Tuberculosis - RNOH/GIRFT review: national report
The GIRFT Tuberculosis (TB) report is a comprehensive, data-driven national review of TB services across Scotland.
9 Organisational support and infrastructure: Policy and Planning
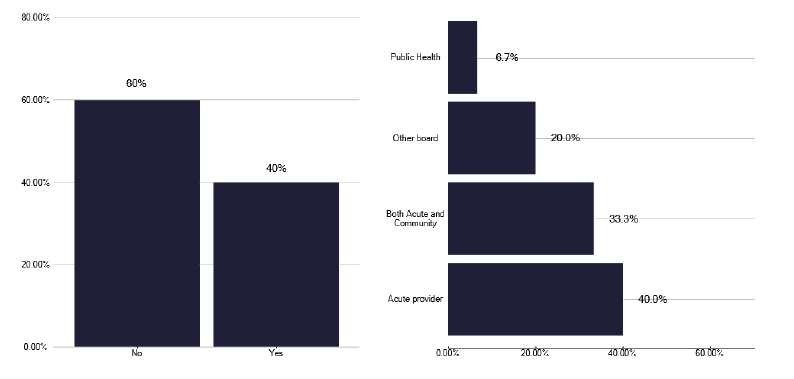
As part of the survey, which was enhanced by information gathered in the deep dives; we explored the underlying policies for managing TB. We noted that 60% of services did not have a local TB policy or specification and when such a policy did exist it was more commonly developed by the acute provider, with only a third of such policies being developed between acute and community, as shown in Figure 10.
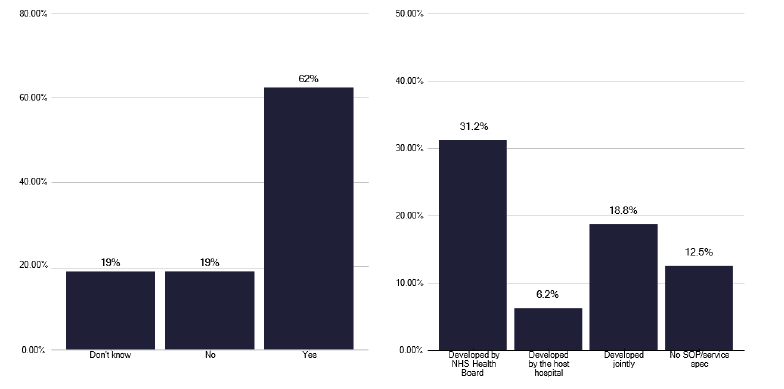
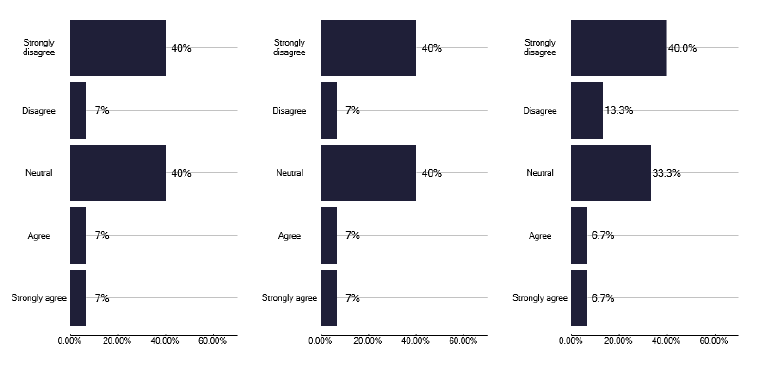
A similar question on whether there was a formal specification as in Figure 11, showed that 62% of services had a policy which was often developed by the Health Board, suggesting there is some inconsistency in the nature of existing policies. Unfortunately, 80% of services did not meet regularly with the management service to discuss service development and funding, and we made recommendations for this to commence during several deep dives. This aspect is important, as the majority of services were split between not having a view on funding or disagreeing that funding was adequate as shown in Figure 12.
Policies for DOT, based upon good practice, were only present in 38% of services but this may reflect the relatively low volume services or those based in urban areas, where patient adherence may not be an issue, and so DOT is not required. We did however identify good practice for DOT using community pharmacists.
Surprisingly 38% of providers said that there was no internal MDT to discuss clinical issues, through this may reflect the services where there are few notifications.
A preventative strategy does not formally exist in Scotland, where 80% of providers said that they did not have a standard operating procedure for several areas. With regard to oversight and supporting services, 87% said that they did not participate in cohort reviews. One service had undertaken a cohort review, which was in fact a local review of the recent increased number of notifications and they did not formally review results from genome sequencing, as shown previously in Figure 4.
Recommendations: Policy
14. Regular meetings between management in acute providers, which would also include Health Protection, Public Health and the Health Board, are an important way of maintaining the profile and supporting funding. Based upon the number of notifications and preventative therapy, these meetings should take place at least on a quarterly basis.
P, HB
15. TB services should meet internally with their management team to discuss practical service delivery issues, the frequency dictated by activity. This time should be built into job plans.
P, HB
9.1 Data Management
Understanding real time issues within TB services is a key aspect of running an efficient service. While surveillance tools such as ESMI and the planned migration to NTBS are helpful, they do not provide ‘real time’ information for managing patients. A more robust clinical management system that can help manage patient flow through the service, generates letters to clinicians and pulls information into national surveillance datasets like ESMI and NTBS, needs to be developed.
The migration of ESMI into NTBS, which has additional data fields, is helpful but gaps may remain in data completion and absence of some fields, as noted in the England TB report. As noted in the workforce section 7, there is a scarcity of administrative staff in TB services to enable this data entry to occur, which may result in data gaps or the need for higher banded Health Protection nurses or TB nurses to spend even more time inputting data. This is a poor use of an expensive and skilled workforce.
Despite the limited resources, we noted one Health Board had invested in a data manager to produce reports for the TB service and the Health Board, informing and raising the profile of tuberculosis management.
Recommendations: Data Management
16. National Services Scotland should consider the development of other data platforms, which would be helpful for in patient management and communication and would feed into national surveillance databases.
N
17. Inputting data into such databases is an essential part of surveillance work and this needs to be appropriately resourced with administrative staff, of which there seems to be few in Scotland, as noted in the workforce section 7. This needs to be reviewed and properly resourced for each service, noting the benefits of accurate data entry and freeing up clinical staff.
P, HB
18. Development of TB Service Analysis Reports, which can influence and support TB development at Health Board level, should be considered and developed by all TB services for Health Board level use.
P, HB
9.2 Diagnostics
For diagnostics, few services had immediate availability of smears but all had access within a timely period and this was not seen as a specific issue. Following developments a few years ago, all but one service had access to GeneXpert Mycobacterium tuberculosis complex (MTBC) and resistance to rifampin (RIF) (MTB/RIF) tests, with one third having the service at regional or Health Board level and 40% by the acute provider, with most services stating there was little issue in accessing the test.
Most, but not all services had access to negative pressure rooms if needed for induced sputum, although we noted from the deep dive meetings this procedure is infrequently used (Figure 13). Most services use fibre optic bronchoscopy for obtaining samples for suspected pulmonary disease, with most having a protocol or SOP (Standard Operating Procedure) for the procedure. An example is included in good practice via the link in Annex E.
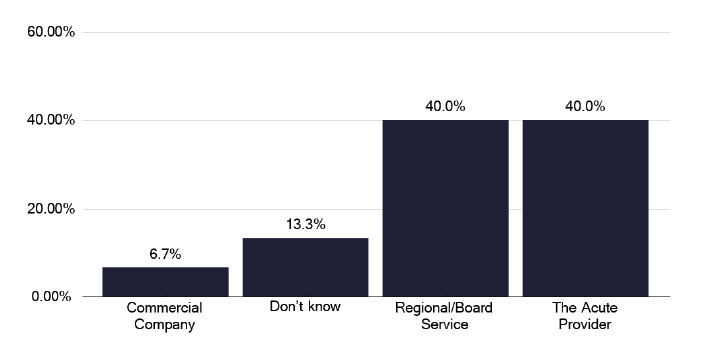
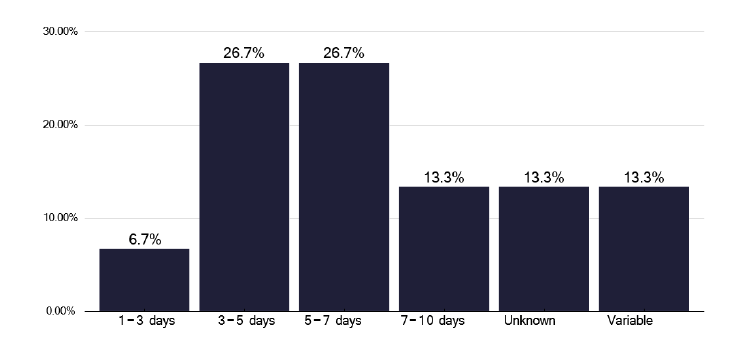
IGRA testing (Interferon-gamma release assay) is key in determining the need for preventative therapy and we noted this was undertaken by the acute provider in 40% of services and by regional services in a further 40%, with one being provided by a commercial company and a further two were uncertain, as shown in Figure 14. There was an adequate turnaround time of the tests of a maximum of 7 days for most services as shown in Figure 15.
We heard from some pathology representatives during the deep dives about who pays for the IGRA testing, as this is an increasing cost burden for pathology. A discussion should take place between departments and services who are requesting IGRAs as a precursor to initiating biological therapies, and with pathology departments about appropriate recognition and cross charging for these tests.
We heard excellent examples where the pathology department sent the results of all, or just the positive IGRA results, to TB clinical leads to ensure that no positive IGRA results were missed. This is excellent practice and should be considered as the norm, as documented in good practice via the link in Annex E.
One feature that became apparent is that not all TB services were aware of the number of IGRAs being performed for different groups of patients as shown in Figure 16 recognising that IGRA is the preferred test prior to preventative action. We heard of wide variation in the reasons for testing, which in part may reflect the size of the service, the nature of any specialised services within that provider and knowledge by the service of the number of tests done. The outlier with 5200 tests, most for biologics, reflects the knowledge of that service of IGRA activity but highlights the significant cost pressure on departments performing IGRA testing.
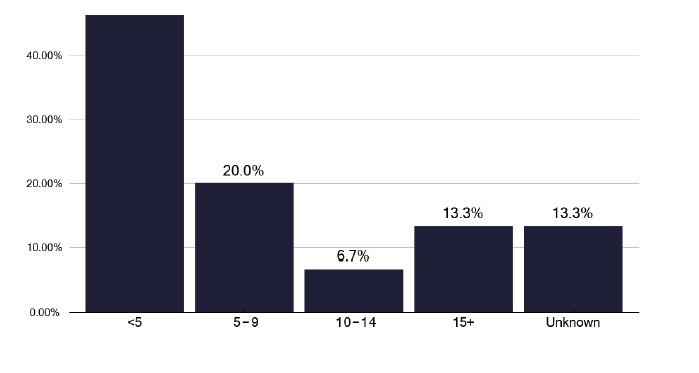
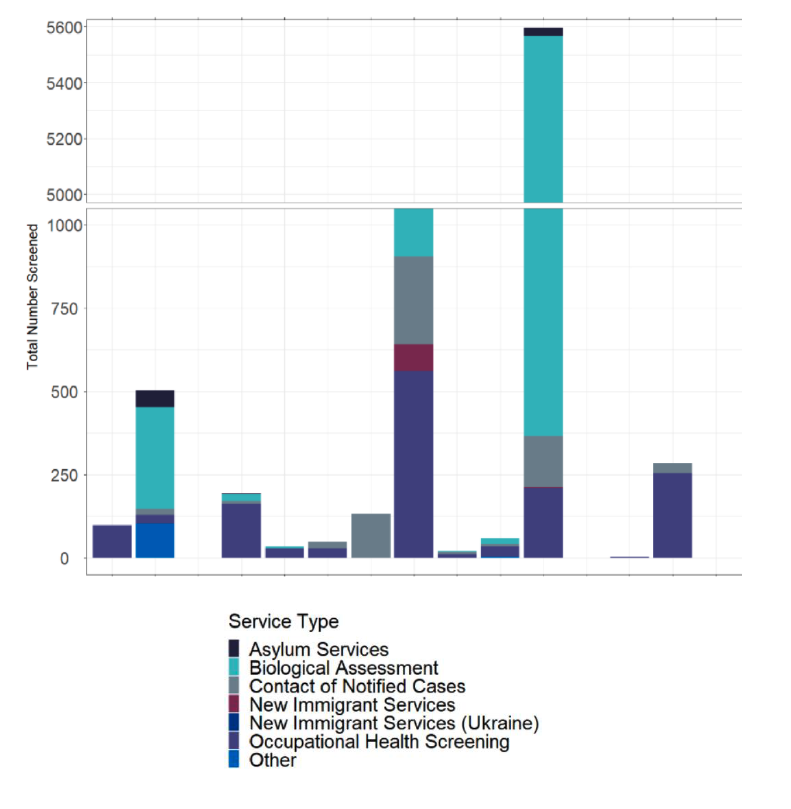
Comparison of the number of LTBI patients screened across services (from survey submissions)
Recommendations: Diagnostics
19. Services should have both protocols and the option for negative pressure facilities if they feel induced sputum is a technique they wish to use for obtaining respiratory samples.
P, HB
20. All IGRA results (or just positives, depending on the service) should be notified to the TB lead, with the opportunity to ensure no positives are missed. This also gives an opportunity, working with pathology, to ensure there are not excessive and inappropriate requests from some speciality services.
P, HB
21. Discussion between speciality services requesting IGRA and pathology departments should take place to ensure appropriate funding.
P, HB
Contact
Email: healthprotection@gov.scot