Offshore wind energy - draft sectoral marine plan: habitat regulations appraisal
The habitats regulations appraisal is completed in accordance with the Habitats Regulations that implement the EC Habitats and Birds Directives in UK waters and has been completed for the sectoral marine plan for offshore wind.
Appendix I: Fish Literature Review
I.1 Migratory Fish Baseline Characteristics
I.1.1 Scope of Review
This appendix presents a review of the ecology of migratory fish populations found in Scottish waters and is designed to provide a context for the HRA of the DPOs in Scottish waters. The review focuses specifically on the migratory behaviour and distribution at sea of the following species as qualifying interest species identified during the HRA for the Sectoral Wind Plan:
- Atlantic salmon Salmo salar;
- Sea lamprey Petromyzon marinus L;
- River lamprey Lampetra fluviatilis;
- Allis shad Alosa alosa; and
- Twaite shad Alosa fallax.
This is a broad-scale characterisation only which seeks to draw upon available information to understand the value of the area in general terms. It does not specifically focus on the interest features of the European/Ramsar sites but provides a context for that assessment in recognition of the importance of migratory fish in this area.
I.1.2 Major Information Sources
This appendix presents a review of the ecology of migratory fish populations found in Scottish waters and is designed to provide a context for the HRA of the DPOs in Scottish waters. The review focuses specifically on the migratory behaviour and distribution at sea of the following species as qualifying interest species identified during the HRA for the Sectoral Wind Plan:
- Atlantic salmon Salmo salar;
- Sea lamprey Petromyzon marinus L;
- River lamprey Lampetra fluviatilis;
- Allis shad Alosa alosa; and
- Twaite shad Alosa fallax.
This is a broad-scale characterisation only which seeks to draw upon available information to understand the value of the area in general terms. It does not specifically focus on the interest features of the European/Ramsar sites but provides a context for that assessment in recognition of the importance of migratory fish in this area.
I.2 Atlantic Salmon Salmo salar
I.2.1 Ecology
The Atlantic salmon is anadromous, its life cycle involving both a freshwater and seawater phase. Salmon utilize rivers for reproductive and nursery phases and the marine environment for adult development and rapid growth (Hendry and Cragg-Hine, 2003). In late autumn and early winter adult salmon spawn in clean gravel in the river or stream of their origin. Eggs are deposited in spawning reeds, depressions in the gravel bed excavated by the female salmon (Birkeland, 2003).
Juvenile salmon usually remain in fresh water for between two and four years, before migrating to sea. Migration takes place between April and June, when the parr begin to turn silver and become smolts. During the smoltification process salmon undergo a number of behavioural, morphological and physiological changes, which prepare them for a life in the sea. Upon reaching the sea, salmon grow rapidly, feeding on a variety of small fish including capelin, herring, sand eels and sprats. After one winter at sea a salmon is called a grilse or 1 sea-winter (1SW), and fish that have spent more than one winter at sea are called a multi-sea-winter (MSW) salmon. The length of time salmon spend in the sea before returning to their river of origin to spawn varies from one winter to five winters (Birkeland, 2003; Hendry & Cragg-Hine, 2003; Malcom et al. 2010).
The UK Atlantic salmon population comprises a significant proportion of the total European stock. Scottish rivers are a European stronghold for the species (JNCC, 2007).
I.2.2 Distribution and Migration Routes at Sea
Adult and sub-adult salmon have been observed using the marine environment at locations extending from Labrador in the west to Faroe and the Norwegian Sea in the east. No tagged Scottish Atlantic salmon have been observed on the Norwegian coast. Holm et al. (2000) investigated the spatial and temporal distribution of post-smolts of Atlantic salmon in the Norwegian Sea and adjacent areas. The highest incidences of post-smolts were in the slope current west of the British Isles, where the highest catch per unit of effort (cpue) value was recorded between northern Scotland and the Faroes in June 1997 (Image I1). The post-smolts appear to follow the main surface currents northwards into the Norwegian Sea where they spread in a fan-like distribution over an area covering most of the international waters between the exclusive economic zone (EEZ) of Norway, the Faroes and Iceland up to about latitude 73-75 N. Hansen and Jacobsen (2003) investigated the origin and migration of wild and escaped farmed Atlantic salmon in oceanic areas north of the Faroe Islands. The tagging experiment found relatively large numbers of returns were observed in Scotland.
Image I1: Dominant Directions of Travel for Atlantic Salmon (1SW and MSW) in Scottish Coastal Waters Based on Tagging Studies
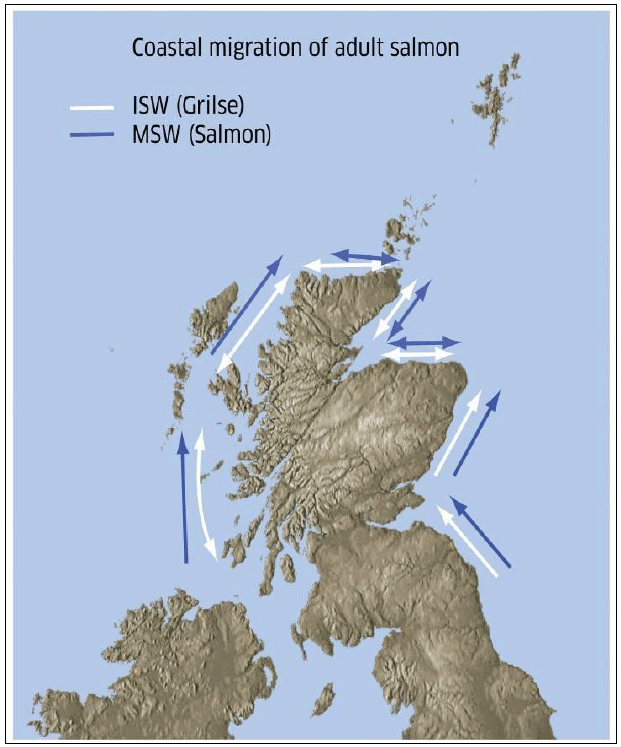
(Source: Malcom et al. 2010)
Greenland and Faroe seas are considered to be particularly important as a feeding area for Scottish salmon based on data from the fisheries in these areas. However, the exact spatial distribution is currently unknown as most data has only come from Greenland and Faroese fisheries, representing only two sampling points for the much larger area of the ocean in which salmon are potentially represented. The concept that salmon tend towards broader, less clearly defined marine habitat use making use of oceans currents has been suggested by some research but data is currently not considered sufficiently detailed to infer specific migration corridors (Malcolm et al. 2010).
Patterns of migration indicated by tagging studies around the Scottish coast suggest that Atlantic salmon make landfalls at many different parts of the coast and then redistribute themselves (Dunkley, 1985). Malcom et al. (2010) concluded that 1SW and MSW sea-age classes may return towards the Scottish coasts across a broad front with the marine origins of the fish are likely to be highly biased towards a range of locations to the north and west of the British Isles. Malcom et al. (2010) also suggested a conceptual model on how Scottish salmon reaching the coast subsequently migrate towards their natal river based on a review of available evidence. The model proposed that salmon and grilse return both to the north and west coasts of Scotland, and may even reach the north east coast directly having passed Orkney and Shetland. In general, this wide geographic distribution of potential arrival locations gives variable and apparent random directions of migration. However, several broad trends in migration patterns are evident:
- Given that MSW salmon rivers dominate the north and east coasts, the dominant direction of movement for MSW fish caught on the west will be north and east. However, for grilse, the pattern of movement would depend on where they reach the shoreline and where their native river was located.
- For east coast rivers south of Aberdeenshire fish generally moving in a northward direction from the Northumberland coast
I.3 Sea Lamprey Petromyzon marinus
I.3.1 Ecology
The sea lamprey is the largest of the British lampreys. This anadromous species occurs over much of the Atlantic coastal area of western and northern Europe, from northern Norway to the western Mediterranean, and eastern North America (Maitland, 2003). Metamorphosis to the adult form takes place between July and September and the process usually takes a few weeks. The time of the main migration downstream seems to vary from river to river and relatively little is known about them after they reach the sea, where they have been found in both shallow coastal and deep offshore waters. The spawning migration in Europe usually takes place in April and May when the adults start to migrate back into fresh water (Maitland, 2003; Harvey & Cox, 2003).
I.3.2 Distribution and Migratory Routes at Sea
Sea lampreys adult (growth) phase is short and lasts around 2 years. In this time, the species is parasitic feeding on a variety of marine and anadromous fishes, including shad, herring, pollack, salmon, mullets, cod, haddock, greenland sharks and basking sharks (OSPAR, 2008; Gallant et al. 2006).
The rarity of capture in coastal and estuarine waters suggests that marine lampreys are solitary hunters and widely dispersed at sea. Sea lamprey has been caught at considerable depth (up to 4099m) suggesting that they can feed in deeper offshore waters (Haedrich, 1977). However, owing to the parasitic nature of the adult phase their distribution is largely dictated by their host. Homing behaviour is not apparent in this species. However, they are selective in their choice of spawning streams and are thought to favour sites where ammocoete larvae are present due to olfactory cues (OSPAR, 2008).
I.4 River Lamprey Lampetra fluviatilis
I.4.1 Ecology
The river lamprey (Lampetra fluviatilis) is intermediate in size between the large sea lamprey and the small brook lamprey. The average adult length is around 30cm with a corresponding weight of about 60 g, but specimens over 40cm can be found, and the unusual race in Loch Lomond is often less than 20 cm. River Lamprey are anadromous migratory species, which grows to maturity in estuaries around Britain and then moves into fresh water to spawn in clean rivers and streams (Maitland, 2003).
I.4.2 Distribution and Migratory Routes at Sea
Unlike sea lamprey the growth phase of river lamprey is primarily restricted to estuaries. In the estuary the river lamprey feeds on a variety of fish, but particularly herring Clupea harengus, sprat Sprattus sprattus and flounder Platichthys flesus. After one to two years in estuaries, river lamprey stop feeding in the autumn and move upstream into medium to large rivers, usually migrating into fresh water from October to December (Maitland, 2003).
I.5 Shad Species
I.5.1 Ecology
The allis and twaite shad Alosa alosa and Alosa fallax respectively are both members of the herring family Clupeidae. The species are anadromous, and have a pelagic-sea life mainly inshore along the coast migrating to the higher, middle watercourse of rivers to spawn (Maitland and Hatton-Ellis, 2003). Mature fish that have spent most of their lives in the sea stop feeding and move into the estuaries of large rivers, migrating into fresh water during late spring (April to June). Males migrate upstream first, followed by females one or two weeks later. At maturity both species stop feeding and gather in the estuaries of suitable rivers in early summer (April and May), moving upstream to spawn from mid-May to mid-July. Allis shad are almost exclusively planktivorous, whereas twaite shad also feed on small fish such as sprats, and this is likely to be reflected in their habitat selection.
No subspecies of allis shad are recognised, but there is some evidence that they return to the natal river, suggesting that there may be some genetic integrity within populations. Several subspecies of twaite shad have been recognised by various authors but only one of these, Alosa fallax fallax, occurs in Britain (Maitland and Hatton-Ellis, 2003).
I.5.2 Distribution and Migratory Routes at Sea
The requirements of shads at sea are very poorly understood, but they appear to be mainly coastal and pelagic in habit, but have both been occasionally recorded over 100 km offshore (Image I2). Allis shad have been reported from depths of 10-150 m, and twaite shad from depths of 10-110 m, with a preference for water 10-20 m deep. A suitable estuarine habitat is likely to be very important for shad, both for passage of adults and as a nursery ground for juveniles (Maitland and Hatton-Ellis, 2003; OSPAR, 2009).
Image I2: Presence/Absence Data for Allis and Twaite Shad
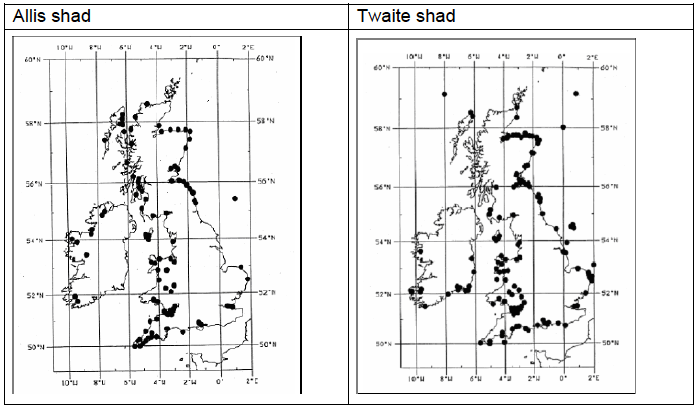
Source: JNCC, 2007)
I.6 References
Birkeland, K. 2003. The River Borgie cSAC Conservation Strategy. Conserving Natura 2000 Rivers. English Nature, Peterborough.
Gallant J., Harvey-Clark C., Myers R.A., Stokesbury M.J.W. 2006. Sea lamprey Petromyzon marinus attached to a Greenland shark Somniosus microcephalus in the St. Lawrence Estuary, Canada, Northeastern Naturalist, 13, 35-38.
Hansen, L. P., and Jacobsen, J. A. 2003. Origin and migration of wild and escaped farmed Atlantic salmon, Salmo salar L., in oceanic areas north of the Faroe Islands. ICES Journal of Marine Science, 60: 110-119.
Haedrich R.C. 1977. A sea lamprey from the deep. Copeia 4: 7678.
Harvey, J and Cox, I .2003. Monitoring the River, Brook and Sea Lamprey, Lampetra fluviatilis, L. planeri and Petromyzon marinus. Conserving Natura 2000 Rivers Monitoring Series No. 5, English Nature, Peterborough.
Hendry, K anf Cragg-Hine D. 2003. Ecology of the Atlantic Salmon. Conserving Natura 2000 Rivers Ecology Series No. 7. English Nature, Peterborough.
Holm, M., Holst, J. Chr., and Hansen, L. P. 2000. Spatial and temporal distribution of post-smolts of Atlantic salmon Salmo salar L. in the Norwegian Sea and adjacent areas. ICES Journal of Marine Science, 57: 955-964.
JNCC, 2007. Second Report by the UK under Article 17 on the implementation of the Habitats
Directive from January 2001 to December 2006. Peterborough: JNCC.
Malcolm, I. A., Godfrey, J and Youngson, A.F. 2010. Review of migratory routes and behaviour of Atlantic Salmon, sea trout and European eel in Scotlands coastal environment: implications for the development of marine renewables. Scottish Marine and Freshwater Science Volume 1 No 14.
Maitland. P.S. 2003. Ecology of the River, Brook and Sea Lamprey. Conserving Natura 2000 Rivers Ecology Series No. 5. English Nature, Peterborough.
Maitland. P.S and Hatton-Ellis, T.W. 2003. Ecology of the Allis and Twaite Shad. Conserving Natura 2000 Rivers Ecology Series No. 3. English Nature, Peterborough.
OSPAR. 2008. Case reports for the OSPAR list of threatened and/or declining species and habitats.
OSPAR. 2009. Background Document for Allis shad Alosa alosa
Contact
Email: drew.milne@gov.scot