED due to the risk of harm substantially increasing at these doses.65 Please note this does not apply when prescribed in palliative care and post-operative care
Tramadol:
- Has high potential for drug interactions. A MHRA Drug Safety Update highlighted the risk of increased International Normalised Ratio (INR) when warfarin and tramadol are used together, with a risk of major bruising and bleeding which could be life-threatening.
- It is good practice not to combine with other opioids for mild to moderate pain
- One study considered tramadol as suitable for neuropathic pain [71], however another found no significant difference between it and placebo59
- Tramadol and paracetamol combination product are not recommended by the SMC for use within Scotland for the symptomatic treatment of moderate to severe pain
Other opioids
- Transdermal buprenorphine or fentanyl could be an option for treatment when there are reasons prohibiting oral administration
Fentanyl:
- other analgesics and other opioids for non-malignant pain should be trialled before prescribing fentanyl patches
- due to the potency of fentanyl the use of transdermal patches is contra-indicated in opioid-naive individuals
- the use of immediate release fentanyl is not recommended as it is restricted for use for the management of breakthrough pain in adults using opioid therapy for chronic cancer pain, when other short-acting opioids are unsuitable
- is suitable in renal impairment and use with caution[72]
Buprenorphine:
- is an alternative for those with renal dysfunction (even severe)
- patches are available in several brands. They are available in different strengths and are not all interchangeable therefore buprenorphine patches should be prescribed by brand name
- patches are not appropriate for individuals with acute pain and those who need rapid dose escalation
- Seven-day transdermal patches (5 to 20 micrograms) should be prescribed in line with SMC restriction: For use in elderly individuals (over 65 years) for the treatment of non-malignant pain of moderate intensity when an opioid is necessary for obtaining adequate analgesia
Oxycodone:
- requires dose adjustment in renal impairment
- oxycodone and naloxone combination prolonged released tablets are not recommended by SMC
Risks and harms
- Risks can be categorised by the timescale on which they may be seen, although some can occur in any timeframe.
- Short-term:
- drowsiness, dizziness, confusion
- constipation, nausea/vomiting
- respiratory depression
- Long-term:
- dependence and addiction, even at therapeutic doses, following prolonged use (longer than three months)42
- may adversely affect the immune system (immunosuppression) [73]
- increased risk of myocardial infarction and cardiovascular revascularisation73
- hyperalgesia[74] – which is increased pain sensitivity
- adrenal insufficiency
- sexual dysfunction, fatigue and decreased levels of testosterone (in men and in women)[75],[76]
- Additional factors to consider during any timeframe:
- risk of overdose, problematic substance use, dependence, diversion
- interactions with other CNS depressant (such as anticonvulsants, benzodiazepines, gabapentinoids), with increased risk of respiratory depression and overdose[77], [78], [79]
- Increased fracture risk
- Individuals who require particular consideration to minimise the risk of harm include:
- Renal impairment
- Recently discharged following surgery
- Older/frail
- Short-term:
- The risks of dependence are increased in individuals with current or history of problem substance use (including alcohol) or mental health conditions (for example, major depression). Additional support and monitoring may be necessary when prescribing for individuals at risk of inappropriate opioid use.42
History of problem substance use
In 2014, the Medicines and Healthcare products Regulatory Agency (MHRA) highlighted the risk of inadvertent exposure to fentanyl patches, emphasising the need for providing information to patients and caregivers regarding the safe handling of patches and safe disposal of used and unused patches.
Prescribers should consider the adverse effects against the limited long-term benefits, as there are few trials examining the efficacy of long-term opioid use (≥3 months) in those with chronic pain. There are limited numbers of studies of variable quality which demonstrate long-term efficacy.61,65,68
Mitigation of harms and reduction of known adverse effects
Laxatives:
- Constipation is common even with low dose weak opioids. Laxatives should be considered when initially prescribing opioids, with a stimulant and softener being first choice, and continued if necessary on a regular basis.
Naloxone:
- Respiratory depression can occur, either with acute higher doses, or lower doses in at risk individuals. Naloxone is commonly used in substitution therapy, however its use to mitigate against respiratory depression in general medicine is an emerging area of understanding.
- Prior to prescribing naloxone, other means to reduce the risk of respiratory depression should be employed. This may include medication review, to reduce opioid dose or deprescribing of other medications which contribute to the risk (see polypharmacy triple whammy section later).
- Clinicians should consider prescribing naloxone for those prescribed an opioid and who may be at risk of an opioid overdose.[80] Take-Home Naloxone Programme is available across all boards in Scotland. The naloxone website has information on how to spot signs of overdose and how to administer naloxone.
| Our recommendations | Strength of recommendation |
|---|---|
| Opioids should not be considered routinely for people with chronic non-malignant pain. In carefully selected individuals, when other therapies have been fully explored, opioids can be considered for short-term treatment (up to three months), if it is considered that the potential benefits outweigh the risks of serious harms such as addiction, overdose and death17,53,58,59,60,61,62,63,64 | Strong |
| Prescribers should consider the adverse effects against the limited long-term benefits, as there are few trials examining the efficacy of long-term opioid use (≥3 months) in those with chronic pain67,68 | Strong |
| Individual review is recommended within four weeks of starting opioid treatment or sooner if required. The frequency of review once the opioid regimen is established depends on individual factors, but should be at least annually to identify possible harms and consider the effectiveness of treatment65 | Conditional |
| All people on an opioid should have a review of their treatment, prioritising individuals prescribed more than 50mg morphine equivalent dose (MED), who should be reviewed at least annually to consider ongoing effectiveness and potential harms, due to lack of evidence to support long-term use37,53,58,59,60,61,62,63,64,67,68 | Conditional |
| Do not prescribe more than 90mg/day MED without seeking advice from a prescriber specialising in pain management | Good practice point |
| Use extreme caution when prescribing more than 120mg/day MED due to the risk of harm substantially increasing at these doses (excluding palliative care) | Good practice point |
| Exercise caution when changing from one opioid to another, especially fentanyl due to its high potency | Good practice point |
| Clinicians should consider prescribing naloxone for those prescribed an opioid and who may be at risk of an opioid overdose | Good practice point |
9.6 Antidepressants
Indications and effectiveness
Recent SIGN evidence review only notes evidence for duloxetine,[81], [82] as the small study sizes for other antidepressants do not provide sufficient evidence to make an analysis of the outcomes. A lack of unbiased, high-quality evidence does not necessarily indicate evidence of no benefit.
Duloxetine
- Clinicians may consider a trial of duloxetine 60mg in people with chronic primary pain (including fibromyalgia, neuropathic pain and musculoskeletal) which is not adequately managed by analgesics,18,81,82, [83] noting that:
- the licensed indication is diabetic neuropathic pain81,82
- other guidelines consider it appropriate to try/consider unlicensed use in non-specific and neuropathic symptoms in lower back pain and fibromyalgia
- Review effectiveness and tolerability of duloxetine at four weeks. Discontinue duloxetine if there is no improvement in pain relief, physical function or subjective improvement after eight weeks. It is good practice to gradually reduce
- Monitor blood pressure in individuals with hypertension or other cardiac disease
- Do not prescribe to those with uncontrolled hypertension
To minimise the risk of side effects, especially nausea, it is good practice to start duloxetine at a lower dose of 30mg daily for two to four weeks before increasing to 60mg
| Medication | Indication | NNT | NNH81 |
|---|---|---|---|
| Duloxetine (50% pain intensity reduction with 60mg daily) | Chronic primary pain | 7.1 | 7 |
Tricyclic antidepressants (TCA)
- Recent evidence review is not conclusive for or against the use of TCAs in chronic pain81
- Other guidelines do consider the use of TCAs in chronic neuropathic pain, and clinicians should consider each person and their individual circumstances and chronic pain experience18,71,83
- Taking TCAs at evening mealtime rather than bedtime might address the problem of people being unable to initiate sleep and reduce fatigue the following morning23
- Due to risk of side effects, especially sedation, it is good practice to start amitriptyline at a lower dose of 10mg daily and an alternative TCA could be considered to reduce side effects
- TCAs should be used with caution in the older adult or in frailty due to anticholinergic effects and risk of falls[84]
- TCAs should not be used for the treatment of lower back pain83,[85]
Selective Serotonin Reuptake Inhibitors (SSRI)
- There is no high-quality evidence to support the use of other antidepressants (such as fluoxetine, paroxetine and citalopram) in the management of chronic pain. Historically they have been used, although unlicensed18
- There are potentially serious side effects of prescribing tramadol with an SSRI, such as serotonin syndrome
Concomitant depression
Optimise antidepressant medication where possible in those with concomitant chronic pain and depression, rather than prescribe additional antidepressant medication.[86] Combinations (e.g. SSRI with mirtazapine or trazodone, or SSRI and TCA) should be considered carefully (see Quality prescribing for antidepressants: a guide for improvement 2024 to 2027) to mitigate against the risk of adverse drug effects and should prompt consideration for specialist review.
Antidepressant good practice points
When an antidepressant is used to manage chronic pain, the individual should understand that these medicines may help with quality of life, pain, sleep, psychological distress, even in the absence of a diagnosis of depression.
Risks and harms
- Suicidality with increased risk of suicidal thoughts and acts, particularly in adolescents and young adults 18–21 years [87]
- Suicide and self-harm rates tend to be higher when antidepressants are started or stopped
- Individuals should be warned of this potential adverse effect during the early weeks of treatment and know how to seek help if required
- Switching antidepressants may be a marker of increased risk of suicidal behaviours in those aged 75 years and over87
- QT prolongation
- Serotonin syndrome
- Increased anticholinergic burden, particularly TCAs
- Inhibition of cytochrome enzymes – pharmacokinetic interactions
| Our recommendations | Strength of recommendation |
|---|---|
|
A trial of duloxetine 60mg may be considered in people with chronic primary pain (including fibromyalgia, neuropathic pain and musculoskeletal) which is not adequately managed by analgesics81,82, noting that
|
Conditional |
| Review effectiveness of duloxetine after four weeks and discontinue if there is no improvement in pain relief, physical function or subjective improvement. It is good practice to reduce gradually | Good practice point |
|
SIGN 2025 evidence review is not conclusive for or against the use of TCAs in chronic pain.81 Other guidelines do consider the use of TCAs in chronic neuropathic pain, and clinicians should consider each person and their individual circumstances and chronic pain experience |
Good practice point |
| TCAs should not be used for the treatment of lower back pain | Good practice point |
| There is no high-quality evidence to support the use of other antidepressants (such as fluoxetine, paroxetine and citalopram) in the management of chronic pain. Historically they have had a place in management, although unlicensed | Good practice point |
| Optimise antidepressant medication where possible in those with concomitant chronic pain and depression, rather than prescribe additional antidepressant medication | Good practice point |
9.7 Anticonvulsants
Indications and effectiveness
- Anticonvulsants could be considered to manage neuropathic pain (see Table 9)
- Much of the trial data for the effectiveness of gabapentinoids is limited to 12-16 weeks duration44
- Gabapentinoids should not be used in low back pain, sciatica (lumbar radiculopathy) and spinal stenosis [88]
- Gabapentin or pregabalin could be considered for individuals with the specific conditions of post herpetic neuralgia or painful diabetic neuropathy44,46
- Gabapentin or pregabalin could be considered for individuals with fibromyalgia, although this is unlicensed46
- Good practice point: Evidence indicates maximal dose of pregabalin 600mg daily/gabapentin 3600mg daily to achieve 30-50% pain intensity reduction)44,90 but if tolerated, minimum effective dose should be used
- Carbamazepine could be considered for trigeminal neuralgia, but individuals should be advised of adverse drug effects and interactions83, [89]
- Review effectiveness of gabapentinoids or carbamazepine after four to six weeks of therapy and discontinue if ineffective
- Good practice point: there is limited place in chronic pain therapy for other anticonvulsants and they should not be started in non-specialist settings
- To minimise the risk of side effects with gabapentin and pregabalin, it is good practice to start at lower doses and titrate to lowest effective dose
- It is good practice that, prior to commencement, agreement is established between the individual and clinician that prescribing is a trial and that if treatment is ineffective (or no improvement in function), it will not be continued
| Medication | Indication | NNT | NNH |
|---|---|---|---|
|
Gabapentin44 doses up to 3600mg daily [90] |
Post herpetic neuralgia | 844 | 8.644 |
|
Gabapentin doses up to 3600mg daily90 |
Painful diabetic neuropathy | 5.944 | 8.644 |
| Gabapentin maximum 2400mg daily90 | Fibromyalgia | 5.4 | 8.6 |
|
Pregabalin 600mg daily44 |
Post herpetic neuralgia | 3.9 | 11 |
|
Pregabalin 600mg daily44 |
Painful diabetic neuropathy | 7.8 | 11 |
|
Pregabalin 600mg daily44 |
Central neuropathic pain | 9.8 | 13.9 |
|
Pregabalin 600mg daily44 |
Fibromyalgia | 11 | 11 |
| Carbamazepine 44,[91] |
Trigeminal neuralgia (50% pain reduction) |
1.7 | 2.1 |
Risks and harms
- Prescribers and individuals should be aware there is a significant risk of adverse effects with gabapentinoids, including drowsiness, dizziness, nausea, weight changes (abnormal appetite), cognition, speech problems and ataxia83,88
- Adverse events lead to cessation of therapy in 18-28% of people44,83,88
- Reduced renal function - requires dose reduction of gabapentinoids to reduce risk of toxicity and side effects
- eGFR is frequently used to quantify the degree of renal impairment and is readily available on laboratory results and clinical systems. However, the licences for gabapentin and pregabalin are based on creatinine clearance (CrCl) which usually relies on manual calculation
- Clinical calculators are available to support calculation for example MedicinesComplete — Calculators
| CrCl ml/min* | Maximum recommended dose (BNF) | Additional Info |
|---|---|---|
| 50-79 | 600-1800mg daily in 3 divided doses | - |
| 30-49 | 300-900mg daily in 3 divided doses | - |
| 15-29 | 150-600mg daily in 3 divided doses | 150mg dose to be given as 300mg in 3 divided doses on alternate days |
| <15 | 150-300mg daily in 3 divided doses | 150mg dose to be given as 300mg in 3 divided doses on alternate days |
*Doses based on Creatinine Clearance (CrCl) and not eGFR
| CrCl ml/min* | Maximum recommended dose (BNF) |
|---|---|
| 30-60 | Initially 75mg daily and maximum 300mg daily in 2-3 divided doses |
| 15-30 | Initially 25-50mg daily and maximum 150mg daily in 1-2 divided doses |
| <15 | Initially 25mg once daily and maximum 75mg once daily |
*Doses based on Creatinine Clearance (CrCl) and not eGFR
- Respiratory depression
- Risk of severe respiratory depression noted with both gabapentin and pregabalin78,79
- Individuals at higher risk who may require adjustments in dose or dosing regimen include:
- compromised respiratory function, respiratory or neurological disease
- renal impairment
- those using concomitant central nervous system (CNS) depressants e.g. opioids, benzodiazepines
- people older than 65 years
- Risk of foetal abnormalities
- MHRA noted findings from a study that showed that pregabalin, like some other antiseizure medications, is associated with a slight increased risk of major congenital malformations [92] if used in the first trimester
- Advice for health professionals can be found in the MHRA Pregabalin Drug Safety Update
- More research is required to define the pregnancy safety profile of gabapentin. Pregnant women and women of childbearing potential should be made aware of the lack of data for most pregnancy outcomes. Gabapentin should only be used during pregnancy where benefits of treatment are considered to outweigh any potential risks
| Our recommendations | Strength of recommendation |
|---|---|
| Gabapentin or pregabalin could be considered for individuals with the specific conditions of post herpetic neuralgia or painful diabetic neuropathy44,46 |
Conditional |
| Evidence indicates maximal dose of pregabalin 600mg daily/gabapentin 3600mg daily to achieve 30-50% pain intensity reduction), but if tolerated, minimum effective dose should be used44,90 | Good practice point |
| Gabapentin or pregabalin could be considered for individuals with fibromyalgia, although this is unlicensed46 | Conditional |
| Prescribers and individuals should be aware there is a significant risk of adverse effects with gabapentinoids, including drowsiness, dizziness, nausea, weight changes (abnormal appetite), cognition, speech problems and ataxia83,88 | Strong |
| Gabapentinoids should not be used in lower back pain, sciatica (lumbar radiculopathy) and spinal stenosis45,88 | Strong |
| Carbamazepine could be considered for trigeminal neuralgia, but individuals should be advised of adverse drug effects and interactions89 | Conditional |
| Review effectiveness of any gabapentinoid/carbamazepine after four to six weeks of therapy and discontinue if ineffective | Good practice point |
High risk individual due to high dose opioid, polypharmacy and multiple co-morbidities
A 55-year-old woman with complex regional pain syndrome, multiple co-morbidities and with polypharmacy was identified for review by the Scottish Therapeutics Utility (STU) high dose opiate search and the mental health ‘triple whammy’ search. She has asthma, sleep apnoea, type 2 diabetes, ischaemic heart disease, depression and obesity (BMI 50kg/m2). She is at high risk of opiate toxicity as she is prescribed 300mg of morphine daily and is also prescribed a benzodiazepine, a z-drug, gabapentin and pregabalin, two antidepressants and an antipsychotic.
She has poor engagement with services and frequently does not attend appointments or reviews. Following multiple attempts, she agreed to come in for a pharmacist medication review. Her pain was assessed using the brief pain inventory and her pain scores and functional impact scores were high. She was unsure if her pain medication was effective and recognised that many symptoms that she was experiencing were due to side-effects of medication.
Discussion was had regarding maximum recommended dose of morphine, the cumulative effects of central nervous system medications and the risk of toxicity due to polypharmacy and sleep apnoea. She was signposted to self-help resources and acknowledged the importance of self-management in chronic pain.
Following this consultation she agreed to slowly withdraw her pregabalin. With the support of the pharmacist this has now stopped. She has agreed a tapering plan for her morphine and has currently reduced her total daily morphine dose by 60mg, to 240mg daily, and plans to continue to reduce this. She has built a good rapport with the practice pharmacist and is now engaging with the practice nurse to address other co-morbidities, including control of her asthma and diabetes.
9.8 Other analgesics
Topicals
There are a number of topical agents including rubefacients and capsaicin products. They may previously have had a role in therapy, but there is limited evidence for their clinical value.
Topical Rubefacients
Indications and effectiveness
- Many rubefacients or counter irritants have been identified as items of low clinical value and should not be prescribed on the NHS.
- Rubefacients are topical preparations that may contain nicotinate compounds, salicylate compounds, essential oils, and camphor. They cause irritation and reddening of the skin due to increased blood flow.
- They have been used to relieve pain in musculoskeletal conditions, however this is not supported by the most recent published evidence.
- Menthol e.g. menthol in aqueous cream can give additional benefit, cooling the skin for several hours. This is available in a variety of strengths.
- Individuals can be signposted to non-pharmacological heat and cold therapies, such as hot water bottles, microwavable wheat bags and reusable gel warmers, icepacks and cooling gel packs.
Risks and harms
- Adverse effects are usually limited to localised reactions such as dry skin, reddening, burning sensations and contact dermatitis. Individuals may be advised to test on a small area of skin first.
Capsaicin
Indications and effectiveness
- Capsaicin products have a limited place in therapy.[93] See licensed indications in Table 13.
- Capsaicin reduces pain messaging to the spinal cord and to the brain, by desensitising sensory (nociceptive) nerve fibres.
- Capsaicin is available as a cream or as a cutaneous patch (see Table 13 for licensed indications).
- Patch use is generally reserved to specialist clinics as there are specific application and monitoring requirements.
- Individuals may or may not feel the warming sensation on application and should be advised this is not linked to efficacy of the product.
| Product | Indication | Dose | NNT |
|---|---|---|---|
| 0.025% Cream | Osteoarthritis | Apply three to four times daily not more often than every four hours | Not known |
| 0.075% Cream |
- Post herpetic neuralgia - Painful diabetic neuropathy |
Apply three to four times daily not more often than every four hours | Not known |
| 8% cutaneous patch | - Peripheral neuropathic pain | Applied under specialist supervision | 11-12[94] |
Risks and harms
With creams:
- Individuals should be advised to wash hands immediately after use and avoid hot showers or baths just before application.
- Localised burning sensation if too much applied or applied too frequently.
- Some people report increased cough and they should be advised to report any exacerbation of pre-existing asthma conditions.
Nefopam
Indications and effectiveness
- Nefopam is a non-opioid, centrally-acting analgesic. The mechanism of action is not well understood. It is known to inhibit the re-uptake of neurotransmitters including serotonin, noradrenaline and dopamine
- There is limited clinical evidence available for the effectiveness of nefopam in the treatment of chronic pain as outlined in Scottish Government guidance on medicines of low and limited clinical value
- Nefopam may be considered for specific individuals,[95] when choice of alternative analgesia is limited due to other co-morbidities e.g. people with renal failure
- Particular caution should be used in adults who would be at elevated risk of anticholinergic side effects e.g. older adults or in cognitive impairment
- There is no evidence that nefopam is more potent than non-steroidal anti-inflammatory drugs (NSAIDs), but is commonly associated with adverse drug reactions and is toxic in overdose95,[96]
Risks and harms
- Common side-effects include: confusion, nausea, nervousness, urinary retention, dry mouth, light-headedness. It is toxic in overdose
- Nefopam scores two on the anticholinergic burden scale (ACB) therefore sympathomimetic and antimuscarinic side-effects may be troublesome
- Confusion and urinary retention are a particular problem in the elderly
The side effects of nefopam can be cumulative with other anticholinergic medications see Scottish Government Polypharmacy Guidance.
Lidocaine plasters
Indications and effectiveness
- Lidocaine plaster use should be limited to licensed indications for pain relief.
- This is symptomatic relief of neuropathic pain associated with previous herpes zoster infection (post herpetic neuralgia) in adults (see Scottish Medicines Consortium advice). Its use is restricted to individuals who are intolerant of first-line systemic therapies or where these therapies have been ineffective.
- The place in therapy of lidocaine medicated plasters is currently unclear. Evidence supporting both their licensed use in post herpetic neuralgia and in other unlicensed indications is limited.
- NICE guidance on chronic pain does not recommend lidocaine plasters for treating neuropathic pain.
Risks and harms
- Caution in use in severe renal impairment.
Medicinal cannabis
Indications and effectiveness
Cannabis-based medicinal products (CBMP) should not be prescribed for chronic pain due to the low numbers of NNH (6) and the high number of NNT (24). [97]
CBMPs have been shown in some systematic reviews to have a low to moderate effect on chronic pain.[98], [99], [100], [101], [102], [103], [104], [105], [106] However, the studies included in these reviews have often been considered low quality and the effect shown has not been considered clinically important.
Risks and harms
Participants receiving CBMPs were more likely to experience side effects compared with placebo. These included anxiety, attention disorder or deficit, confusion, delusions, depression, suicidal thoughts, disorientation, dizziness, euphoria, falls, hallucinations, mania, memory impairment, nausea, paranoia, sedation, diarrhoea and vomiting.
Although trials of CBMPs in pain have shown limited serious adverse effects the trials are usually of short duration. High-quality harms data from long-term controlled studies for chronic non-cancer pain are lacking.
Methocarbamol
Indications and effectiveness
- It is licensed for short term symptomatic treatment of acute painful muscular tension, especially in the lower back in adults.
- Methocarbamol is a centrally acting muscle relaxant which does not directly relax tense skeletal muscles.
- There are few high-quality studies and no meta-analyses comparing methocarbamol to placebo or alternative agents for muscle spasms.
- It is less suitable for prescribing due to clinical efficacy not being well established.
Risks and harms
Side effects include: confusion, convulsions, drowsiness, restlessness, anxiety, hypotension, vertigo, nausea and vomiting.
- If methocarbamol is used concomitantly with centrally acting drugs such as barbiturates, opioids and appetite suppressants, the effects can be mutually enhanced
- The consumption of alcohol during treatment with methocarbamol can increase the effect
- The effects of anticholinergics, such as atropine and some psychotropic drugs, may be potentiated by methocarbamol
- Methocarbamol may reduce the effect of pyridostigmine bromide. Therefore, methocarbamol should not be used by people with myasthenia gravis, particularly those being treated with pyridostigmine
Benzodiazepines
Indications and effectiveness
- There is no indication in chronic pain, specifically:
- Sciatica (lumbar radiculopathy): benzodiazepines should not be used
- chronic primary pain in people aged 16 years and over: NICE recommends ‘do not initiate benzodiazepines to manage this’
Risks and harms
- Adverse events are considered as cognitive (memory loss, confusion, disorientation), psychomotor (reports of dizziness, loss of balance, or falls); and morning hangover effects (residual morning sedation)
- The number needed to harm for any adverse event is 6
- Older adults and frail people are more susceptible to adverse effects and harms
- Increased risk of respiratory depression if prescribed with an opioid
- Consider the total benzodiazepine-type medicine load prescribed per day – as benzodiazepines, z-drugs and gabapentinoids provide synergistic effects such as sedation and respiratory depression
| Our recommendations | Strength of recommendation |
|---|---|
| Topical rubefacients and counter irritants should not be prescribed on the NHS, having been identified as items of low clinical value | Strong |
| Nefopam may be considered for specific individuals, when choice of alternative analgesia is limited due to other co-morbidities e.g. those with renal failure, accounting for elevated risk of adverse effects, especially anticholinergic | Good practice point |
| Lidocaine plaster use should be limited to licensed indication of previous herpes zoster infection (post herpetic neuralgia) in those who are intolerant of first-line systemic therapies or where these therapies have been ineffective, as it has been identified as an item of limited clinical effectiveness | Strong |
| Medicinal cannabis-based products are not recommended for routine use in the management of chronic pain, due to very limited evidence of clinically significant improvements in pain or wider impact and the consistent evidence of adverse effects/ harms97,98,99,100,101,102,103,104,105,106,[107] | Conditional |
| Methocarbamol should not be used in the management of chronic pain - it is considered less suitable for prescribing due to lack of clinical efficacy and increased risk of adverse effects and interactions | Conditional |
| Benzodiazepines should not be used in chronic (primary) pain18 | Strong |
9.9 How medications should be stopped
Reduction of therapy should be controlled, not sudden, to manage the risk of withdrawal symptoms.
Successful reduction requires shared decision-making and a person-centred review that assesses risk and benefit before reducing or stopping medication.
9.10 Safety - specific medication safety issues to highlight
Opioid use is receiving increasing attention, with opioid deaths in Scotland and the USA a major cause of public concern:
- Media coverage may raise specific concern about drugs which are in everyday use, such as fentanyl
- Individuals prescribed opioid medication should understand that many of these deaths are associated with unregulated products (with potentially very high doses of drug) and illicit drug use – and should understand their own situation in context
- National Records of Scotland shows that there were 1,017 deaths due to problematic drug use in Scotland in 2024[108] Of these deaths, medication implicated included:
- opiates/opioids (such as heroin/morphine and methadone): 810 deaths (80%)
- benzodiazepines (such as diazepam and bromazolam): 574 deaths (56%)
- gabapentin and/or pregabalin – 374 deaths (37%)
Generally:
- Potentially fatal interactions between medicines that cause CNS depression, particularly opioids, gabapentinoids and benzodiazepines
9.11 Specific areas/situations where medication may need to be introduced or increased
- If the presenting pain condition changes or symptoms worsen, undertake a further pain assessment, considering potential cause of flare-up and consider appropriate management (which may include rest, heat packs, pacing, use of short-term NSAID), dependent on the cause of the presenting pain.
- Antidepressants:
- Before starting or continuing treatment with an antidepressant, ensure that all suitable management options, including non-pharmacological approaches and watchful waiting, have been discussed and offered. Explain and discuss the risk of dependence and the factors that may increase this risk.
- If treatment is reviewed and the individual has noticed an improvement in their quality of life, pain, sleep or psychological distress since the antidepressant was initiated, then a joint decision with the individual should be made whether continued treatment is needed. Always ensure ongoing review is in place to determine the benefits of continued prescribing.
- Consider delaying prescribing if the person needs more time to think about their options or the prescriber needs to consult with other members of the healthcare team. If prescribing is delayed, ensure that a follow-up appointment and timeframe is agreed and arranged.
9.12 Polypharmacy in chronic pain
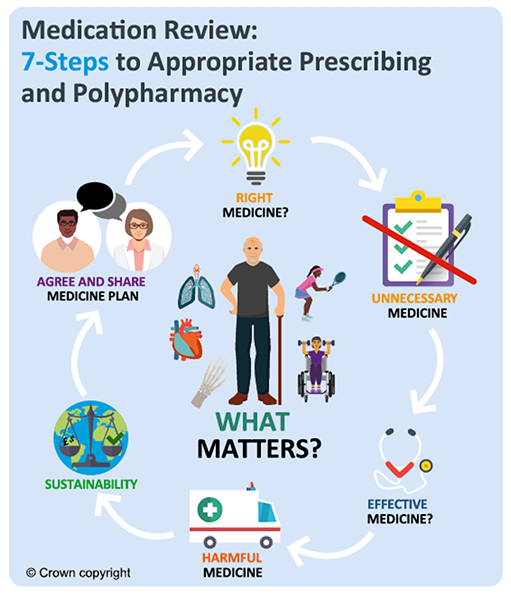
Polypharmacy is common in those with chronic pain due to other co-morbidities. A person-centred approach should be considered, utilising the 7-Steps medication review process.
A high-risk combination which increases the risk of sedation, falls and the potential for dependency is three or more of:
- opioid
- antidepressant
- gabapentinoid
- benzodiazepine/z-drug
- antipsychotic
Prescribers should consider the total benzodiazepine-type drug load prescribed per day. Benzodiazepines, z-drugs and gabapentinoids have similar adverse effects such as sedation and respiratory depression. Along with this, an individual’s comorbidities may make them more susceptible to medicine-related harms e.g. increased breathlessness, fatigue and respiratory depression, which can be potentially fatal.
Reduction should be gradual. Stopping any of these medicines abruptly can cause discontinuation/withdrawal symptoms, such as dizziness, drowsiness, insomnia, irritability, headache, nausea and vomiting.
9.13 Specific situations to consider
Pregnancy and pre-conception advice
- Medication can affect both conception and foetal development.
- For most medication, the effects on conception and foetal development are not well understood. It is crucial to have open discussions with women of childbearing age before initiating or prescribing medication and weigh the benefits against the risks. Preconception planning should include a medication review for those on long-term pain medication.
- Pregabalin can impact foetal development,92 and similar considerations may apply to gabapentin.
For additional guidance, refer to:
- British National Formulary (BNF)
- UK Teratology Information Service (UKTIS)
- Bumps - Best use of medicines in pregnancy
Menopause and Hormone Replacement Therapy (HRT)
- Muscle aches and pains are common during perimenopause and menopause [109]
- In women aged 45 – 60 years presenting with new musculoskeletal pain consider perimenopause/ menopause as the cause and whether a trial of HRT is appropriate109
Renal and Hepatic Function
- Pain medication is metabolised either by the kidneys or the liver. Some medicines, such as codeine, are prodrugs that require metabolic conversion to an active form to be effective. Impaired renal or liver function can increase the risk of toxicity or reduce the effectiveness of certain pain medication.
- NSAIDs, gabapentinoids and opioids may need adjustment of their doses or may be contraindicated in cases of significant renal or hepatic dysfunction.
- check whether dosage adjustments are based on estimated glomerular filtration rate (eGFR) or creatinine clearance, for example gabapentin (see licence). For severe impairments, seek specialist advice.
Older adults and frailty
- Older adults often have multiple co-morbidities, higher levels of polypharmacy and increased sensitivity to commonly used medicines. This increases the risk of drug interactions as well as adverse effects.
- Older adults may have weight loss and/or weigh less than 50kg, affecting prescribed dosages.
- Older adults may be impacted by impaired renal or hepatic function, or medicines affecting the central nervous system. This can lead to increased risk of toxicity and adverse effects such as falls.
- Use of medication with strong or very strong anticholinergic effects increases the risk of adverse effects, both cognitively as delirium, or in the peripheral effects such as blurred vision, urinary retention. Multiple medications compound this problem.
- General guidance for prescribing in older adults is in the BNF- Prescribing in the elderly. Scottish Government Polypharmacy Guidance also contains detailed sections on prescribing in frailty and for older adults.
- Recommended actions:
- Simplify regimens
- Rationalise medication as appropriate
- Reduce dose in line with hepatic or renal function
- Minimise anticholinergic burden using calculators such as mARS calculator
Problematic substance use
- Many individuals with problematic substance use may also suffer from chronic pain and require treatment with pain medication.
- The Faculty of Pain Medicine provides guidance for acute pain management.66
- Shared decision-making is crucial when standard analgesics are insufficient and medication with dependency risks is being considered.
- Establish clear treatment goals and develop a strategy for discontinuation if clinical benefits are not realised.
- Non-pharmacological strategies should be discussed.
- Be aware of the risks associated with not prescribing, such as individuals obtaining illicit substances to manage their pain, which may increase the risk of accidental overdose. Seek advice from specialist services, including addiction services.
- Consider prescribing naloxone (see section on naloxone).
9.14 Practitioner experience
Practitioner’s experience – Moving from novice to expert
“I started running a pain clinic two days a week approximately two years ago. I felt apprehensive about taking on this role for many reasons. Chronic pain is a complex condition to manage, there is rarely a cure and the symptoms are unpleasant. I felt that consultations were often going to involve difficult conversations, and individuals might not be willing to engage with me. Medication changes could worsen pain when I want to make people feel better, not worse. Did I have enough knowledge in the area to take on this patient cohort?
Often these people feel isolated, not listened to, unbelieved and unsupported. I was pleasantly surprised by how appreciative they were of my reviews. Feedback included:
‘Nobody has taken time to talk me about my condition and medication like this before’
‘Coming off pregabalin has changed my life; I wish I met you years ago’,
‘Thanks for listening and giving me the confidence to trial changing my medication’
and the most commonly heard phrase:
‘I feel more like me again, now that I have reduced my medication’.
Most people feel anxious at the prospect of reducing their medication. However, I always give them full control of any changes, going at their pace, pausing if needed and increasing the dose back to the previous dose if pain worsens (but this has only happened a few times). I remind the individual that if they never trial any changes then they will never know if they could manage on a lower dose or even without the medication completely.
The majority of people with chronic pain have received little or no information on self-management – you don’t need to be a specialist in this area to provide them with resources.
Helping them to reduce the impact of pain on their quality of life by optimising medication and introducing self-management strategies is extremely rewarding.”
Contact
Email: EPandT@gov.scot