The management of wild deer in Scotland: Deer Working Group report
The final report of the Deer Working Group.
Part of
Section 10 Wild Deer and Diseases
1 Wild deer in Scotland and the rest of the UK are susceptible to a range of animal diseases. These diseases are classed as either endemic or exotic to the UK. Some are also classified as zoonotic, meaning that the disease can be transmitted from the animals to humans. Wild deer are also host to a number of internal and external parasites, some of which can transfer to humans and can affect human health either directly or indirectly by transmitting a disease.
2 Wild deer in Scotland have generally been considered to have a relatively low level of disease.[1] However, there has long been attention given to the occurrence of diseases in wild deer. This is because of the possible impacts of the diseases on the deer themselves and the risks of the transmission of diseases to and from farm livestock (including farmed deer) or other wildlife sources and, in some cases, to humans. As a result, there are statutory provisions and government procedures covering some diseases, as well as practical guidance available to help those involved in managing deer and handling deer carcases to recognise the symptoms of diseases.[2]
10.1 Notifiable and Non-Notifiable Diseases
10.1.1 Notifiable Diseases
3 The principal legislation covering animal diseases is the Animal Health Act 1981, with the Act and secondary legislation under the Act listing Notifiable Diseases where there is a legal requirement to report the disease. The occurrence or suspected occurrence of one of the Notifiable Diseases has to be reported to the government body responsible, the Animal and Plant Health Agency (APHA).
4 APHA is part of the UK Department for Environment, Food and Rural Affairs (DEFRA) and acts for the devolved Scottish and Welsh administrations for their devolved powers over these matters. More detail about some Notifiable Diseases that can affect wild deer are listed in Annex 7, including Bovine Tuberculosis, Foot and Mouth, Chronic Wasting Disease (CWD), Warble Fly, Bluetongue Virus (BTV) and Epizootic haemorrhagic disease. There are others, for example Anthrax, which is highly lethal to mammals but not currently present in the UK.
5 Several factors make diseases in deer an increasingly important topic for the management of wild deer in Scotland. A key reason is that the expansions in deer species ranges and numbers in Scotland mean that there is generally more proximity between wild deer and livestock (including farmed deer) in many parts of the country, increasing both the scope for transmissions between them and the potential for deer populations of one or more species acting as disease reservoirs.[3]
6 Climate change can also be a factor in increasing the risks of disease by improving the conditions for disease vectors. With BTV, for example, while its occurrence in Scotland in 2017 was the result of cattle imported from France via England, the spread of BTV in continental Europe has been associated with a northwards expansion due to climate warming of the midge species that transmits it.[4]
7 The prominent new disease threat to wild deer is CWD. It affects the nervous system of deer, is highly infectious and has no known treatment or vaccine. While CWD only appears to affect deer species (Cervids), it has been causing severe losses to many farm and wild deer populations in North America. A major concern was the discovery in 2016 of the first cases of CWD in Europe in reindeer and moose in Norway.[5]
8 The CWD infected reindeer in Norway were in the Nordfjella mountains where the Norwegian Government has culled the entire local reindeer population of nearly 1,500 animals, confirming 18 cases of CWD.[6] While the CWD in the reindeer was similar to the type prevalent in North America, the CWD in three moose in Norway were a different type. The same has been the case with a moose with CWD recorded in 2018 in Finland.[7] All four moose were older animals and it seems that this type of CWD may occur sporadically and spontaneously.[8] Since then, no further cases of CWD have been reported from Norway.
9 Surveys are now underway in many European countries to monitor for CWD. In Scotland, the Scottish Deer Health Survey 2017-19 is being carried out to determine the prevalence of three diseases in Scotland’s wild deer populations: CWD, E.coli 0157 and Cryptosporidium.[9] The survey is funded by the Scottish Government and the government agency Food Standards Scotland (FSS), and is being undertaken by the Moredun Research Institute and University of Edinburgh.
10 No results have been published at the time of writing for the screening of deer for CWD by the Deer Health Survey. However, if the North American type of CWD was to start to become established in Scotland, it could have a severe impact on wild deer populations through both the disease and control schemes designed to contain it. There could also be a major impact on the venison market due to customer concern.[10]
10.1.2 Non-Notifiable Diseases
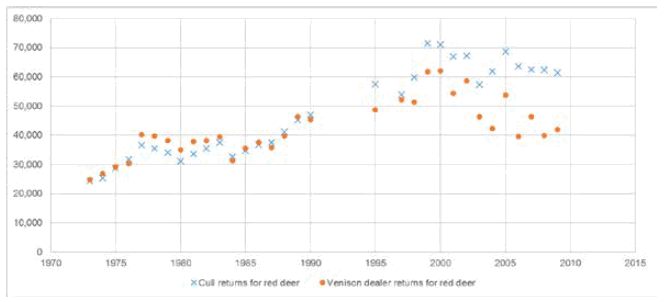
11 The other two diseases in the current Deer Health Survey are not notifiable diseases, but each represents a significant risk to human health. E.coli 0157 is a bacterium, while Cryptosporidium is a parasite. Both occur in deer faeces and can be transmitted to humans mainly through contaminated meat and water respectively. Each can cause serious human illness through gut infections.
12 While it has been suggested in the past that the prevalence of harmful E.coli in wild deer may be low, there has been increased attention paid to it following an outbreak in Scotland in processed wild venison products in 2015.[11] In that year, there was also a study published that showed a very high incidence of Cryptosporidium in wild deer in part of the Cairngorms National Park, which concluded that this posed a significant risk for public safety.[12]
13 The initial results from the Deer Health Survey have confirmed a very low prevalence of harmful E.coli 0157, with only 0.3% of the faecal samples testing positive.[13] However, the levels in the positive samples were very high and the results have emphasised the need for strict hygiene precautions when processing deer carcases to avoid faecal contamination.[14] No results have been published yet for the incidence of Cryptosporidium.
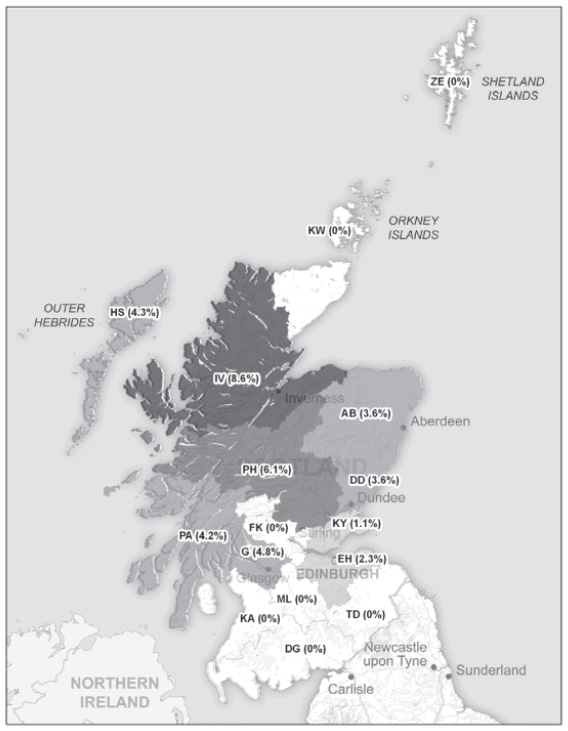
14 There are also a number of other disease organisms that can present a significant risk to someone improperly handling deer carcases (for example, liverfluke, tapeworm). The importance of the market for wild venison to deer management in Scotland and the importance of food safety to that market, are discussed in the next Section of the Report.
10.2 Lyme Disease
15 Amongst the disease vectors hosted by wild deer in Scotland, ticks are particularly significant. Ticks may be infected with Borrelia burgdorferi bacteria and transmit that to humans, resulting in Lyme disease (Lyme borreliosis). While Borrelia burgdorferi is the species responsible for Lyme disease, it consists of different strains and genospecies.[15] The species is therefore often described as the B.burgdorferi complex or B.burgdorferi sensu lato (in the broad sense).
16 Lyme disease is the most common zoonotic disease transmitted by ticks in Europe and North America.[16] In Europe, Lyme disease is mainly carried by the sheep or deer tick (Ixodes ricinus). In the UK, in addition to Ixodes ricinus, Lyme disease is carried by the hedgehog tick (I.hexagonus), the seabird tick (I. uriae) and the fox or dog tick (I.canisuga), although these tick species are more host-specific and pose little threat to humans.
17 Lyme disease is of growing importance in Scotland as the number of people in Scotland affected by Lyme disease is continuing to increase. This is reflected in the results from Scotland’s National Lyme borreliosis testing laboratory in Inverness in Figure 22. Similar statistics are also produced by Health Protection Scotland, the national surveillance centre for communicable diseases and health problems associated with environmental hazards.[17]
18 Official statistics for confirmed new cases of Lyme disease in Scotland are recognised to underestimate considerably the full extent of cases, with the true extent considered to be several times greater.[18] This is a result of under-diagnosis due to the difficulty of diagnosis as the symptoms are non-specific, and under-reporting as Lyme disease is no longer a Notifiable Disease.[19]
19 If Lyme disease is treated early, there is more chance of a person recovering to full health. However, if left untreated it can become a chronic, debilitating and disabling condition. As full recovery may not take place in some cases, the total number of people affected is accumulating. The increasing number of cases is leading to growing awareness of the disease in Scotland and to more media coverage.[20]
20 The prevalence of the disease in different parts of the country varies. This is illustrated by the results of a study of blood samples from 1,440 blood donors in 2010/11, in which 4.2% of the samples tested positive for antibodies for the Borrelia bacteria that causes Lyme disease.[21] The distribution of the positive test results were then mapped by the donors’ postcodes as shown in Figure 23.
21 Wild deer in Scotland are a major host for ticks. This is due to the relative abundance of deer and their large size, with deer carrying heavy tick loads in some circumstances. However, while deer can increase the size of local tick populations, deer themselves do not carry or transmit the Borrelia bacteria that cause Lyme disease. Thus, a tick feeding on a deer cannot become infected and subsequently transmit that infection to a human or other host. As deer are not a transmission host, they are referred to as a non-competent host for Borrelia.
22 Research has shown that there is a clear correlation between increasing deer densities and increasing tick populations, as the deer provide an important feeding and reproduction host.[22] However, this increase in ticks does not lead directly to an increase in ticks infected with Borrelia as deer are not a transmission host. This has led to suggestions that there can in theory be a ‘dilution effect’, due to the number of ticks that are not infected with Borrelia increasing faster than a minority of the tick population that are infected.[23]
23 The suggested ‘dilution effect’ might also be produced in some situations by other wildlife species that are not competent transmission hosts.[24] However, this effect does not reduce the absolute densities of Borrelia infected ticks, it only reduces them as a proportion of the tick population. A critical variable in any situation is the extent to which there are competent transmission hosts present. If there are transmission hosts in an area for the increased number of ticks to feed on, the overall number of infected ticks will increase.
24 Competent transmission hosts in Scotland include birds and rodents, with the latter including voles, mice, rats, squirrels and beavers. Sheep can also transmit Lyme disease through their skin between co-feeding (close-together) ticks.[25] While the extent to which dogs might be competent transmission hosts is not known, they become ill with Lyme disease and researchers in the USA have been testing dogs as a proxy for humans in trying to understand the spread of Lyme disease.[26] More generally, however, there appears to be a lack of information on the full extent of competent host species in Scotland including, for example, whether rabbits and hares can be transmission hosts.
25 The evidence described above shows clearly that more deer means a greater density of ticks, while the prevalence of Borrelia amongst the ticks will be influenced by the availability of other transmission hosts. However, there are no studies that show that more deer results in a lower risk of Lyme disease to people.[27]
26 Wild deer and ticks both occur in a wide range of environments in Scotland and the level of Lyme disease risk to people can vary in different circumstances. A tick population with a certain level of infected ticks may pose a lower risk in a rural area with relatively low potential for human contact with ticks, than a tick population with the same prevalence in urban green spaces with much greater potential for human contact with ticks.[28]
27 The colonisation of Scotland’s peri-urban and urban environments by deer is thus an important factor in the spread of Lyme disease. The times of greatest tick activity are spring and summer and these can also be periods of higher human activity in urban green spaces such as parks. A study in an English city found an 18% level of infected ticks in the green spaces sampled, in comparison to the studies they examined of rural habitats in the UK where the prevalence ranged from 3-8%.[29] The city study suggested that there could be a need in some urban parks to have warning notices about ticks and the risk of Lyme disease.
28 Deer can, like other host species, spread ticks between green spaces so that ticks tend to occur in all green spaces where there is suitable vegetation and sufficient host species.[30] This can include domestic gardens where ticks drop off after a reproduction feed and become established, posing a risk to pets as well as to people with the level of risk of Lyme disease determined by the density of infected ticks.
29 The average prevalence of infected ticks in Scotland is considered to be 2-6% as a result of two major studies.[31] This average figure is lower than the prevalence rates reported in mainland Europe.[32] However, prevalence rates of up to 20% have been found in a small number of localised areas in Scotland and prevalence rates can vary both seasonally and year to year in any one location.[33] The important factor for the risk of Lyme disease is the density of infected ticks in an area, rather than simply the prevalence of infected ticks in the tick population.
30 There is ongoing research into the relationship between deer and Lyme disease, so that the position can be understood more fully.[34] While it is clear that increasing deer density means a greater density of ticks, the relationship between deer densities and the risk of Lyme disease is not linear. This is due to other variables, such as the influence of the abundance of other transmission hosts on the density of infected ticks in any situation and the likelihood of human-tick contact in that situation.
31 Scottish Natural Heritage (SNH) briefly described Lyme disease in its 2016 Report on Deer Management and referred to some previous attempts to attribute costs to the number of cases of the disease.[35] While there appear to be no recent or reliable estimates for the financial costs of Lyme disease in Scotland each year, these will continue to rise with the increasing number of cases.
32 SNH has not done any direct work on the relationship between deer, ticks and Lyme disease, and the role that deer can play in the risk of the disease.[36] The Group considers, however, that the expanded ranges and increased numbers of wild deer in Scotland are likely to be a major factor in contributing to ongoing rises in the cases of Lyme disease. The Group considers that the risk of Lyme disease should be seen as a more important factor than currently in the need to reduce deer densities in locations, particular in those areas where there is likely to be greater chance of human-tick contact.
33 The Working Group recommends that the Scottish Government should ensure that the role of wild deer in increasing the risk of Lyme disease is given greater prominence in its policies for deer management in Scotland, and that greater priority is given to that risk in considering the need to reduce deer densities in locations across Scotland.
10.3 Monitoring Deer Health
34 There are now estimated to be well over 750,000 wild deer spread throughout mainland Scotland and some islands. The greater numbers of deer and the greater proximity between deer and both livestock and people compared to earlier decades, add to the risk of existing or new diseases being transmitted. The Group therefore considers that it is important to monitor deer health at a national level for diseases, due to their potential impacts on people, livestock and the deer themselves.[37]
35 In the past, there has been a history of monitoring different aspects of the health of wild deer to examine the risks to the deer, livestock and people. The results of these studies were included in the Deer Commission for Scotland’s Annual Reports, and before that the Red Deer Commission’s Reports.[38] The current Scottish Deer Health Survey described above continues that history of monitoring by examining three risk factors. The Group considers that monitoring for different components of the disease risk should continue on a planned basis.
36 The Working Group recommends the Scottish Government and its agencies should, following the current Scottish Deer Health Survey, develop and maintain an ongoing national programme to monitor wild deer in Scotland for existing and potential diseases.
37 The Group considers that the risk or occurrence of diseases is likely to become a more significant factor in the management of wild deer in Scotland. While monitoring deer health is important, there also needs to be adequate information available to deal with a disease outbreak.
38 Researchers have highlighted the need in this context for adequate information on deer culls and the use of venison.[39] The Group considers that Scotland is ill-prepared on both those requirements. Reference has been made previously to the limited deer cull information available for many parts of Scotland.[40] The limited information available on what happens to the carcases of the deer culled each year is discussed in the following Section of the Report.
39 Important components of any strategy to monitor for diseases and also to protect food safety, are adequate awareness, education and training amongst practitioners to aid early identification of disease symptoms and ensure appropriate hygiene standards. There is Wild Deer Best Practice guidance available on these and they are covered by the Deer Stalking Certificate Level 1 (DSC1) qualification. However, as discussed earlier, there is no requirement to have any level of training to be able to shoot deer in Scotland and no information on the proportion of the cull each year that might be shot by someone with the DSC1 qualification.[41]
Footnotes
1 The Wild Deer Best Practice (WDBP) guidance on ‘Deer Health’ states that “Wild deer tend to be remarkably free of disease.”
2 For example, WDBP guides on ‘Deer Health’ and ‘Notifiable Diseases’.
3 Böhm, M., White, P.C.L., Chambers, J., Smith, L and Hutchings, M.R. (2007). Wild deer as a source of infection for livestock and humans in the UK. The Veterinary Journal, 174, pp.260-276.
4 Falconi, C., Lopez-Olvera, J.R. and Gortazar, C. (2011). BTV infection in wild ruminants, with emphasis on red deer: a review. Veterinary Microbiology, 151, pp. 209-219.
5 British Deer Society website, ‘CWD in Norway’, 16 November 2017.
6 Ministry of Agriculture and Food website (Norway), ‘Chronic Wasting Diseases: All known animals in Nordfjella dispatched’, 27 February 2018.
7 Norwegian Veterinary Institute website, ‘CWD in Finland is different from the Nordfjella CWD type’, 8 March 2018.
8 Norwegian Veterinary Institute website, Op cit.
9 ADMG website, ‘Scottish Deer Health Survey 2017-19’, 1 September 2017.
10 For example, as with ‘mad cow disease’.
11 ADMG website, Op cit.
12 Wells, B., Shaw, H., Hotchkiss, E., Gilray, J., Ayton, R., Green, J., Katzer, F., Wells, A. and Innes, E. (2015). Prevalence, species identification and genotyping Cryptosporidium from livestock and deer in a catchment in the Cairngorms with a history of a contaminated public water supply. Parasites & Vectors, 8, p.66.
13 Scottish Deer Health Survey 2017-19: STEC 0157 Results (Moredun Research Institute, December 2018).
14 Scottish Deer Health Survey, Op cit.
15 Dr Lucy Gilbert correspondence with DWG, 14 June 2019.
16 Strnad, M., Hönig, V., Růžek, D., Grubhoffer, L. and Rom, R. (2017). Europe-Wide Meta-Analysis of Borrelia burgdorferi Sensu Lato Prevalence in Questing Ixodes ricinus Ticks. Applied and Environmental Microbiology, 83(15).
17 Dr Lucy Gilbert, Op cit.
18 Dr Lucy Gilbert, Op cit.
19 It was removed from the list of Notifiable Diseases in Scotland in 2010, when the criteria for Notifiable Diseases were tightened to diseases that require urgent action.
20 BBC Scotland, ‘Disclosure: Under the Skin’, first broadcast 17 June 2019.
21 Munro, H., Mavin, S., Duffy, K., Evans, R. and Jarvis, L.M. (2015). Seroprevalence of lyme borreliosis in Scottish blood donors. Transfusion Medicine, 25(4), pp.284-286.
22 Dr Lucy Gilbert, ‘Do deer increase Lyme disease risk?’, presentation shared with DWG, March 2018.
23 Dr Lucy Gilbert, Op cit.
24 Entomology Today website, ‘Why the variety of ticks in your back yard might be a good thing’, 12 January 2018.
25 Dr Lucy Gilbert correspondence with DWG, 17 June 2019.
26 Liu, Y., Nordone, S.K., Yabsley, M.J., Lund, R.B., McMahan, C.S. and Gettings, J.R. (2019). ‘Quantifying the relationship between human Lyme disease and Borrelia burgdorferi exposure in domestic dogs. Geospatial Health, 14(1), p.750.
27 Dr Lucy Gilbert correspondence with DWG, 16 March 2018.
28 Hansford, K.M., Fonville, M., Gillingham, E.L., Coipan, E.C., Pietzsch, M.E., Krawczyk, A.I., Cull, B., Spring, H. and Medlock, J. (2017). ‘Ticks and Borrelia in urban and peri-urban green space habitats in a city in southern England. Ticks and Tick-borne Diseases, 8(3), pp.353-361.
29 Hansford et al. (2017) Op cit.
30 Hansford et al. (2017) Op cit.
31 Dr Lucy Gilbert correspondence with DWG, 14 June 2019.
32 Strnad et al. (2017), Op cit.
33 Dr Lucy Gilbert, Op cit.
34 For example, involving the James Hutton Institute and the University of Glasgow.
35 SNH (2016). Deer Management in Scotland: Report to the Scottish Government from SNH, October 2016.
36 DWG meeting with SNH, 19 June 2019.
37 As recommended, for example, by Böhm et al. (2007), Op cit.
38 For example, the DCS reported in 2002/03 on a review of ‘Diseases of Deer relevant in Scotland’, while in 2003/04 the DCS reported on ‘An Analysis of Key Endoparasites and Recommendations for Monitoring Disease’.
39 For example, see Holland, J., Mc Morran, R., Morgan-Davies, C., Bryce, R., Glass, J., Pollock, M., McCracken, D., Glass, R., Woolvin, A. and Thomson, S. (2017). Meeting the challenge of wild deer research to support delivery of sustainable deer management in Scotland. SNH Commissioned Report No.963.
40 See Section 2.
41 See Section 8.
Contact
Email: brodie.wilson@gov.scot