Offshore wind energy - draft sectoral marine plan: habitat regulations appraisal
The habitats regulations appraisal is completed in accordance with the Habitats Regulations that implement the EC Habitats and Birds Directives in UK waters and has been completed for the sectoral marine plan for offshore wind.
Appendix G: Seabirds Literature Review
G.1 Seabird Foraging Baseline Review for the Marine Sectoral Plan HRAs
This appendix reviews what is currently known about the foraging behaviour, feeding locations and distribution for coastal and offshore bird colonies (mainly breeding populations). It has been prepared to inform the HRA for the Sectoral Wind Plans for Offshore Renewable Energy (ORE) within Scottish waters. For the Sectoral WindPlan HRA, it is important to understand what current data exists to describe seabird distributions to inform the review of baseline conditions within Scottish waters and then evaluate the Plan’s impacts. In particular, it is recognised, that a lot of work has been done in recent years to describe the distribution and behaviours of seabirds offshore in order to inform assessments of the impacts arising from coastal and offshore developments as well as to underpin marine spatial planning initiatives. The review presented in this appendix identifies this recent work to produce a stand-alone statement about the current ‘state of knowledge’ on sea bird distributions.
The focus of this review has been on birds foraging behaviour and ‘at sea’ distributions because it is during foraging excursions that birds are at risk of impacts from proposed marine renewables energy development (typically wave devices pose less of a threat to seabirds than tidal devices, and both present less of a hazard than offshore wind farms). This review has also considered seasonal migratory flyways and species-specific vulnerability to wind energy generation.
G.2 Key Data Sources
The recent work reviewed in this appendix includes the Future of the Atlantic Marine Environment (FAME) project which is a large-scale GPS bird-tagging exercise that is being lead by the RSPB. This project considers the foraging behaviour, direction and distances of seabirds from breeding colonies. FAME is designed to inform the designation of Marine Protected Areas (MPAs) and develop best practice management recommendations for these areas. This is a pan-European Union (EU) partner scheme involving the UK (as lead), Ireland, France, Spain and Portugal. In addition, STAR (Seabird and Tracking Research) which is a twin project to FAME, has also collected further tracking data from sites in the UK and Ireland. Data from these projects have been used to develop predictive species distribution models and high-density usage maps for a variety of species (Wakefield et al., 2017; Cleasby et al., 2018).
In addition to the FAME work, a review of the foraging ranges of 25 species of UK seabirds has recently been prepared jointly by BTO, RSPB and Birdlife International (Thaxter, 2012) with the aim of being used for scoping candidate MPAs has been included in this report. This recent paper provides a robust and most up to date assessment of data which is useful to this HRA and helps towards a comprehensive view of seabird populations and behaviour in the area.
BirdLife International also created a online seabird foraging range database (http://seabird.wikispaces.com) which has also been reviewed in this appendix. This database was compiled by BirdLife and included seabird ecology and foraging ranges to be used to help identify important marine bird areas to help inform MPA designations and marine spatial planning.
In addition, a survey of the Pentland Firth and Orkney Waters area (PFOW) has been completed by APEM (2012) for Marine Scotland and The Crown Estate (TCE) and the report of this work has been obtained and reviewed. This report aims to gain a better understanding of the distribution and utilisation of sea space by birds in the Pentland Firth and Orkney Waters (PFOW) area. APEM (2012) undertook seven aerial surveys of the area between November 2010 and August 2011 . It has been included here because the PFOW area is an important location for marine energy resources as well as being a known important area for seabirds throughout the year. Also as part of the PFSA HRA (ABPmer, 2010) the BTO reviewed the degree to which seabird species are potentially exposed to effects within the PFOW area and identified which European/Ramsar sites outside the area may be affected. We have carried some of this information through into this HRA.
In addition to reviewing these data sources, throughout this appendix we have also drawn upon a Joint Nature Conservation Committee (JNCC) report looking at the distribution of seabirds in the study area (Pollock et al, 2000). This report considers data from surveys conducted in waters north and west of Scotland from 1979 to 1999. The results of this report give us a clearer understanding of where individual sea bird species are during different times of the year and provides good visual interpretations of the distribution of these species.
In addition to the above, European Seabirds at Sea (ESAS) information has been included (www.seamap.env.duke.edu last accessed 13/02/12). This is a collaborative dataset with inputs from the JNCC, and other north western European organisations. The dataset was established in 1991 with the aim of collecting data on the distribution of seabirds in north-west European offshore areas.
These data sources provide information at a wide range of spatial scales from a national scale (e.g. the OBIS Seamap) to a more regional scale (e.g. the recent APEM surveys of PFOW) and then to a local level (e.g. the individual bird tagging routes provided by the FAME project). Each also employs different methods and analysis and they cover different areas. This needs to be borne in mind when making any between-survey comparisons. However, collectively these data give a very comprehensive description of the broad distributions and behaviours of the key bird species.
Information on the ecology of seabirds based on recent tagging studies for a range of species including Gannets (Langston et al., 3013; Soanes et al., 2013; Wakefield et al., 2013), Great Skua (Wade et al., 2014), Kittiwakes (Trevail et al., 2019) and Fulmars (Quinn, 2014) were also reviewed. In addition, research on the distribution and ecology of species during the non-breeding season has also be presented (such as Bogdanova et al., (2017); Glew et al., (2018) and Furness (2015)).
G.3 Seabird Foraging Behaviour
The distances over which waterbirds forage offshore has been reviewed to inform particularly the screening review for the Sectoral WindPlan HRA. For this review, information was drawn upon from a range of studies including those listed above and the other studies of seabird foraging distances that were undertaken by BTO (Roos et al 2009) as part of the PFSA HRA work (ABPmer, 2010), and reports reviewed therein including; Furness and Tasker (2000), Hamer et al., (2000, 2001, 2007), Suryan et al. (2000), Thaxter et al. (2010) and Wanless et al. (1990, 1991, 1998). The online BirdLife International (2018) seabird foraging database was also reviewed along with a number of other papers including; BirdLife International (2000), Fenny & Walls (2009), Ratcliffe et al. (2000) and Thaxter, 2012 and the maximum foraging ranges for each species were identified. Thaxter et al. (2012) was also reviewed to identify the mean maximum foraging ranges for each species. The reference section in each of the retrieved papers was also scanned to make sure that no relevant studies were missed. Where possible the methodology used in each study was identified, as different approaches may have intrinsic problems. The results are presented as a summary table showing mean maximum foraging ranges for each species along with text summaries for each of the species that forage over the greatest distances offshore (Table G1).
This summary review is designed to provide guidance of the sites and species that need to be considered during the screening and assessment work for this Plan-level HRA as well as for future project-level HRAs. Focus is on seabirds foraging offshore as opposed to species (especially waders) that forage in intertidal areas. In summary, the factors that might affect offshore seabird foraging range distances include:
- Spatial variation in food abundance (including anthropogenic factors such as fishing vessels);
- The risk of predation/kleptoparasitism by other bird species;
- The importance of nest attending to incubate eggs and protect nest from predators; and
- Weather and climatological factors (British Trust for Ornithology (Roos et al., 2009).
The distances that are typically covered by seabirds foraging offshore vary greatly between species and research and understanding of their foraging behaviour has increased in recent years. As part of this research, increasingly novel techniques and technologies are being developed to track birds and to investigate foraging behaviour. Over relatively short distances and timescales transects, ship-based surveys and radio-telemetry have been used (Pollock et al, 2000; Perrow et al., 2006; Wilson et al., 2009). To track birds over considerably greater distances and over larger time scales GPS data loggers are deployed, however they necessitate recovery of the data logger to extract information, or close approach for remote data download (Guilford et al., 2008; and the FAME work). The greatest opportunity to follow birds over potentially large distances and over extended time periods comes from satellite tracking, but at present this is only useful for birds of large body size such as northern gannet (Hamer et al., 2000; 2001; 2007). Radar is one of the more powerful tools available to describe the movement of birds in three-dimensional space and its use has been long-established and well described (Desholm et al., 2004). Relationships between foraging trip duration and, respectively, maximum distance from the colony and total trip distance have also been estimated for many bird species to determine foraging behaviour (Hamer et al., 2007). From the available range of literature sources and different methods, Table G1 presents the maximum and mean maximum (i.e. the maximum range reported from the studies reviewed in Thaxter et al., 2012 averaged across studies), foraging distances of seabirds (from breeding colonies).
A review as part of previous HRA screening work (ABPmer, 2010) highlighted seabirds that typically forage within 100km of breeding site and those which travel beyond 100km. Thaxter et al (2012) and the FAME project (Table G1) have been used to update foraging distance information taking account of advice from SNH and the PSG. Taking this updated information and following advice from SNH, an overall list of species with mean maximum foraging distances greater than 100km has been produced for screening the Sectorla Wind Plan HRA . These species are as follows (mean maximum foraging distances are highlighted in brackets):
- Atlantic Puffin (105 km);
- Manx Shearwater (330 km);
- Northern Gannet (229 km);
- Northern Fulmar (400 km); and
- Lesser Black-backed gull (141 km)
Although foraging distances are relatively well understood, less information is available to indicate foraging directions. However, the recent FAME studies are providing a lot of new and useful data on this issue. The results from this and other previous analyses are reviewed below.
Table G1: Maximum and mean maximum foraging distances of seabirds (from breeding colonies) from a number of literature sources
| Species Common Name | Maximum Foraging Range (km) | Mean Maximum Foraging Range (km) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| BirdLife International (2010) | BLI Mean Max |
BTO Review (Roos et al., 2009 in ABPmer 2010) |
Furness & Tasker (2000) | Ratcliffe et al. (2000) |
Fenny & Walls (2009) | FAME/STAR | Thaxter et al (BTO/BLI/RSPB) (2012) | Thaxter et al (2012) | |
| Arctic Skua | 100 | 40 | Unknown | <10 | ≤5 | 75 | 63 | ||
| Arctic Tern | 20.6 | 12 | 25 | <5 | ≤15 | 20-30 | 30 | 24 | |
| Atlantic Puffin | 200 | 62 | 137 | <50 | ≤40 | 40 | 200 | 105 | |
| Black Guillemot | 55 | 12 | ≤5 | ||||||
| Black-headed Gull | ≤15 | 40 | 26 | ||||||
| Black-legged Kittiwake | 200 | 66 | 83 | <50 | ≤40 | 40 | 300 | 120 | 60 |
| Common Eider | 100 | 38 | 80 | 80 | |||||
| Common Guillemot | 200 | 61 | 123 | <50 | ≤40 | 40 | 340 | 135 | 84 |
| Common Gull | ≤15 | 50 | 50 | ||||||
| Common Scoter | 2 | 8 | Unknown | - | |||||
| Common Tern | 37 | 34 | ≤15 | 20-30 | 30 | 24 | |||
| European Shag | 20 | 16 | 17 | <10 | ≤15 | 15 | 17 | 15 | |
| European Storm Petrel | Unknown | <50 | >100 | >65 | |||||
| Gt. Black-backed Gull | Unknown | <10 | ≤40 | 40 | |||||
| Great Cormorant | 50 | 32 | 35 | <5 | ≤15 | 15 | 35 | 25 | |
| Great Skua | 100 | 42 | 31 (est.) | <10 | ≤40 | 219 | 86 | ||
| Herring Gull | 54 | <10 | ≤40 | 40 | 92 | 61 | |||
| Leach’s Storm Petrel | Unknown | <50 | >100 | <120 | 92 | ||||
| Lesser Black-backed Gull | ≤40 | 40 | 181 | 141 | |||||
| Little Gull | 50 | ||||||||
| Little Tern | 11 | 7 | ≤5 | 20-30 | 11 | 6 | |||
| Manx Shearwater | 400 | 196 | 330 | >50 | >100 | >330 | 330 | ||
| Northern Fulmar | 664 | 311 | 245 | >50 | >100 | >100 | 580 | 400 | |
| Northern Gannet | 640 | 308 | 540 | >50 | >100 | >100 | 590 | 229 | |
| Razorbill | 51 | 31 | 150 | <20 | ≤40 | 40 | 312 | 95 | 49 |
| Red-throated diver | 50 | 12 | 13 | <5 | 9 | 9 | |||
| Roseate Tern | 30 | 18 | ≤15 | 20-30 | 30 | 17 | |||
| Sandwich tern | 70 | 42 | ≤15 | 20-30 | 54 | 49 | |||
| Velvet Scoter | 20 | ||||||||
| Highlighted species are those with mean maximum foraging ranges of ≥ 100km which meant that those European/Ramsar sites which support these species but lie outside the 100km pre-screening buffer zone needed to be considered at the screening stage. | |||||||||
G.3.1 Species review
Those species listed-above which forage over long distances have been included in more detail in the following review. These species are reviewed particularly because they could forage within the short and long term option areas even though the European/Ramsar sites for which they are a qualifying interest features lie at some distance away. In particular, because they are likely to forage over distances of greater than 100km, the sites for which they are designated features could lie outside the 100km boundary area that was used to inform the screening of the AA. In addition, the Leach’s storm petrel, great skua, black-legged kittiwake, razorbill, European storm petrel and common guillemot have maximum foraging distances >100km and these have also been reviewed. These reviews of foraging distances for individual species draw predominantly upon the information contained within the online BirdLife International (2018) seabird foraging database.
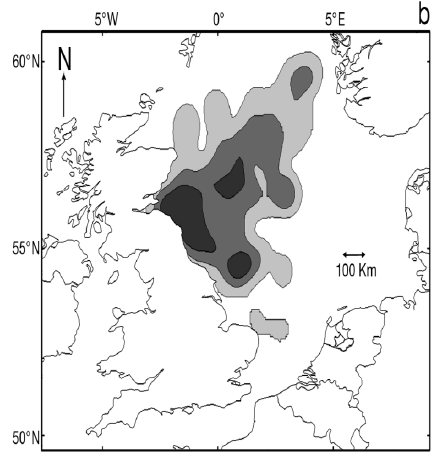
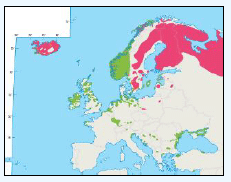
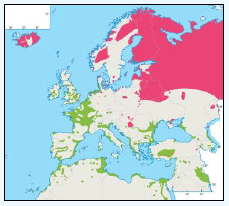
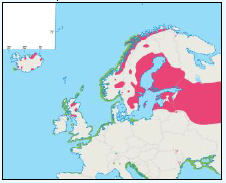
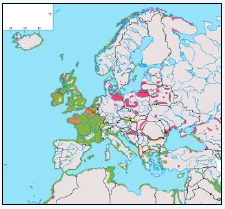
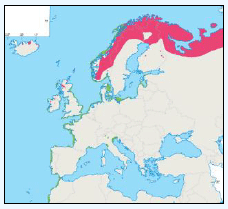
As a first stage analysis, for each species the relevant seabird distribution from OBIS Seamap is presented in Image G1 which presents data from the OBIS Seamap website. The data is presented in terms of density (birds/km2). This is achieved by summing the number of birds recorded in the transect, both on the water and flying, in each of the 1/36 ICES rectangles. This value is then divided by the area surveyed within that rectangle. In order to account for the variations in detection of birds on the water at different distances from the ship, the numbers of birds (excluding those flying) were multiplied by a factor according to species and width of the strip transect. The data in Image G1 represents the density for each species from 1978 to 2002. The information here demonstrates the value of Scottish waters and the SREZ as a whole to seabirds. This information and other data are reviewed for each key species below.
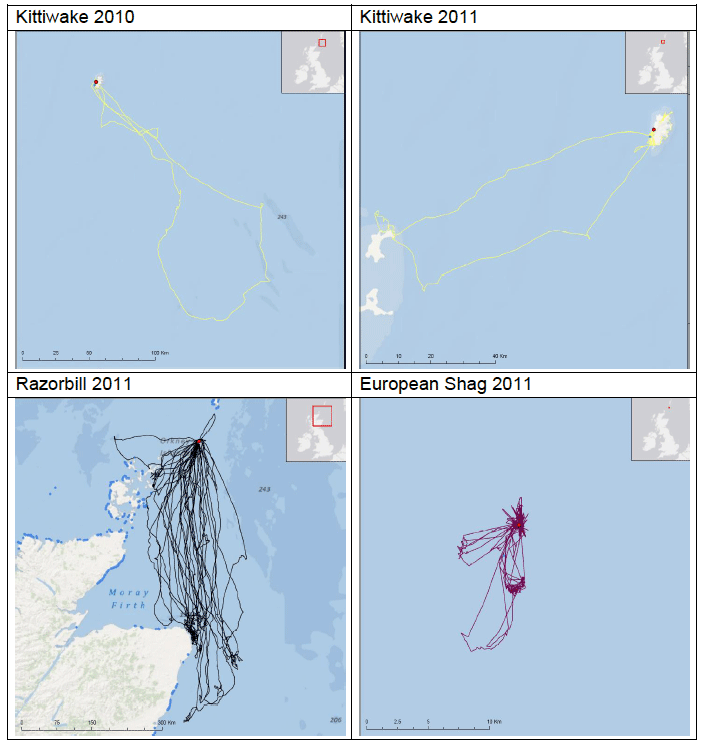
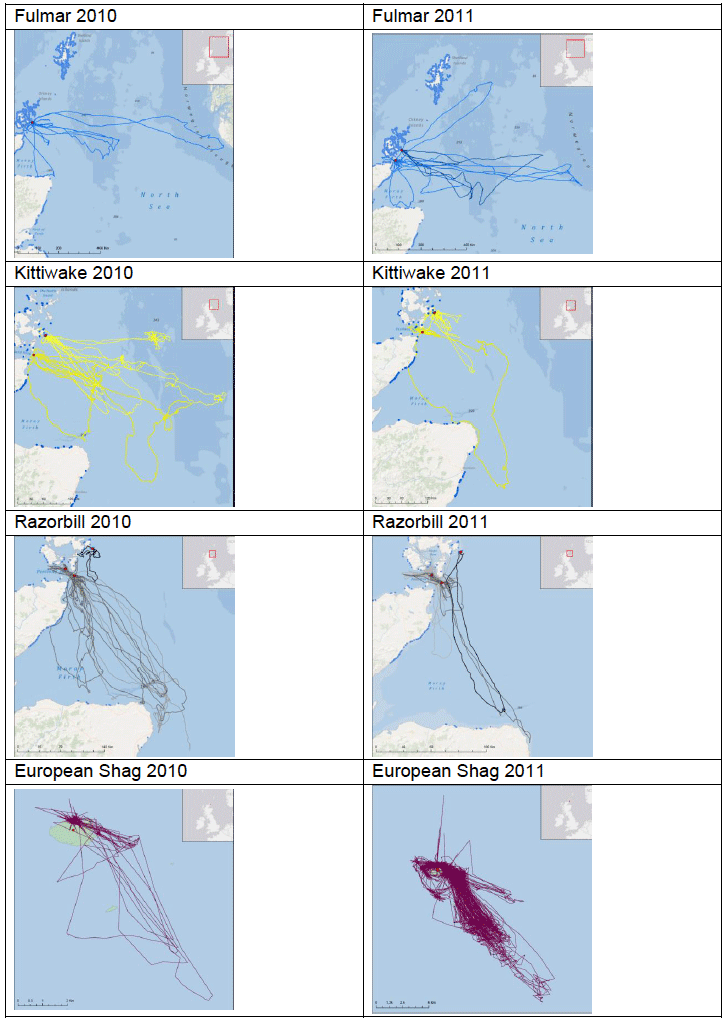
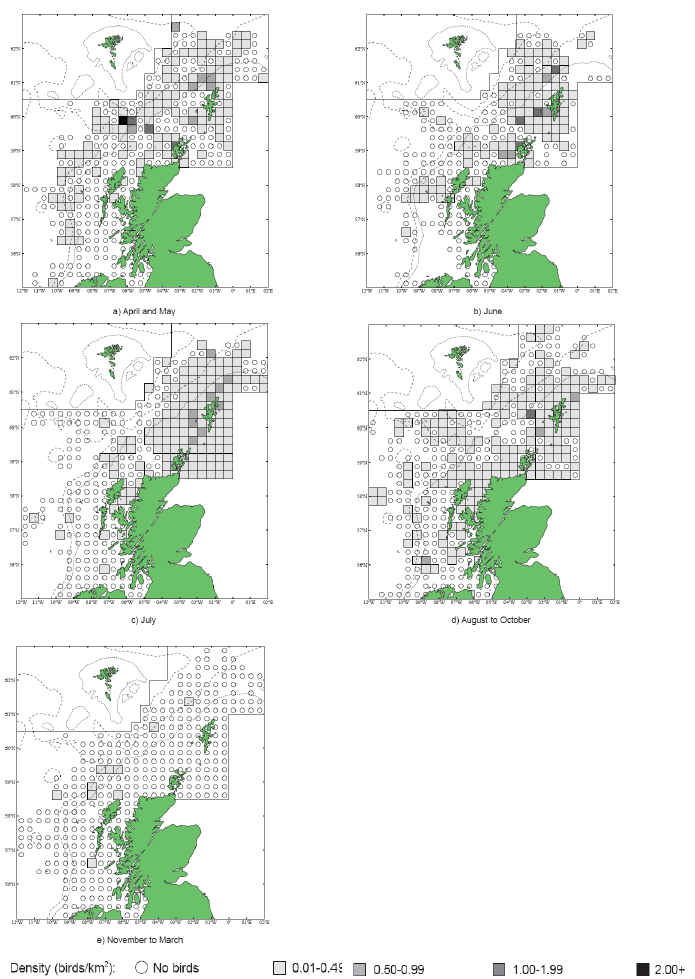
Some of the species listed above were tagged as part of the FAME project. Tagging work was carried out between April and August both in 2010 and in 2011 at various locations around the SREZ. GPS loggers (I-gotU GT-120, Mobile Action Technology) were attached to breeding birds using a temporary attachment method that would allow the tag to fall off the birds in the event that they were not re-caught. The tags recorded the bird’s location once every 100 seconds to an accuracy of approximately 15 m which provided high resolution location data. Data collated from the tags were only accessible by re-catching the bird to remove the tag between one and five days after attachment. Images G2-G4 show foraging patterns over a two-year period of Common Guillemot, Kittiwake, Razorbill, European Shag and Fulmar at various locations around the SREZ. It is clear from the images that at some locations, for example Colonsay there are foraging hotspots for the majority of the species tagged. Further research into foraging activity of these birds in relation to physical processes (e.g. tidal flows) and other biological surveys (e.g. seabird prey location) is being undertaken by RSPB (Owen, 2012) but there are no publicly available results to date.
Image G1: Data From OBIS-Seamap (Seabird Distribution and Abundance Data for Each Month From 1978 to 2002)
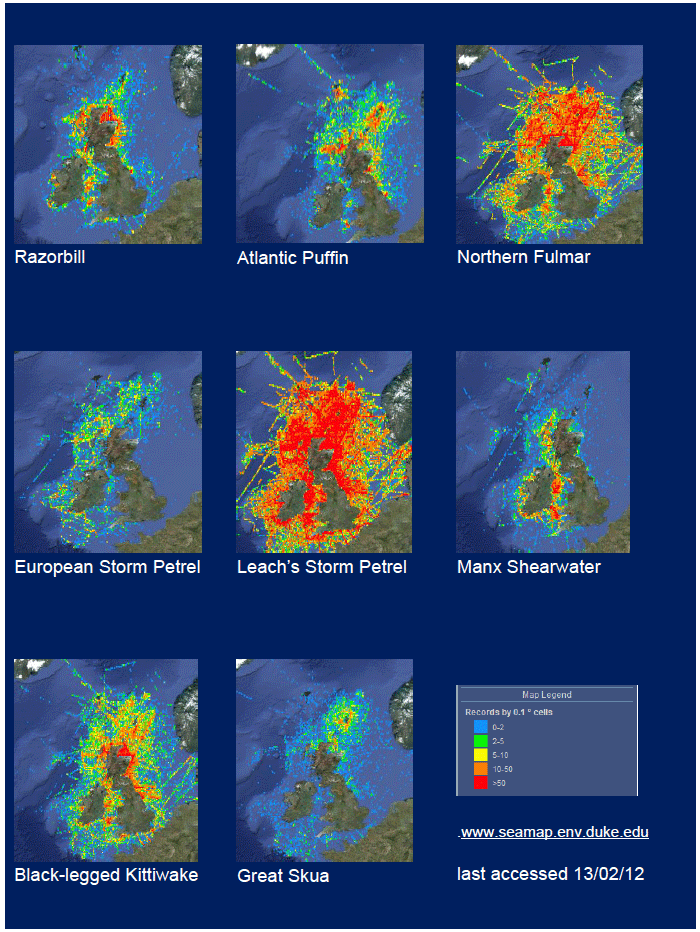
(Source: RSPB/FAME project)
Image G2: Foraging tracks of seabirds from Colonsay
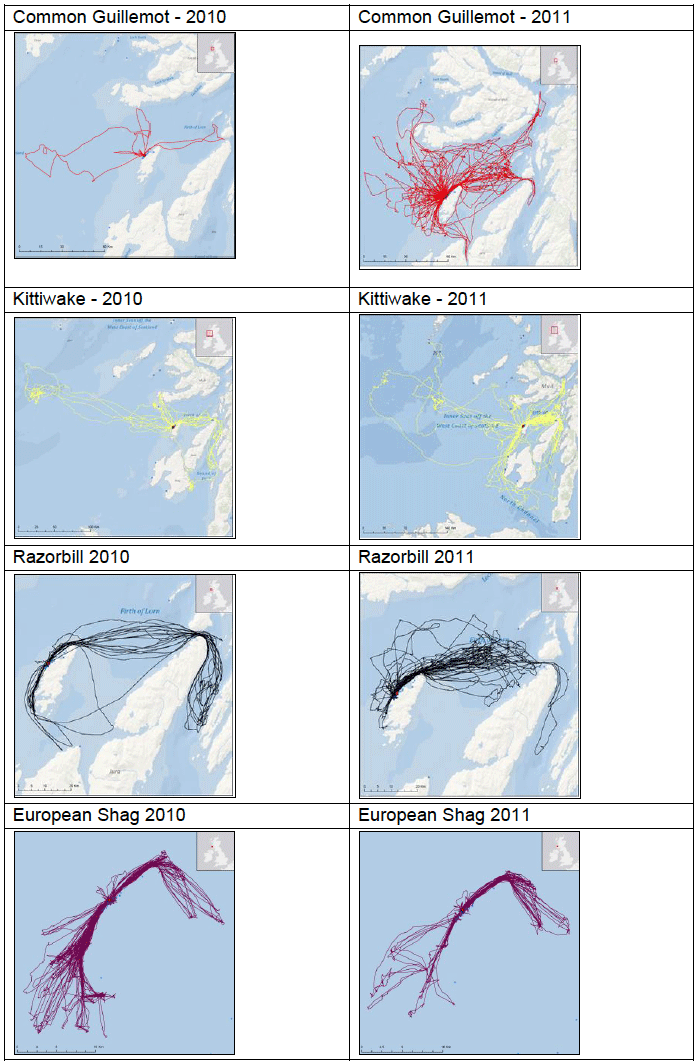
(Source: RSPB/FAME project)
Image G3: Foraging tracks of seabirds from Fair Isle
(Source: RSPB/FAME project)
Image G4: Foraging tracks of seabirds from Orkney
G.3.2 Atlantic Puffin Fratercula arctica
Atlantic Puffins (hereafter Puffin) from north west Britain disperse widely and have been observed in Newfoundland, the Canary Islands and the Mediterranean. Puffins are pelagic and more work is required to gain a better understanding of where they spend their time away from the colony.
One study in 1987 used direct tracking to study the foraging range of Puffins breeding on Isle of May, Scotland, using a VHF radio transmitter. Of the trips made 64% were within 2km, 7% between 2-10km and 29% more than 10km (Wanless et al., 1990). Transect surveys around the Isle of May also found that Puffins were most common close to the colony but on some occasions birds occurred at a sandbank, the Wee Bankie, 40km away.
After a crash of herring stocks near to the colony, Puffins at Røst Island, Norway were observed to travel at least 137km to fishing grounds (Anker-Nilssen and Lorentsen, 1990). Another study (Pearson, 1968) also estimated that Puffins breeding on the Farne Islands could forage a maximum distance of 137km from the colony. Based on average trip durations at UK colonies (various sources) and flight speeds of 82km/h one study suggested a potential range of over 200km from Great Island, Newfoundland, Canada (Bradstreet & Brown, 1985), which has now been supported by Birdlife International and Thaxter et al, (2012). Therefore, for the purposes of the Sectoral Marine Plan HRA, this mean maximum quoted foraging range has been assumed for Puffin (see Table G1).
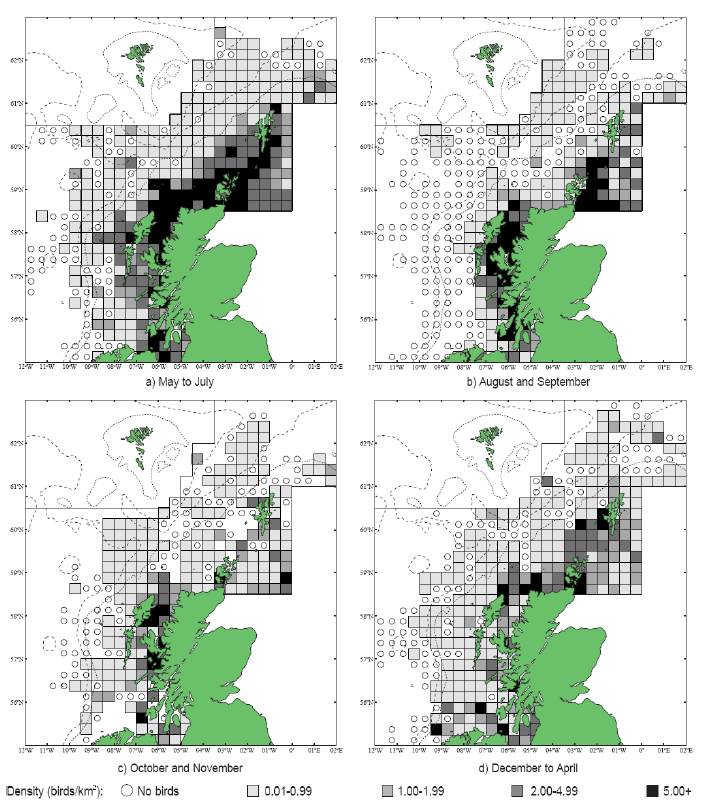
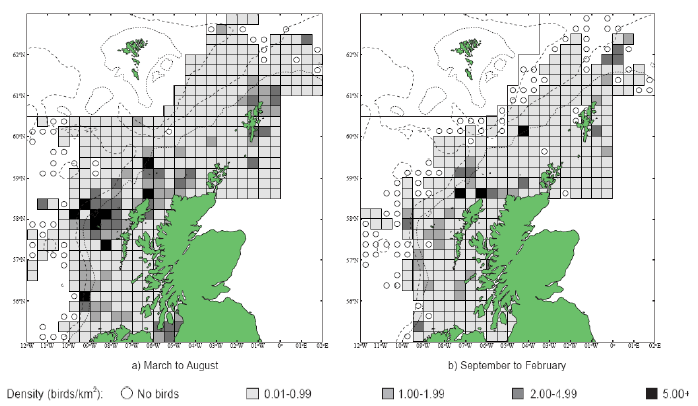
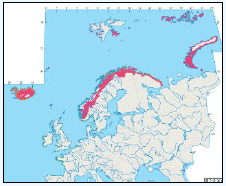
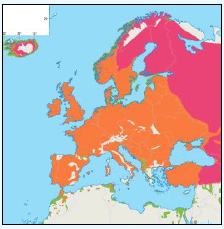
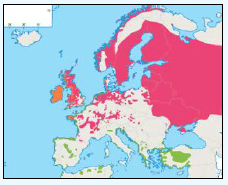
Puffins arrive during April and May for the breeding season, with a large percentage foraging over shelf waters (Image G5a). From June to July, highest densities of Puffins occurred with concentrations around the main breeding sites (Image G5b) of Shetland, Orkney, North Rona, the Shiants and St. Kilda (Pollock et al, 2000 and APEM 2012) with Orkney having particular importance to breeding auks, with an estimate by Mitchell et al (2004) of 250,000 individuals in the area. Image G5b demonstrates that low to moderate densities are observed beyond the shelf edge and over deep water as far west as the Rockall Trough and north to the Norwegian Sea. It is suspected that these are most likely non-breeding individuals as it is thought that breeders will generally forage nearer the colony. From August onwards the species are most widespread in deep waters (Image G5c), possibly due to leaving the colonies as the chicks have now fledged (Stone et al, 1995). October onwards birds leave the area and migrate south for the winter (Image D6d). The Pollock et al., (2000) data does not cover the east coast area but the OBIS mapping (Image G1) fills this gap to some degree and, in particular, shows how this area is also of high value for this species including especially the Forth Islands SPA site and the waters around it.
Images G5a-d: Puffin Distribution
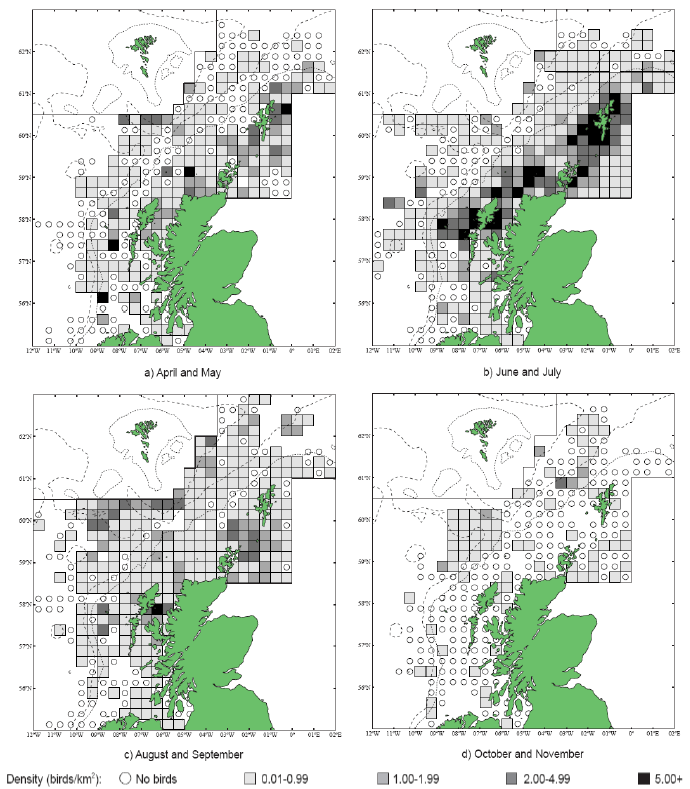
(Source: Pollock et al., 2000)
G.3.3 Black-legged Kittiwake Rissa tridactyla
The foraging range of Kittiwakes has been widely studied using a range of techniques. Radio tracked Kittiwakes were found to forage a maximum of 62km in Prince William Sound, Alaska (Ostrand et al., 1998), and over 40km at Sumburgh Head, Scotland (Hamer et al., 1993). Kittiwakes have also been calculated to forage 55km from a colony at Farne Island based on mean trip duration of 158 min during chick-rearing, and a flight speed of 48km/h (Pearson, 1968). A study at Prince Leopold Island, Canada used aerial surveys of Kittiwake densities. They found that during chick-rearing Kittiwakes were most abundant within 48km of the colony, although some large concentrations were found up to twice that distance away (Nettleship & Gaston, 1978). High concentrations of Kittiwakes have also been observed 20-30km from the nearest colonies at Caithness, Scotland (Mudge & Crooke, 1986) and at 30km from a colony on the Isle of May (Wanless et al., 1998). Both Daunt et al., (2002) and Humphreys et al., (2006) recorded that the maximum foraging range for Kittiwakes on the Isle of May was 83km.
Foraging distances have been shown to be dependent on prey availability. Suryan et al., (2000) found that increased foraging distances at two separate colonies at Prince William Sound, Alaska were associated with low prey abundance. In years where herring dominated diets mean distance to the farthest feeding location was 40km, whereas when Kittiwakes consumed other species foraging distance increased to 60km, with the maximum recorded foraging range being 120km (Suryan et al., 2000). GPS tagging of 464 Kittiwakes as part of the FAME/STAR projects recorded a maximum foraging range of 300km (Wakefield et al., 2017).
According to Thaxter et al. (2012) their mean maximum foraging distance is 60km and this value will be used for the purpose of screening the Sectoral Wind Plan HRA (see Table G1).
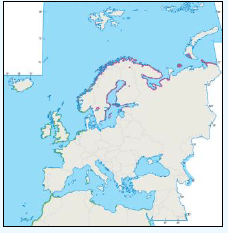
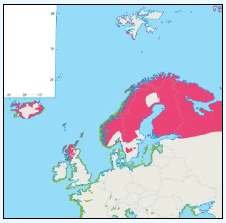
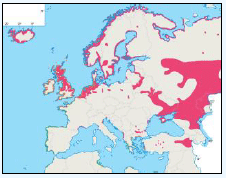
The distribution of Kittiwakes has been considered for the JNCC by Pollock et al 2000 and APEM 2012. From January to April highest densities were over the continental slope along the Rockall Trough (Pollock et al 2000) (Image G6a). Kittiwakes are scavengers and this area is associated with fishing vessels and so the high abundance in this area is most likely due to this. May to July Kittiwake distribution is much more coastal and close to colonies (Image G6b) as this is the breeding season. August and September the distribution is patchy, Image G6c. October to December Kittiwakes are in inshore waters west of Scotland, lowest numbers were recorded offshore at this time (Image G6d) (Pollock et al, 2000). Of the small gulls identified in the APEM (2012) report Kittiwakes were the most abundant species resulting in 72% of all small gulls recorded. Kittiwakes also had the most consistent pattern of abundance which is likely to be a consequence of their nomadic behaviour across the north Atlantic in the winter (Wernham et al, 2002). The Pollock et al., (2000) data does not cover the east coast area but the OBIS mapping (Image G1) fills this gap to some degree and, in particular, shows how this area and the coastal waters are also of high value for this species including those from a range of SPA sites along this coastline (e.g. Troup, Pennan and Lion’s Head SPA; Buchan Head and Collieston Coast SPA).
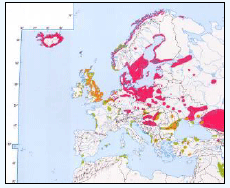
Important areas of high seabird density at sea, based on hotspot mapping techniques derived from large-scale seabird tracking as part of the FAME and STAR projects
emphasized the importance of areas along the entire east coast of Scotland and off the coast of Yorkshire (in proximity to where some of the largest Kittiwake colonies are located) for Kittiwake. In addition, hotspot areas were identified around the coast of Shetland, the Hebrides, Northern Ireland and North-East England (Image G7) (Cleasby et al., 2018; Wakefield et al., 2017).
Kittiwake foraging behaviour has been shown to be influenced by temporal cycles. For example, Trevail et al., (2019) found that Kittiwakes adjusted their foraging habitat selection according to the tidal cycle. Kittiwakes not only showed a preference for foraging nearer breeding colonies but that habitat selection changed over the 12.4-h tidal cycle, most likely because of changes in resource availability.
Spatial and temporal variations in migration has been linked to breeding success in Kittiwakes. Bogdanova et al. (2017) found that both successful and unsuccessful breeders wintered primarily in the north-west Atlantic. However, at some colonies, breeding success affected the timing of migration, whereby unsuccessful breeders departed the colony earlier, arrived at the post-breeding and main wintering areas sooner, and departed later the following spring.
Images G6: Map displaying hotspots identified at the UK-level for Kittiwake.
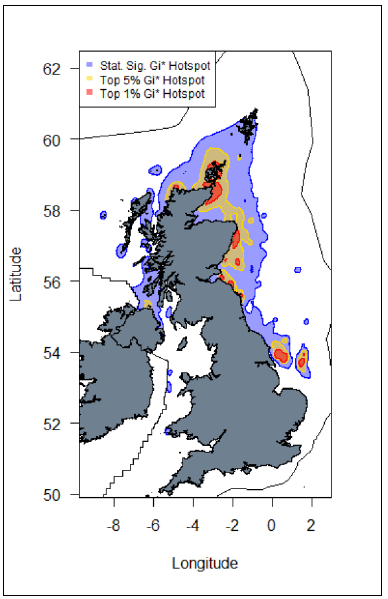
(Source: Cleasby et al., 2018)
Images G7a-d: Kittiwake Distribution
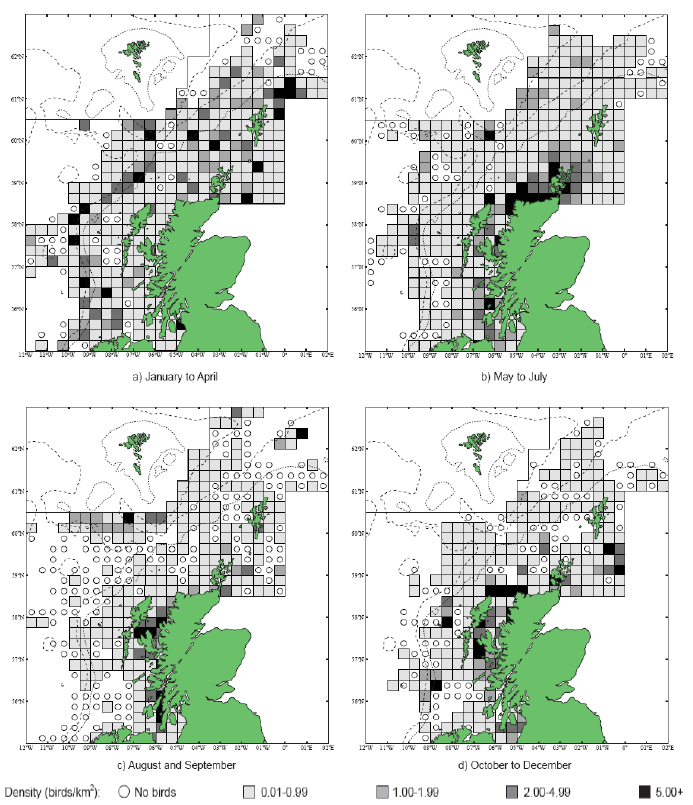
(Source: Pollock et al., 2000)
G.3.4 Common Guillemot Uria aalge
The foraging behaviour of auks, including foraging range, has been studied in depth for many years throughout the UK and North America using a variety of different methods. Numerous studies have taken place in Scotland, in particular at the Isle of May (Tasker et al., 1987, Thaxter et al., 2009; 2010, Wanless et al., 1990; 1998). The most recent of these surveys by Thaxter et al. 2009 and 2010, used bird-borne compass loggers to study foraging ranges. Thaxter et al., (2009) found that of 19 Common Guillemots breeding on the Isle of May (28 trips from 11 males plus 29 trips from 8 females), the maximum range for males was 14.4 ± 6.6km and 7.9 ± 5.3km for females. During a similar study Thaxter et al., (2010) observed 61 Guillemots travel an average distance of 34.5km/trip (±29.8), with the maximum observed distance from the colony being 14.1km (±12.2).
A study from North America (Cairns et al., 1987), used data loggers in combination with recorded flight speed, to estimate that the longest foraging range for Guillemots was 123km during incubation and 80km during chick rearing. However, median foraging ranges were much smaller.
GPS tagging of 178 Guillemots as part of the FAME/STAR projects recorded a maximum foraging range of 340km (Wakefield et al., 2017). The FAME project has had some recent findings using high resolution tagging with GPS tags which are able to record a bird’s location with a high frequency (as described above). Using this method it has found that the maximum known foraging distance for Guillemots is currently 340km. According to Thaxter et al. (2012) their mean maximum foraging distance is 84km and this distance has been used for the purpose of screening the Sectoral Wind Plan HRA (see Table G1).
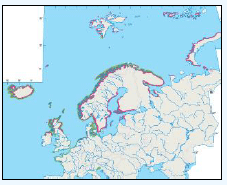
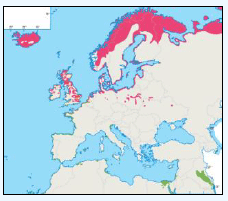
The JNCC have considered the distribution of guillemots in Scottish waters (Pollock et al, 2000), see Image G8. Near-shore concentrations were very high May to July with the highest densities of Guillemots found around Shetland, Orkney, along the northern coast of Caithness and Sutherland (Image G8a). It is thought that the reason for this is because this is during the breeding season and so birds are expected to stay relatively close to the nest with most breeding birds feed within 55 km of their colony (Leaper et al, 1988). August to September Guillemots undergo a complete body moult and so are unable to fly. During this time they gather inshore (Image 89b). There are fewer numbers present than during the breeding season, due to some Guillemots moving to the Irish Sea (Harrison et al, 1989; Pollock et al, 1997; Pollock et al, 2000), into the North Sea and Faroe Bank (Tasker et al, 1986). October to November the moulting flocks have moved offshore (Image G8c). December through to April Guillemots are more widely dispersed with the highest concentrations over the continental shelf (Image G8d). Although birds do visit colonies during autumn and winter months, highest densities were during April (Pollock et al, 2000).
Important areas of high seabird density at sea, based on hotspot mapping techniques derived from large-scale seabird tracking as part of the FAME and STAR projects
emphasized the importance of emphasized the importance of areas along the entire east coast of Scotland with other hotspots also evident around some of the larger UK colonies, e.g. Flamborough Head, Yorkshire or Rathlin Island, Northern Ireland. In addition, Guillemot hotspots in the Irish Sea were identified, including areas off the Pembrokeshire coast and Anglesey (Image G9) (Cleasby et al., 2018; Wakefield et al., 2017).
Images G8: Map displaying hotspots identified at the UK-level for Guillemot
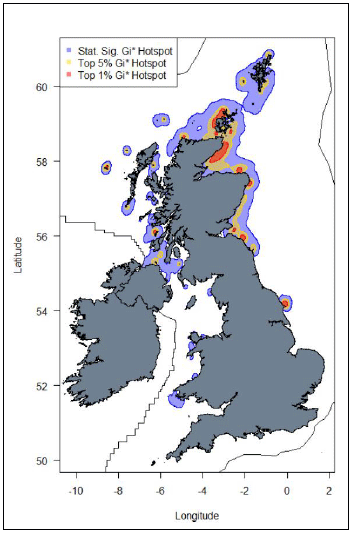
(Source: Cleasby et al., 2018)
Images G9a-d: Common Guillemot Distribution
(Source: Pollock et al., 2000)
G.3.5 Common Scoter Melanitta nigra
A number of studies into the foraging range of Common Scoter have taken place off the Welsh coast. At Llanfairfechan concentrations of birds were found between 1-4km offshore and at Red Wharf Bay the majority of birds were more than 3km offshore (Kaiser, 2004). In Conwy Bay birds were also recorded 3-4km offshore (Kaiser et al., 2006). However, studies over Lithuania and Estonia, using radar observations, recorded massive summer movements of Scoter with birds travelling 20-30km from the shore over the Baltic Sea (Kumari, 1979; Zalakevicius & Jacoby, 1992).
Based on an estimate by BirdLife International (2010), the maximum foraging range of Common Scoter has been given in Table G1 as 8km and this value has been used for the purposes of the Sectoral Wind Plan HRA. Therefore, this species is not included within the screening report as a long-distance foraging species that then requires the additional consideration of sites outside the 100km buffer area. It is worth noting however that a value of 200km is incorrectly quoted on the BirdLife International website and was used for screening the OWE HRA (ABPmer 2011). Care will be needed to avoid using this erroneous value in future project-level assessments and Plan reviews.
G.3.6 European Storm Petrel Hydrobates pelagicus
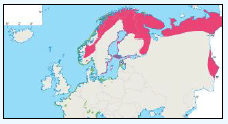
There is a lack of detailed information on the foraging ranges of the European Storm Petrel. A study by Thaxter et al., (2012) found that the maximum foraging distance of European Storm Petrels is >65km. Image G1 shows that there is a high abundance of European Storm Petrels off the North-west coast of Scotland, especially around Orkney and Shetland.
G.3.7 Great Skua Catharacta Skua
As with the Arctic Skua, there is little detailed information on the foraging ranges of Great Skua. A number of studies (Votier et al., 2004; 2006, Wood, 1989) have found that Great Skuas feeding on seabird prey tend to have small foraging ranges confined to the coastline of their breeding colonies, and seldom move more than 2km from their nest site (Votier et al., 2004). In contrast Great Skuas feeding on fish will often travel long distances to find them at sea. As such Thaxter et al (2012) has spilt this species into colony based foraging and offshore foraging activities. The study by Votier et al., (2004) used radio-transmitters to track Great Skuas at Hermaness. Those feeding on discards often flew several tens of km to fishing fleets off Muckle Flugga. Smith & Jones (2006) indirectly estimated a foraging range of 31km for Great Skua following the recovery of a bird ring from a Razorbill chick in a Skua pellet.
During the early-mid 1970s the estimated maximum foraging range of breeding Great Skua on Foula was 14km, based on the mean length of foraging trips of around 35 minutes (Furness, 1978). However, probably due to a reflection of changes in diet, foraging trip durations have become far longer in more recent years. Throughout the 1994 and 1995 breeding seasons, feeding trips were taking in excess of one hour, with a median length during chick-rearing of more than 3.5 hrs, and some breeding adults could easily have been ranging much further afield, perhaps 50-100km or more (BirdLife International, 2010). Increased foraging distances in more recent years compared with historic data was also identified by Wade et al. (2014).
As recorded by Thaxter et al¸ 2011 and 2012, the maximum distance for Great Skuas feeding on discards (offshore) is 219km. This distance has been rounded up to 220km for the purpose of screening this Sectoral Wind Plan HRA, (see Table G1).
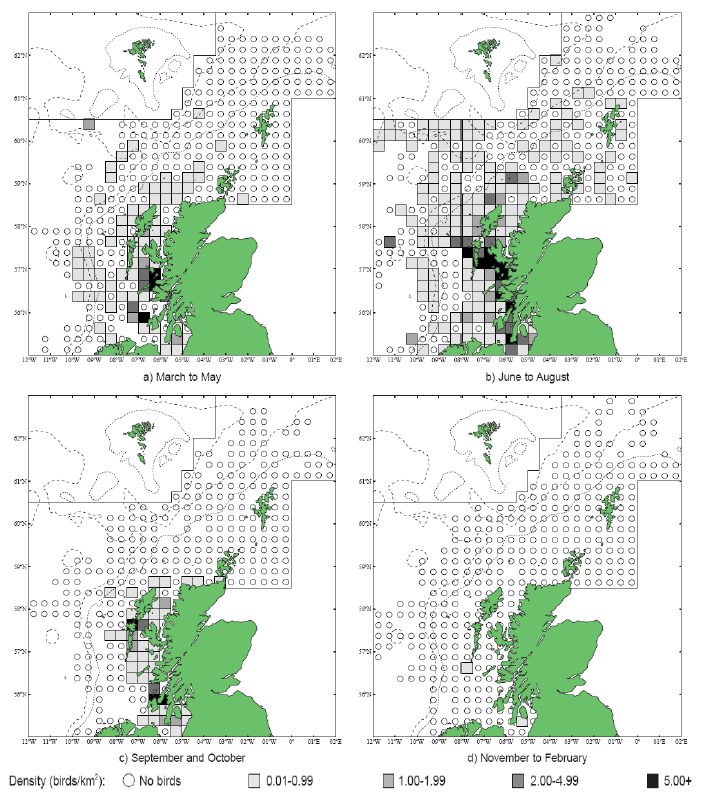
The JNCC have considered the distribution of Great Skuas in Scottish waters (Pollock et al, 2000). From April to May Great Skuas are widespread but in fairly low densities (Image G10a), with larger numbers around the colonies in Orkney and Shetland and in offshore waters Great Skuas are found along the shelf break. There is a high concentration of birds in the south-west area of the Faroe-Shetland Channel, where the birds are associated with fishing vessels (Pollock et al, 2000). The distribution of Great Skuas in June is still widespread inshore and around Orkney and Shetland (Image G10b) but the number offshore has reduced as mature birds will have started nesting at this stage (Pollock et al, 2000). Along the continental shelf around the Northern Isles low numbers of Great Skuas are recorded across a wide area (Image G10c), with moderate numbers recorded around the colonies as breeding birds forage in close proximity to the colonies. The offshore distribution of birds were again in low numbers and in waters closest to the Northern Isles possibly due to immature birds which visit club sites (sites made up of non-breeders) within colonies before departing in August (Tasker et al, 1985b and Pollock et al, 2000). Associations with fishing vessels are widespread north of Scotland although again the densities recorded are low. Fishing vessels and their discards form an important part of breeding Skuas diet (Pollock et al, 2000).
While the Pollock et al., (2000) data does not cover east coast but the OBIS mapping (Image G1) shows that while the PFOW area is of highest value (as indicated above by Pollock and JNCC studies), the east coast also supports areas of Great Skua foraging. This includes particularly the mouth of the Moray Firth. However, there are no European/Ramsar sites for this is an interest feature species outwith the PFOW area and Outer Hebrides (Handa and St Kilda SPAs).
Image G10a-e: Great Skua Distribution
(Source: Pollock et al., 2000)
G.3.8 Leach’s Storm Petrel Oceanodroma leucorhoa
There is a lack of detailed information on the foraging ranges of the Leach’s Storm Petrel. A study by Thaxter et al., (2012) found that the maximum foraging distance of Leach’s Storm Petrels is <120km and their mean maximum foraging distance is 92km.
G.3.9 Lesser Black-backed Gull Larus fuscus
Lesser Black-backed Gulls (LBBG) fly readily and strongly, at low and medium altitudes, and less frequently in upper airspace. Furness et al., (2013) recorded that an estimated 30% of LBBGs fly at blade height and they, amongst other large gull species, have a high collision risk with offshore wind turbines. ORJIP (2018) findings were higher again, showing that 67% fly at blade height and found that they also display variable flight patterns and foraging behaviour.
LBBGs have a wide breeding range which covers the area from Iceland, all the way east to the Taimyr peninsula and south to Portugal. They have colonies of breeding populations on the west coast of England and Wales and in Central Scotland, specifically Alde Ore Estuary and the Firth of Forth.
LBBGs are a highly migratory species outside of their breeding season, travellimg along the west coast of Europe and Africa, sometimes as far as Nigeria.
A study by Thaxter et al., (2012) found that the maximum densities of LBBGs were recorded foraging 181km from colonies. The mean maximum foraging distance was recorded as 141km and has been used for screening the Sectoral Wind Plan HRA.
Thaxter et al., (2015) made an interesting study of foraging behaviour of LBBGs from Alde-Ore, England using GPS-telemetry. These birds were tracked and only small spatio-temporal overlaps with the offshore wind developments were observed. Similar findings were also observed by Champhyusen (2011).
G.3.10 Manx Shearwater Puffinus puffinus
Guilford et al., (2008) made an interesting study of foraging behaviour of Manx Shearwaters from Skomer Island, Wales using GPS tags attached to individuals. These birds were tracked flying north to the Mull of Galloway to feed, travelling a straight-line distance of more than 330km each way. Another study by Brooke (1990) estimated that Manx Shearwater could travel as far as 360km from the colony during chick rearing, based on an absence of two days, a flight speed of 45km/hr and 8hr flying in each direction (although the values chosen for the time spent flying were arbitrary). A conservative estimate of 400km, as set out in Table G1, has been given as maximum foraging range for Manx Shearwater, based on the assessment by BirdLife International (2010). This value has been used for screening the Sectoral Wind Plan HRA.
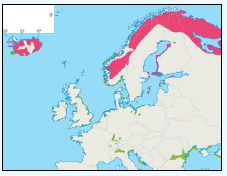
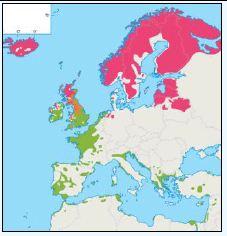
The JNCC have considered the distribution of Manx Shearwaters in Scottish waters (Pollock et al, 2000). They use this area to breed during the summer and start to return to the study area in March. Relatively low numbers were recorded during this time with the highest number recorded near Rum (Image G11a) which holds one of the largest Manx Shearwater colonies in the world with an estimated population of 61,000 pairs (Furness 1997) as in the Natura 2000 standard data form. Between June and August Manx Shearwaters are very widespread, as far west as Lousy Bank, but, with highest numbers recorded close to the colonies along the west coast (Image G11b). Birds were also recorded north-eastwards along the Faroe-Shetland Channel and east towards Shetland and Orkney (Pollock et al, 2000). It is supposed that sightings in these areas are due to the immature non-breeding birds (Brooke, 1990). From September and October, the numbers reduced dramatically (Image G11c) as birds migrate southwards. The birds that are still in the area were found to be restricted to inshore waters along the west coast, this population is made up of adults that have finished breeding and left the colonies and chicks that fledge in mid-September (Brooke, 1990). During the winter months, November to February, Manx Shearwaters are rarely recorded in the region (Image G11d) as most have migrated by this time, spending their winter off the east coast of South America (Brooke, 1990).
The Pollock et al., (2000) data does not cover the east coast area but the OBIS mapping (Image G1) fills this gap to some degree. It shows how this area and the coastal waters are also of relatively low value for this species and there are no designated SPAs along the east coast for which Manx Shearwater is a qualifying species.
Images G11a-d: Manx Shearwater Distribution
(Source: Pollock et al., 2000)
G.3.11 Northern Fulmar Fulmarus glacialis
Most of the information on Northern Fulmar (hereafter Fulmar) foraging ranges is based on trip durations, which, given the many assumptions involved in calculating ranges from such data, should be viewed as maxima. Foraging trip durations for Fulmars are variable among sites and years.
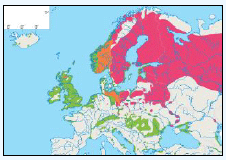
Hamer et al., (1997) studied populations of Fulmars on St Kilda, Outer Hebrides and Foula (Shetland). They estimated that the maximum foraging trip distances would be 245km for birds on St Kilda and 122km for Fulmars on Foula (Hamer et al., 1997). A study in Scotland also trapped one live breeding adult 460km from its nest site (Dunnet & Ollason, 1982) and in north east Greenland satellite tracked radio-tagged adults foraged 40-200km during incubation and chick-rearing, with failed breeders travelling more than 300km (Falk & Møller, 1995). Other authorities have given potential maxima of foraging radii of between 140km and 750km, assuming linear flight and negligible time spent on the water. While these will be overestimates of the radii travelled (as Fulmars deviate from straight line flight and spend time on the water feeding and resting, and may make overnight visits to feed chicks, which are often not recorded), they demonstrate that Fulmars have time to travel very long distances during foraging trips and could easily exceed a 100km radius from the colony (BirdLife International, 2010). As set out in Table G1, a maximum foraging range of 664km has been given for Fulmar based on BirdLife International (2010) data. A rounded up value of 670km value has been used for screening the Sectoral Wind Plan HRA.
The JNCC have considered the distribution of Fulmars in Scottish waters (Pollock et al, 2000). Prior to the breeding season (January to April) Fulmar distribution is concentrated along the continental slope (Image G12a), which was recorded as most obvious in April when the birds are known to leave the colonies in a ‘pre-laying exodus’ (Macdonald et al, 1977, Pollock et al 2000 and APEM 2012) and have a nomadic offshore distribution during the non-breeding season (APEM 2012). Many Fulmars along the continental shelf are associated with fishing vessels as they scavenge on discards (Pollock et al, 2000). The Fulmar breeding season is May to July where higher densities occur over the continental shelf (Image G12b). Beyond the continental shelf, Fulmar were recorded over the deep waters of the Norwegian Sea and the continental break north and west of Scotland (Pollock et al, 2000), this evidence that Fulmars will forage at large distances from the colony even during the breeding season. August to October (Image G12c) Fulmar distribution was largely in shallower waters, apart from the high numbers over the Wyville-Thomson Ridge and the shelf break to the north-west of Western Isles. In the shallow waters (<200m) there are areas of high density north of Scotland and around Shetland, suggesting this may be recently fledged birds (Pollock et al, 2000), although this may also be a reflection of fishing vessels in the area. Numbers are lowest during November and December (Image G12d). Ringing recoveries have suggested that much of the breeding population stay within a few hundred kilometres of the breeding colonies in winter, whilst the young travel over large distances during this time (Dunnet et al, 1979 and Pollock et al, 2000). During this time very few birds were recorded associated with fishing vessels.
The Pollock et al., (2000) data does not cover the east coast area but the OBIS mapping (Image G1) fills this gap and in particular shows how this species is widely recorded across the coastal and offshore waters of much of the SREZ. There are also several designated SPAs along the east coast for which Fulmar is a qualifying species.
Images G12a-d: Northern Fulmer Distribution
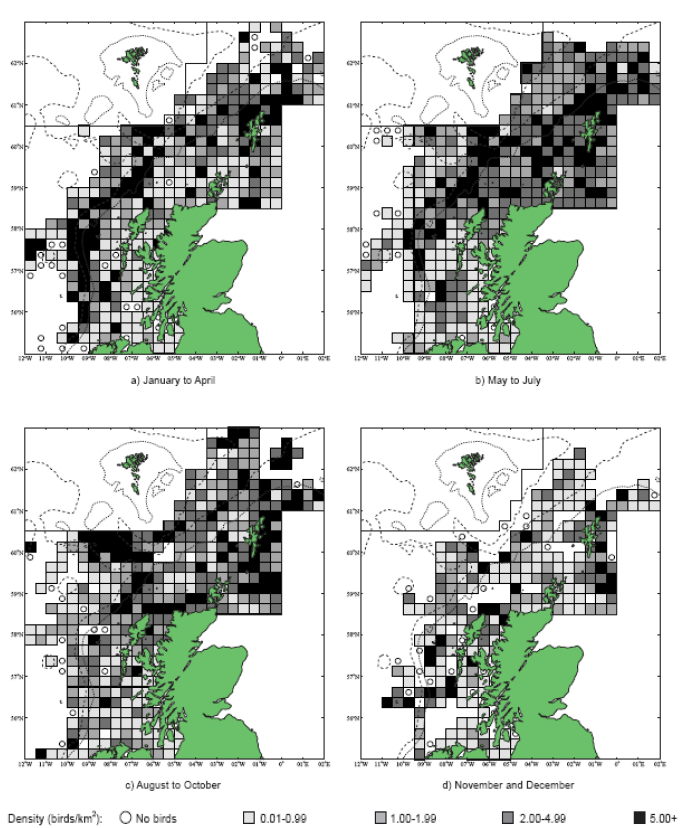
(Source: Pollock et al., 2000)
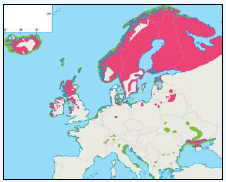
G.3.12 Northern Gannet Morus bassanus
Gannets breeding on Bass Rock, Scotland have been studied in detail using GPS transmitters (Hamer et al., 2000; 2001; 2006; 2007 and 2009). Hamer et al. (2001) recorded a maximum foraging range of 540km from Gannets at Bass Rock, Wanless et al. (1998) recorded feeding at sites 33km (Wee Bankie) and 20km (Fife Ness) away whilst another study (field work undertaken in July and August 2003) by Hamer et al., (2009) found the mean foraging range per trip by Gannets was 155km, with a maximum of 276km recorded.
Hamer et al. (2007) further investigated the foraging behaviour of gannets in the North Sea and found that they tended to forage repeatedly over a narrow range of bearings, despite marked annual variations in foraging ranges and trip durations (Image G13).
Image G13: Representation of One of the Maps Produced in Hamer et al. (2007) Indicating the Foraging Locations of Northern Gannet From Bass Rock in 2002
Hamer et al. (2001) also recorded gannets foraging repeatedly over a narrow range of bearings from their breeding location at Bass Rock. This study also observed gannets switching to a separate range of bearings in a markedly different direction from the breeding colony, providing strong evidence that on subsequent foraging trips individuals remembered the directions to feeding sites (Hamer et al., 2007). However, conversely, birds at a much smaller site in the Celtic Sea (Great Saltee) did not show this pattern (Hamer et al., 2001). Hamer et al. (2007) concluded that these differences in the consistency of foraging locations were related mainly to difference in the spatial and temporal predictability of prey resources in the North Sea and Celtic Sea.
Northern Gannets at Great Saltee, Ireland were recorded travelling a maximum of 240km (Hamer et al., 2001) and 293km (Hamer et al., 2006) to foraging grounds. At Rouzic, France a breeding colony of Gannets were found to have a mean foraging range of 100km, with a maximum of 176km (Grémillet et al., 2006). Large feeding flocks of Gannets were also recorded, using ship-based transects around St Kilda, within only a few km of Boreray whilst several smaller groups were seen 40km away (at the Whale Rock Bank) (Leaper et al., 1988). A study by Tasker et al., (1985a) also observed a maximum foraging range of Gannets to be 150km from Noss, UK, however the majority were seen within 37km. Nelson (1978) estimated a range of 320 to 480km for breeding Gannets from a colony, based on observed trip durations and assuming continuous flight at 65 to 80km/h.
Tagging of fourteen adult Northern Gannets from Bempton Cliffs found that most foraging trips from Bempton occurred within 50 - 100 km of the colony (Langston et al., 2013). Similar foraging distances were recorded by Soanes et al., (2013).The study tracked 15 Northern Gannets breeding on Les Etacs, Channel Islands, to determine their use of both local and international waters and examine the consistency between an individual’s foraging trips. The study found that the birds had a mean foraging trip of approximately 18 hours and had a mean foraging range of 106 km.
A study by Lewis et al., 2001 suggests that foraging range increases significantly for Northern Gannets with increasing colony size, probably as a result of intra-specific competition causing birds to forage further away from the colony during years with higher numbers of breeding Gannets (Lewis et al., 2001 & Thaxter et al, 2012). Northern Gannets from different colonies have also been shown to forage in largely mutually exclusive areas and that these colony-specific home ranges are determined by density dependent competition (Wakefield et al., 2013).
Based on an estimate by BirdLife International (2010) the maximum foraging range of Northern Gannet has been given in Table G1 as 640km. This value has been used for the Sectoral Wind Plan HRA.
The distribution of Gannets has been considered for the JNCC by Pollock et al 2000 and APEM (2012). Adults return to the region in March with immature birds arriving later in May (Pollock, 2000). Between March and August Gannets use a lot of the study area regardless of the water depth, with the highest concentrations of gannets found near the breeding colonies at Shetland, North Rona and especially St. Kilda (Image G14a).
It is likely that these are breeding birds and so will not be foraging at great distances from the colonies (Pollock et al, 2000) which is in contrast to the conclusion of APEM (2012) in the PFOW area who suggest that the individuals in this apparent population could be from a number of Gannetries as this species has a large foraging range. The distribution of Gannets during this time is often associated with fishing vessels as they scavenge the discards. During September and October Gannets leave the study area and so there are fewer numbers found during the winter months (Image G14b). Any birds that do remain the in the area stay around the colonies and along the continental shelf.
Images G14a-b: Gannet Distribution
(Source: Pollock et al., 2000)
G.3.13 Razorbill Alca torda
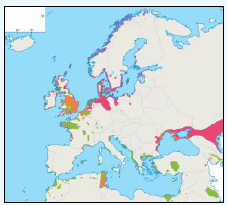
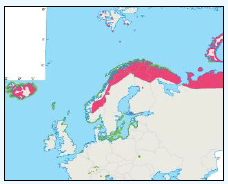
A study by Webb et al., (1985) at Flamborough Head found that maximum densities of Razorbill were recorded foraging at 26-28km from colonies, although large numbers were also seen from the coast within 1km from the colony. A more recent study by Thaxter et al., (2010) supports this conclusion. Using bird-borne data loggers to record foraging trips made by 14 Razorbills from the Isle of May breeding colony, they found that of the 76 foraging trips made just under half were within 10km of the coast, with the remainder of trips to areas 30-40km offshore, with the longest foraging trip being approximately 60km. Gaston and Jones (1998, cited in Thaxter et al., 2010) however, have reported Razorbills carrying prey more than 150km from the nearest breeding colony. This distance has now been superseded by the FAME project data and it is now considered that the maximum foraging range of Razorbill, as set out in Table G1 is 312km. FAME used GPS tracking data from Orkney during 2010 and 2011 and are displayed in Image G15. A rounded-up value of 320Km has been used as the maximum foraging distance for screening the Sectoral Wind Plan HRA.
Image G15: Razorbills Tracked From Orkney During the Breeding Season in 2010 and 2011
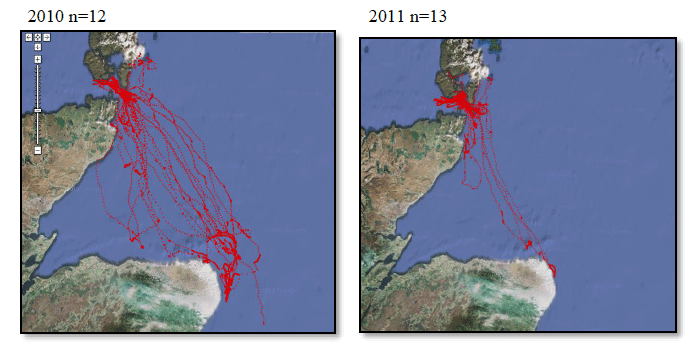
(Source: FAME project.)
The distribution of Razorbills has been considered for the JNCC by Pollock et al 2000 and Scottish Government (APEM, 2012). During the breeding season (May-July) highest densities of Razorbills are found inshore off the west coast of Scotland (Image G14a). During August there is a high proportion of Razorbills inshore on the west coast of Scotland (Image G16b). In the PFOW area, Orkney in particular is considered to be of great importance to breeding auks with an estimate by Mitchell et al (2004) of 250,000 individuals in the area.
Images G16a-d: Razorbill Distribution
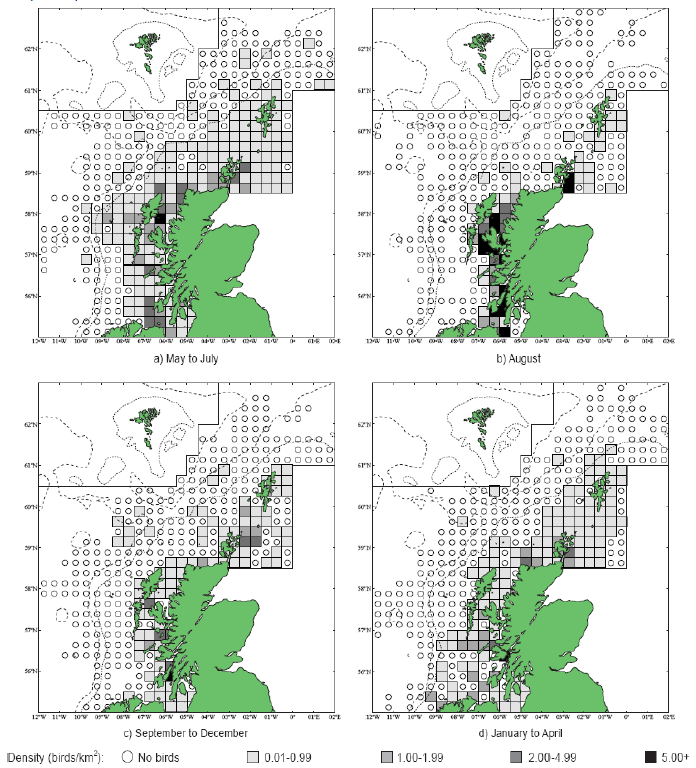
(Source: Pollock et al., 2000)
The Minch is an important area for moulting Razorbills with an estimated two thirds of the west coast population congregating in the area during this time (Pollock et al, 2000). From September to December razorbills start to spread out in the study area once again and forage offshore (Image G16c). During the winter early spring, the distribution is fairly evenly spread along the continental shelf with birds tending to stay close to shore (Image G16d).
The Pollock et al., (2000) data does not cover the east coast area but the OBIS mapping (Image G1) shows how this area is also of high value for this species. There are also several SPAs (including Forth Islands SPA) along this coastline for which Razorbill is a qualifying interest feature species.
Important areas of high seabird density at sea, based on hotspot mapping techniques derived from large-scale seabird tracking as part of the FAME and STAR projects emphasized the importance of areas along the east coast of Scotland and the Orkney Islands as well as hotspots in the Hebrides and around Foula and Shetland. Outside of Scotland, hotspots were identified along the Northern Irish coast, around the Yorkshire coast in England and around the Pembrokeshire coast in Wales (Image G17) (Cleasby et al., 2018; Wakefield et al., 2017).
Image G17: Map displaying hotspots identified at the UK-level for Razorbill
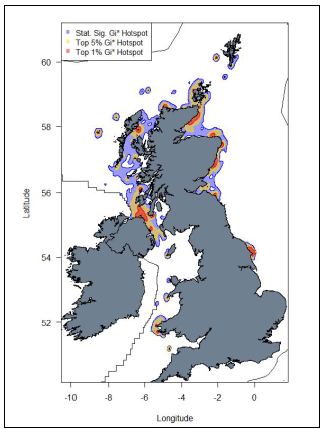
(Source: Cleasby et al., 2018)
G.3.14 Velvet Scoter Melanitta fusca
The Velvet scoter is seen in small numbers around Britain, mainly off the east coast. It is known to have difficulty in becoming airborne and commonly flies low, especially over water.
A recent draft plan (AEWA, 2018) highlights specific management measures for designated sites to ensure the protection of the Velvet Scoter in the UK.
G.4 Migration Behaviour and Flyways
Seabird species undertake large scale seasonal migrations outside of the breeding season (Furness, 2015). Within the same local populations or breeding colony, different non-breeding strategies can be employed. For example, Quinn (2014) found that individual differences in winter distribution existed within a single North Sea Fulmar colony, with a higher proportion of females than males utilizing the West Atlantic region, compared to local North Sea waters. Older individuals were more likely to remain within North Sea waters than travel further from the colony. The distribution of auk species outside of the breeding season during moulting was investigated by Glew et al (2018). The study aimed to quantify species-specific foraging locations and individual trophic variability during feather regrowth in three sympatric auk populations breeding on the Isle of May, Scotland (Common Guillemot, Razorbill and Atlantic puffin). Foraging areas during moult differed between species and feather types. Guillemots likely underwent moult within the southern North Sea, Razorbills along the east coast of England and into the southern North Sea and Puffins off the east coast of Scotland. Estimates of individual trophic position varied considerably within feather types (up to one trophic level difference between individuals), among feather types grown during different time periods and across the three species, with Guillemots consistently foraging at higher trophic positions than Razorbills and Puffins.
The UK lies on some of the major migratory flyways of the east Atlantic, with large numbers of coastal waterbirds attracted each year by the relatively mild winter climate and extensive estuarine and wetland habitats. Every year, large numbers of birds traverse the North Sea and Irish Sea during the spring and autumn migrations on their way between breeding and wintering grounds. There appear to be no fixed corridors preferred by migratory birds and instead migration usually takes the form of broad-front. There can be high intensity migration (i.e. a few days with extremely high numbers) and flight heights can vary depending on the species and weather conditions (DECC, 2009). Heights can range from just above the water surface to several thousand metres and as migration is costly in terms of energy expenditure, birds will generally try and fly at altitudes where effort is least (Köller et al. 2006).
The height a given species flies at can influence the risk of collision with wind turbines (Furness et al, 2012). Recent studies have investigated the use of LiDAR to determine concentrations of seabirds flying at heights which may place them at risk of collision from wind turbines (Cook et al., 2018).
The issue of potential impacts to migratory birds is key for the Sectoral Wind Plan HRA and it is understood that there is a limited amount of robust data on this aspect. This is recognised within the SEA for the Draft OWE Plan (Marine Scotland, 2010) which records that there is a lack of accurate data showing migratory routes and this has also been confirmed via correspondence with the RSPB and SNH as part of consultations . It is noted however, that the Wildfowl and Wetlands Trust (WWT) are researching migration routes of several bird species in relation to proposed UK wind farm sites. The WWT work included mapping routes of Greenland Barnacle Geese, Greenland White-fronted Geese and Whooper Swans (Griffin et al., 2010) from or along the west coast of Scotland to Iceland and Svalbard Barnacle Geese routes from South-West Scotland to Norway and Svalbard (WWT, 2010). Geese and swans were the focus for this research as they tend to fly at less than 100m above sea level and could be at risk of collision with wind turbines. This study was commissioned by COWRIE and involved a tracking study of Whooper Swans to analyse flight heights and migration routes between their breeding grounds in Iceland and the UK in relation to offshore wind farms. The project entailed fitting satellite transmitters to forty known Whooper Swans in winter 2008/2009 at various wintering locations in mainland Britain. These included WWT sites at Caerlaverock (SW Scotland), Welney (SE England) Martin Mere (NW England). Of the 40 individuals tagged, 35 were tracked to Iceland during spring migration 2009. These included 5 birds from Caerlaverock, 13 from Welney and 17 from Martin Mere. In addition, 10 transmitters were fitted in August 2009 in Iceland to provide information during autumn migration of the swan’s southbound tracks.
As part of this summary review, key scientific literature relating to migratory pathways has been further reviewed to provide additional information on the routes that are taken. In particular, the above study by Griffin et al. (2010) into migration routes and flight heights of Whooper Swans has been reviewed here. In addition, the review by Cramp (2000) and the literature that has been compiled as part of this review was summarised to identify the locations of migrant, resident and winter distributions for a range of bird qualifying features. This review is presented in tabular for at the end of this appendix and includes maps of bird distributions that are provided within the Cramp (2000) review. Recently there have been reviews of flight heights, avoidance rates and seabird vulnerability and sensitivity to offshore wind farms (Cook et al, 2012, Marine Scotland, 2012, Furness et al, 2012). A summary of these is also provided below.
G.4.1 Flight Heights
Flight height appears to be a key factor influencing the collision mortality risk of seabirds colliding with wind turbines. Griffin et al. (2010) concluded that on average, the flight heights for swans were lower for birds migrating over water along the British coast (9m) compared to when crossing from Britain to Iceland (32m), thus flights occurred below or within the rotor swept area and so potentially at risk of collision. Two records however may provide evidence of avoidance of offshore windfarms. At 8.9km north of Barrow (operational) and 3.8km north of Robin Rigg (near operational) swans were recorded flying at altitudes of 172 metres and 160 metres respectively, where one hour previously their flight heights had been recorded at approximately 50 metres above land (Griffin et al., 2010).
A study into seabird vulnerability reviewed collision index calculations and identified populations of gulls, white tailed eagles, northern gannets and skuas as being of particularly high concern in Scottish waters (Furness et al, 2013). This index is influenced by the flight height of species. In contrast, when considering the displacement index calculations, different species were identified as most vulnerable, namely divers and common scoters. Collision is more likely to occur if seabirds fail to avoid the wind farms (displacement), whereas displacement from foraging habitats is more likely to occur if seabirds can avoid wind farms, i.e. displacement is a function of avoidance behaviour. There are two kinds of avoidance observed by birds; ‘macro-avoidance’ when birds alter their flight path to completely avoid the whole wind farm; and ‘micro-avoidance’ when the bird will fly into the windfarm but acts to avoid the individual turbines. If species specific rates of macro- and micro-avoidance behaviour were known, a clearer understanding of species specific vulnerability would be understood. In addition, if more robust data were gathered regarding bird flight height, the most ‘at risk’ populations could be a focus in the monitoring and mitigation of these developments.
Cook et al., (2018) used LiDAR and digital aerial imaging to collect estimates of the altitude of birds in flight. The study successfully collected flight height information on 2,201 birds of which 806 were believed to be black-legged kittiwakes and 377 were identified as northern gannets. This data was used to derive continuous flight height distributions for these species. The study found that the proportion of birds recorded at collision risk height (20-120 m) was 0.081 and 0.043 for Gannet and Kittiwake respectively based on all surveys undertaken.
G.4.2 Migration Routes
Griffin et al. (2010) found that swans migrating from southeast England (Welney) generally migrated north along the east coast of Britain whereas those from northwest England and southwest Scotland (Martin Mere and Caerlaverock) followed the western coastline (see Image G18).
Image G18: Migration routes of 35 Whooper Swans tracked from the UK to Iceland from WWT Wetland Centres at Welney (red lines), Martin Mere (blue lines) and Caerlaverock (green lines)
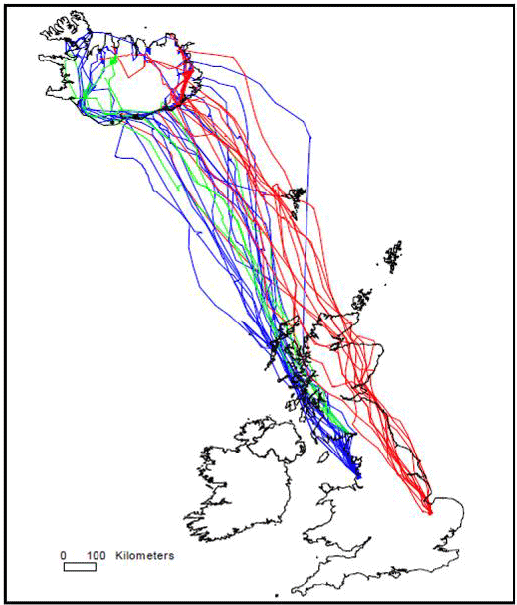
(Griffin et al., 2010)
The swans’ satellite tracks were inspected in relation to the distribution of the potential and actual footprint areas of individual windfarms in six key overlapping areas: the East Irish Sea, the Solway Firth, the North Channel/Inner Hebrides, the Greater Wash/North Sea, the Firth of Forth and the Moray Firth. A total of 48 Round 1 (R1), Round 2 (R2), Round 3 (R3) and Scottish Territorial Water (STW) offshore sites were considered in the analysis.
Of the 19 Whooper Swans tagged at Martin Mere seven flight lines (37%) across the East Irish Sea passed across or immediately adjacent to existing or proposed R1 and R2 windfarm sites with an additional eight tracks (42%) passing within 5km of these sites. Whilst up to ten flight lines (50%) crossing the Solway Firth passed across or immediately adjacent to the existing or proposed R1 or STW windfarm sites with another two (10%) passing within 5 km of these sites. Of these birds crossing the North Channel/Inner Hebrides area no birds were likely to have flown across or adjacent to (within 5km) of STW sites. Of the 15 swans tagged at Welney only one bird stopped and flew across a proposed STW site (Neart na Gaoithe) and one bird flew within 4km of the site proposed at Westermost Rough (R2).
Of the eight birds producing reasonably detailed tracks during the return autumn migration from Iceland, five birds passed across or immediately adjacent to planned offshore windfarm sites with six birds passing within 10km of the planned sites.
Griffin et al. (2010) concluded that swans migrating along the east coast of Britain tended not to pass within or near the footprints of existing or proposed offshore windfarms, preferring to stick close to the coast or overland. However, swans migrating along the west coast more frequently flew into or close to the footprints of several offshore windfarms, preferring to fly over coastal waters. Although this tracking study is not representative of the migration pathways taken by Whooper Swans every year, it is a useful study to add to understanding and risk evaluation to birds arising from windfarms.
Information and knowledge on specific migration routes is also expanding rapidly with recent technological advances and through the application of useful tools (e.g. Burger & Shaffer, 2008). However, it is recognised that identifying distinct flyway routes is difficult both because of the nature and limitations of available information on this subject and because these movements are likely to occur across broad fronts rather than along clearly definable routes. Although little information is known about actual routes taken between breeding and non-breeding areas, a relatively large amount of information is known about the origins and destinations for many bird species, through data from the UK ringing scheme (Langston, 2010 - Wernham et al., 2002, BTO Migration Mapping Tool http://blx1.bto.org/ai-eu/). The destinations outside of the UK where tagged birds migrate to and from Scotland has been reproduced in Table G2. This table is derived from information within the BTO Migration Atlas Project report (Wernham et al., 2002) and is presented within the SEA Environmental Report (Marine Scotland, 2010). As recognised within the SEA document, this information does not specifically describe the migration routes but presents a useful collection of ringing recovery information ‘for birds that fly to and from Scotland (not necessarily from other parts of the UK)’.
Table G2: Principal recovery locations for likely focal bird species
| Species | Principal Recovery Locations of Ringed Birds Outside of Mainland UK |
|---|---|
| Arctic Skua | Concentrated around Orkney and Shetland Isles |
| Arctic Tern | Concentrated around Orkney and Shetland Isles, Scandinavia, W Africa |
| Atlantic Puffin | Canary Islands, Mediterranean |
| Barnacle Goose (Nearctic) | Iceland, Greenland, Svalbard |
| Black-legged Kittiwake | USA, Greenland, Faroes, Continental Europe |
| Brent Goose (light bellied, E. Canada) | Svalbard |
| Common Tern | Continental Europe, Ireland, W Africa |
| Common Guillemot | Continental Europe, Scandinavia, Ireland, Orkney and Shetland Isles, Iceland |
| Common Scoter | Moray Firth, Firth of Forth, Northern Europe, North Africa. |
| Corncrake | Unrecorded in Bird Atlas |
| European Storm Petrel | Ireland, Iceland, Faroe Islands, Scandinavia, Portugal, S Africa |
| European Shag | Ireland, Faroe Islands, Continental Europe, Scandinavia |
| Great Northern Diver | Unrecorded in Bird Atlas |
| Greenland White Fronted Goose | Iceland, Greenland, Orkney |
| Herring Gull | Ireland, Orkney, Shetland Isles |
| Lesser Black-backed Gull | Northern Ireland, Firth of Forth, Alde Ore Estuary, Africa |
| Manx Shearwater | Ireland, Continental Europe |
| Migrating waterbirds | Various destinations |
| Northern Gannet | Ireland, Iceland, Continental Europe, Orkney, Shetland Isles, northern Scandinavia, NW Africa |
| Pink-footed Goose | Iceland, Greenland |
| Razorbill | Continental Europe, Scandinavia, Ireland, Orkney and Shetland Isles |
| Velvet Scoter | Firth of Forth, Tay and Eden Estuary, Moray and Naim coast, Northern Europe, West and Central America |
| Whooper Swan | Ireland, Western Isles, Orkney |
(Marine Scotland, 2010)
Further details and maps describing the origins and destinations for many bird species are also presented in Table G3 which provides general information on migrant, resident and winter distributions around the UK and the rest of Europe for many bird species (Cramp et al., 2000). The birds listed in this table have been included as they have been identified as qualifying features within SPA sites within 100 km of the DPOs.
Table G3: Migrant, resident and winter distributions, as well as general flight heights, for a number of bird species
| Species | Summary Information |
|---|---|
| Common Redshank | Migrate between Iceland and Britain (Orkney to Hampshire) (Ogilvie 1963; Hale 1973; Folkstad 1975; Minton 1975; Boere 1976; Holden & Cleeves, 2002). Some birds from Scotland and northern England winter in Ireland, but southward movement within Britain more usual, and others continue south to continental coasts (Netherlands to Portugal, especially western France). Flies freely in lower airspace. |
| Great Crested Grebe | As few ringed, little known of migration routes, though apparently broad-front overland as well as coastal. Some autumn and winter immigration into British waters from Denmark and Netherlands. Often active aerially on moonlight nights, visiting neighbouring waters in search of mate or territory. Uses airspace somewhat sparingly, although strong flier once airborne, not often at much height. |
| Whooper Swan | Migrate between Iceland and Britain (Pennycuick et al., 1996; Holden & Cleeves, 2002). Once thought to fly at high altitudes between Iceland and the UK; more recent evidence shows that the birds also migrate at just above sea level, and land on the sea in adverse weather or poor visibility (Pennycuick et al., 1996). |
| Barnacle Goose | East Greenland population migrates via Iceland to winter in west Scotland and Ireland (Salomonsen 1967; Holden & Cleeves, 2002). The Svalbard population moves down the west coast of Norway and across the North Sea, crossing overland in north England and South Scotland to winter in Solway Firth (Holden & Cleeves, 2002). |
| Greylag Goose | Migrates between Iceland and Scotland and Ireland (Mitchell 1996; Hearn 2000; Swann and Brockway 2002; Swann 2000; Mitchell and Sigfússon 1999; Cranswick et al., 1997; Holden & Cleeves, 2002) |
| Greenland White-fronted Goose | Geese collared in Scotland, Iceland, and Greenland as part of continuing international marking programme. |
| Pink-footed Goose | Migrates between Iceland and Britain (Fisher 1951, Thom 1983, Newton et al., 1990, Dix 1991, 1992; Fox et al., 1992; Holden & Cleeves, 2002) |
| Light-bellied Brent Goose | Migrates between Spitsbergen and Franz Josef Island to Britain and Denmark (Løvenskiold 1964) Although strong flier, takes wing with some reluctance, and flies no higher than essential. |
| Bar-tailed Godwit | Winters North Sea and Atlantic coasts of Europe. Major passages in spring and autumn are through Baltic and North Seas and thence along western seaboard. Exceptional flight capability used at relatively low height except on migration. |
| Eurasian Wigeon | Migrates between Iceland and Britain Fenno-Scandia and USSR east populations migrate to winter quarters in west and south-west Europe, especially Britain and Ireland. Usually fly at no great height. |
| Northern Pintail | Migrates between Iceland to Britain and Ireland (Bruun 1971; Holden & Cleeves, 2002). Breeding populations of north Russia east to north-west Siberia (Omsk), Fenno-Scandia, and Baltic migrate south-west to winter Netherlands and British Isles. Use of airspace even freer than for other Anas and often at considerable heights. |
| Red Knot | Migration occurs from Canada and Greenland to western Europe, including Britain and southern north sea (Andreasson and Råd 1977; Netterstrøm 1970; Holden & Cleeves, 2002) Winter range of Spitsbergen birds unknown. |
| Purple Sandpiper | Migrates between Iceland and Norway to Britain (Atkinson et al., 1981; Holden & Cleeves, 2002) Not known to fly regularly in upper airspace. |
| Ruddy Turnstone | Migration occurs between Northern Europe, Greenland and Canada to Britain and Ireland (Holden & Cleeves, 2002; Morrison 1975; Clapham 1979; Wilson 1981; Branson et al., 1978) Also a strong flier, most often within lowest airspace. |
| Slavonian Grebe | Breeding populations in Iceland, Norway and Scotland winter around Scotland and Atlantic coast of Ireland. Flies readily, visiting neighbouring waters but observations of high flight lacking |
| Red-throated Diver | Migration routes not well known. Migration occurs between Spitsbergen, Lapland and USSR to Atlantic and North Sea coasts around Britain and Ireland. 2 chicks from Scotland (Shetland) also found French coast (Finistère, Vendée). Migrants from Europe and Iceland winter around British and Irish coasts (Holden & Cleeves, 2002) Entirely aquatic and aerial, normally on land only for nesting. Inadequacy of food on small breeding pools requires frequent flighting to better fishing lakes or sea, involving dual aquatic habitat and much use of airspace up to at least 1000 m. Mobility aided by ability to rise readily from small water surfaces, and exceptionally from land. |
| Common Eider | Partially migratory. Small movement within British and Irish breeders, rarely over 200km (Milne 1965a). Recoveries of Dutch birds in Fife. British and Irish birds do not generally move far from their breeding grounds (Holden & Cleeves, 2002). Although flying freely, normally keeps to lowest few metres of airspace, mostly over water. |
| Common Shelduck | Breeders and immatures leave nesting grounds in Britain, Ireland, Belgium, Netherlands, Denmark, Scandinavia, north Germany, north Poland, and Baltic States, for annual moult in German Waddenzee area (Goethe 1961a). Birds from Germany and farther east arrive in Britain in winter (Holden & Cleeves, 2002). Flies strongly, rising on passage to 1000 m or more above ground. |
| Great Cormorant | British and Irish populations non-migratory but show extensive dispersal with small proportion crossing English Channel; mainly coastwise, but also inland usually within 60 km of coast (Mills 1965). Recoveries in France and Iberia (mainly Brittany to north Portugal) continuance of southward dispersal in Britain and Ireland, and originate mainly from Irish Sea and Galloway. North Sea effective barrier to eastward movement from Britain, recoveries Norway to north France being quite exceptional. Circles or soars up to considerable heights and flies overland, often for long distances, occasionally or regularly. |
| Mallard | Many Icelandic birds winter Britain and Ireland. Those from north-west Russia, Fenno-Scandia, Baltic states, north Poland, north Germany, and Denmark winter from Denmark to north France and in Britain, with slight onward passage as far as north Spain. Many birds that breed in Iceland and northern Europe spend the winter in Britain and Ireland (Holden & Cleeves, 2002). Can rise steeply up and away even in awkwardly confined spaces; much given to circling around, usually at heights below c. 500 m. |
| Greater Scaup | In Iceland, ringing of young showed migration chiefly to Ireland, Britain, and Netherlands (Boyd 1959), with smaller numbers reaching north France, Denmark, and north Germany. Important winter concentrations in Firth of Forth (Scotland). Take-off somewhat laboured, and use of airspace relatively infrequent. |
| Long-tailed Duck | No evidence of Icelandic birds reaching continental Europe; yet ones ringed west Greenland moved south-east to Iceland and Denmark, and to Atlantic seaboard of North America, while others wintered in Greenland seas. Origins of British winterers unknown. Makes maximum use of airspace, readily flying between coast and inland sites at up to 200-1200 m over land, although usually low over water. |
| Black (Common) Scoter | Icelandic breeders winter around Ireland, Britain, and Atlantic coasts France, Spain, and Portugal Movement by small British and Irish population largely unknown, but breeding lakes deserted. Breeders from Arctic USSR, Sweden and Finland migrate WSW to winter on both sides of North Sea (west coasts of Britain). Uses airspace often in long formations, and normally at low altitude, but flies fairly high overland; generally flies only when need pressing. |
| Velet Scoter | Small numbers winter around Britain (mainly east coast) Has difficulty in becoming airborne and commonly flies low, especially over water. |
| Common Goldeneye | Breeding populations of Fenno-Scandia, north Germany, Poland, Baltic States and north-west Russia mostly winter Baltic States, Denmark, Netherlands, Britain and Ireland. Airspace over water and land freely used at wide range of altitudes. |
| Red-breasted Merganser | Breeding population of Iceland partially migratory; some resident, others migrate to Britain and Ireland. Movements of British breeders not fully known, but at least disperse from inland breeding waters. Migrants from Finland, Poland and north-west USSR winter in Britain. Spends much time in aerial excursions, usually at fairly low altitude. |
| Eurasian Curlew | Large resident population in Ireland, with local movements to coast, though many also winter inland; movements unconfirmed in absence of ringing. Some British birds winter near breeding site, though most move south-west. Thus birds from Scotland and northern England move to British west coast and Ireland; those from southern England to south-west England, some to France and Iberia (Bainbridge and Minton 1978). Scandinavian population moves south-west: Norwegian birds to northern Britain, Ireland, and western France; main wintering area for Swedish birds is Britain and Ireland, though also some on Continental coasts from Denmark to western France (Nørrevang 1959). Some USSR, German and Dutch populations also reach Britain and Ireland (Holden & Cleeves, 2002). Except on migration flies mainly in lower airspace. |
| Eurasian Oystercatcher | Birds reared Iceland, Faeroes, and northern Britain moult and winter mainly around Irish Sea and on other western coasts of Britain and Ireland; few reach North Sea coast (Dare 1970; Dare and Mercer 1974b; Anderson and Minton 1978). Some British birds, especially in southern England, remain near breeding areas, but the general trend for these to move south to moult and winter takes some to France and Iberia and exceptionally to Morocco (Buxton 1957; Andrew 1959; Dare 1970; Pienkowski 1972). Migration also noted frequently across southern Scotland, between Firth of Forth and Solway Firth (e.g. Andrew 1959), and west of south from north-east England (Evans 1966); by no means certain, however, that Continental immigrants predominate in such movements. 3 recoveries of birds moving between eastern England (all autumn dates) and USSR (Murmansk, Arkhangel'sk), but clearly these atypical. Considerable movement may occur within a linked estuarine complex, such as Wash-Humber system (England), though apparently little movement between sites within Firth of Forth (Scotland) (e.g. Minton 1975; M W Pienkowski). Although a strong flier, shows marked reluctance to venture out of sight of land, and usually travels within lower airspace. |
| Ringed Plover | Largely migratory but some British birds resident. North-western populations of Canada and Greenland, majority pass through Britain and Ireland European populations - Birds from coasts of Scandinavia and Baltic and from East and West Germany south-westwards move widely to wintering areas extending from Britain and Ireland to West Africa. |
| European Golden Plover | Partially migratory in Britain and Ireland; wholly migratory elsewhere. 23 recoveries of birds ringed Britain as chicks, 21 recovered within Britain, so only minority emigrate. British internal recoveries showed mean movement of 115 km (0-500). Icelandic population (115 ringing recoveries) wholly migratory, with western route to and through Ireland, western Britain, western France, and Iberia. Norway to Western Siberian birds, many found in Britain between November to April Use of airspace (especially above the lowest) normally confined to migration, advertising flights over territory, etc. Wading also infrequent. |
| Grey Plover | Those occurring in west Palearctic come from breeding population of northern USSR. In Britain, arrives July onward through autumn and winter (Mason 1969, Mason 1972; Branson and Minton 1976). Wariness leads to frequent use of outstanding powers of flight, mostly in low airspace but migrating flocks fly high in massed formations (Bent 1929). |
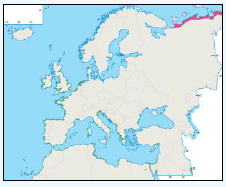
| Northern Lapwing | Britain and Ireland constitute northernmost regular wintering areas. In contrast to continental birds, British breeders mainly make short summer dispersals (within 100 km), though some indication of adult movement west to south-west into Ireland. Birds from Hungary, Czechoslovakia, Switzerland, and southern West Germany move to Britain. British Isles especially important as winter quarters for Scandinavian birds; south-westerly movements by those from south and east Baltic countries largely bypass Britain. In flight, lower airspace usually preferred except on movements, when altitudes of 250-400 m are common and up to 2000 m or more recorded (Meinertzhagen 1920). |
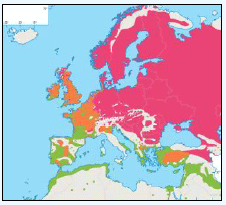
| Dunlin | Holarctic breeding distribution, wintering on most ice-free coasts of northern hemisphere Iceland to Britain and western France (Rosenberg et al., 1970; Meltofte 1975; Pienkowski and Dick 1975; Green 1978; Hardy and Minton 1980). Baltic region to Britain (Minton 1966, Minton 1975; Pienkowski and Dick 1975). Flies much in lowest airspace, infrequently higher. |
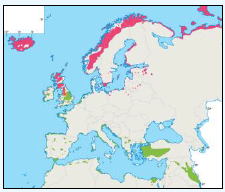
| Sandwich Tern | Some birds from Britain and Ireland disperse to Netherlands, and vice versa (Møller 1981a). Recoveries of British and Irish birds extend from France to Angola, with 30% of recoveries in Sénégal (Langham 1971; A P Møller). In some circumstances, as in display, rises into upper airspace, and normally flies somewhat higher over water than most seabirds. |
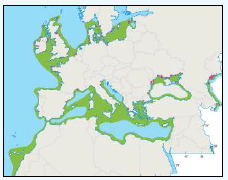
| Sanderling | With Holarctic breeding range, winter distribution very large: both sides of Americas and Africa, western and southern Europe, and across southern Asia to Micronesia and Australia (rare in New Zealand). Although readily taking wing, tends to stay in lowest airspace and not to indulge much in unnecessary flights. |
| Black-tailed Godwit | Icelandic population winter in large numbers in Ireland and Britain and in western France (Williamson and Ruttledge 1957; Vernon 1963; Fournier and D'Elbée 1974; Prater 1975a). Although primarily a ground and wading species (up to belly-depth), flies readily at all seasons, rising freely above lower airspace. |
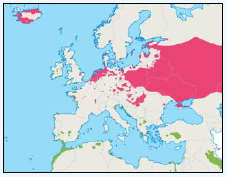
| Goosander | Migratory and partially migratory. No evidence yet that any Icelandic breeders emigrate. Similarly, British breeders almost entirely resident, moving short distances (mainly within 150 km) from breeding waters to lakes and sheltered estuaries |
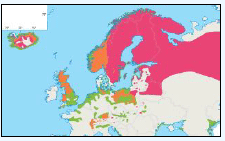
| Eurasian Teal | All but a few Icelandic breeders migrate to Britain (chiefly Scotland) and Ireland. Most or all breeders from north Russia, Baltic states, Fenno-Scandia, north Poland, north Germany, and Denmark fly south-west in autumn to North Sea wintering grounds, chiefly Netherlands and Britain. Winter distribution between Netherlands, England, Wales, and Ireland greatly dependent on weather. Breeders of Britain largely resident, moving only in severe weather. Makes full use of flight, mainly at medium and low heights, for reconnoitring secure places and for daily movements between feeding and resting areas. |
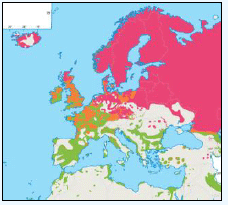
| Gadwall | Icelandic breeders winter Ireland and Britain. Breeders from Scotland move to Ireland (chiefly) and England. Those breeding north Germany, Poland, south Sweden, and west-central Russia winter North Sea, chiefly Netherlands and Britain, with some as far as Mediterranean. Mobile on ground as well as swimming and flying. Takes off less often in steep ascent from confined spaces than A. platyrhynchos or Teal A. crecca, but equally prone to make circling reconnaissance flights at low or medium heights, and frequent local movements between resting, foraging, and breeding haunts. |
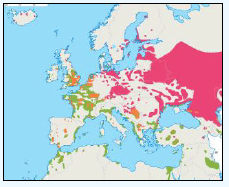
| Common Greenshank | Scottish population - passage occurs through Britain and Ireland Those to and from Fenno-Scandia and Denmark show movement through Poland, West Germany, eastern Britain, Low Countries, France, Italy, Iberia, and Morocco. Flight normally in lower airspace except during display and migration when upper airspace up to some hundreds of metres used. |
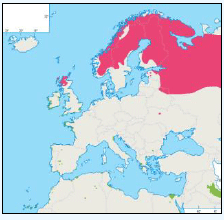
| Black-headed Gull | European winter range extends from southern Iceland, Faeroes, southern Norway, western Baltic, Balkans, and southern Russia southwards; common winter visitor in Iberia and around Mediterranean. Icelandic population recovered in Scotland and Ireland Fenno-Scandian and Baltic Populations - many migrate to western seaboard countries (North Sea and British Isles to north-west Africa) Norwegian birds winter mainly around North Sea, with those from western Norway especially in Britain. |
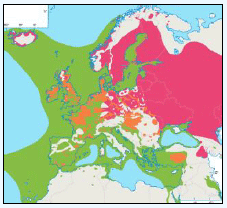
| Common Tern | Migratory Behaviour - Radar-tracked terns (both Arctic and Common) crossing south Sweden in late July took off in small flocks during evening, ascended at rates of c. 1.2 m/s to heights of 1000-3000 m flying 12-15 m/s (40-55 km/h) in level flight, suggesting that terns start migratory flights in evening and fly at high altitudes at night (Alerstam 1985). In north Germany, >50% of visible terns migrate <20m or >80% <100 m, and only c. 10% higher than 250 m, single birds >500m (3985 migrating terns observed). North and east European populations migrate westwards to Great Britain (Norman 2002). Main wintering areas of British and Irish birds are Ghana and Senegal. |
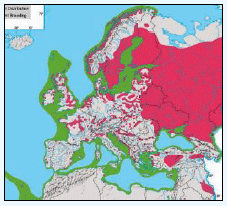
| Common Gull (Mew) | Many of the Common Gulls that winter in Britain are birds that nested in Scandinavia (Holden & Cleeves, 2002). |
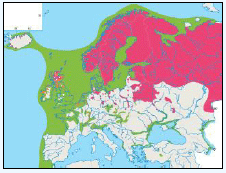
| Northern Shoveler | Mostly migratory. Breeders of Iceland all migrate, probably to Ireland or Britain. Most British breeders move southwards to south France, south Spain, north and central Italy, a few to north Africa. Breeders from south Fenno-Scandia and USSR east migrate west and south-west to western seaboard, chiefly Netherlands, Britain, and Ireland. Circles like other Anas and usually flies below 100 m; shares generic capacity for quick and ready movement between sites. |
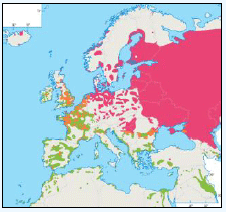
| Bewick’s (Tundra) Swan | Wintering sites in Denmark, northwest Germany (few), Netherlands, Britain, and Ireland Bewick’s swan leave Siberia and migrate to Britain, the Netherlands or Germany (Holden & Cleeves, 2002). |
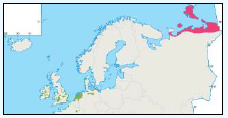
| Common Pochard | Mainly migratory, but some resident or partially migratory. Many of the birds that winter in Britain migrate here from northern and eastern Europe and central Russia (Holden & Cleeves, 2002). Freely uses excellent powers of flight, and will rise on occasion to considerable altitudes. |
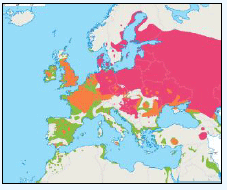
| Tufted Duck | Predominantly migratory though largely resident in a few areas. Some Icelandic breeders winter on coast and other ice-free waters there, but most move to Ireland (chiefly) and Britain. Breeders of northern Britain mainly move south-west to Ireland, though Scottish juveniles recovered exceptionally north to Norway and south to Portugal; those of south Britain believed largely resident. Vigorous flier making full use of surrounding airspace over fairly wide radius and to considerable heights. |
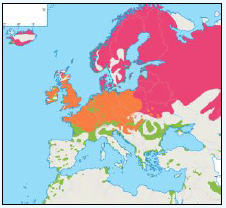
| Lesser Black-back Gull | Total winter range now extends from Britain, Mediterranean, Black and Caspian Seas (uncommon), Turkmeniya, and Persian Gulf to Arabian Sea and West and East Africa (sparingly further south). Flies readily and strongly, at low and medium altitudes, and less frequently in upper airspace. |
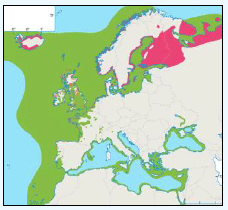
| Taiga Bean Goose | In west Europe, winters in Britain (few), Denmark, south Sweden, East and West Germany, Netherlands, Belgium, France and Spain. Birds from Scandinavia migrate to Britain (Holden & Cleeves, 2002). As forest breeder, can rise steeply from confined spaces, but frequency and patterns of aerial activity otherwise resemble other geese. |
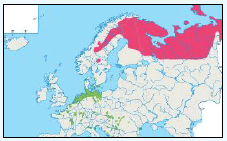
Map Legend
(Source: Cramp et al., 2000 and references therein)
G.5 References
ABPmer (2010). Screening and Scoping Review for the Pentland Firth Strategic Area (PFSA) Leasing Round Habitats Regulations Assessment. Appendix E: Informing Appropriate Assessment of the Pentland Firth Strategic Area Leasing Round. A report by BTO.
ABPmer (2011). Habitats Regulations Appraisal of Draft Plan for Offshore Wind Energy in Scottish Territorial Waters: Information for Appropriate Assessment Report for the Scottish Government January 2011; ABP Marine Environmental Research Ltd. Report No. R. 1722 (overall summary) and R1772a -c (pre-screening, screening and assessment information reports)
ABPmer (2014). Wave and Tidal Further Leasing: HRA Principles Document, Screening Report and Appropriate Assessment Information Report. Reports for The Crown Estate. ABP Marine Environmental Research Ltd. Report numbers R.2160a-c.
AEWA (2018). Draft AEWA/EU International Single Species Action Plan for the Conservation of the Velvet Scoter (Melanitta fucas)- Western Siberia and Northern Europe/NW Europe population.
Anker-Nilssen, T. and Lorentsen, S.-H. (1990). Distribution of Puffins Fratercula arctica feeding off Røst, northern Norway, during the breeding season, in relation to chick growth, prey and oceanographical parameters. Polar Res. 8:67-76.
APEM (2012). Investigation of the Utilisation of Sea Space by Seabirds in the Pentland Firth Orkney Area. Report to Scottish Government. 78pp
BirdLife International (2000). The Development of Boundary Selection Criteria for the Extension of Breeding Seabird Special Protection Areas into the Marine Environment. OSPAR Convention for the Protection of the Marine Environment of the North-East Atlantic. Meeting of the Biodiversity Committee (BDC) Vlissingen (Flushing): 20 - 24 November 2000.
BirdLife International (2010). BirdLife Seabird Wikispace. Available online: http://seabird.wikispaces.com/ Accessed: 19/11/10.
Bogdanova, M. I., Butler, A., Wanless, S., Moe, B., Anker-Nilssen, T., Frederiksen, M and Harris, M. P. (2017). Multi-colony tracking reveals spatio-temporal variation in carry-over effects between breeding success and winter movements in a pelagic seabird. Marine Ecology Progress Series, 578, 167-181.
Bradstreet, M.S.W. and Brown, R.G.B. (1985) Feeding ecology of the Atlantic Alcidae. Pp. 263-318 in The Atlantic Alcidae: the evolution, distribution and biology of the auks inhabiting the Atlantic Ocean and adjacent water areas (D. N. Nettleship and T. R. Birkhead, eds.). Academic Press, London, U.K.
Briggs, K.T., Tyler, W.B., Lewis, D.B. and Carlson, D.R. (1987) Bird communities at sea off California: 1975-1983. Stud. Avian Biol. 11:1-74.
Brooke, M. (1990) The Manx Shearwater. T & AD Poyser, London.
Burger, A. E. & Shaffer, S. A. 2008. Application of tracking and data-logging technology in research and conservation of seabirds. The Auk 125: 253-264.
Cairns D.K., Bredin K.A. and Montevecchi W.A. (1987). Activity budgets and foraging ranges of breeding Common Murres. The Auk 102: 218-224
Cleasby IR, Owen E, Wilson LJ, Bolton M (2018). Combining habitat modelling and hotspot analysis to reveal the location of high-density seabird areas across the UK: Technical Report. RSPB Research Report no. 63. RSPB Centre for Conservation Science, RSPB, The Lodge, Sandy, Bedfordshire, SG19 2DL. Available from https://www.rspb.org.uk/globalassets/downloads/documents/conservation-science/cleasby_owen_wilson_bolton_2018.pdf
Cooch, F.G. (1965) The breeding biology and management of the Northern Eider (Somateria mollissima borealis) in the Cape Dorset Area, North-west Territories. Canadian Wildlife Service Wildlife Management Bulletin. Series 2, Number 10.
Cook, A.S.C.P., Johnston, A., Wright, L.J., & Burton, N.H.K. 2012. A review of flight heights and avoidance rates of birds in relation to offshore wind farms. Strategic Ornithological Support Services: Project SOSS-02. BTO and The Crown Estate.
Cook, A.S.C.P., Ward, R,M., Hansen, W.A and Larsen, L. 2018. Estimating Seabird Flight Height using LiDAR. Scottish Marine and Freshwater Science Vol 9 No 14
Cramp S (ed.) (2000) Birds Of The Western Palearctic Edited by Stanley Cramp 1832 pages, col plates, illus, figs, maps. [2000] BWPi DVD Oxford University Press
Daunt, F., Benvenuti, S., Harris, M.P., Dall’Antonia, L., Elston, D.A. and Wanless, S. (2002) Foraging strategies of the Black-legged Kittiwake Rissa tridactyla at a North Sea colony: evidence for a maximum foraging range. Mar. Ecol. Progr. Ser. 245:239-247.
DECC, 2009. UK Offshore Energy Strategic Environmental Assessment. Future Leasing for Offshore Wind Farms and Licensing for Offshore Oil & Gas and Gas Storage. Environmental Report.
Defra (2011) Charting Progress 2 An Assessment of the State of UK Seas Author: Department for Environment Food and Rural Affairs (Defra) Publisher: TSO (The Stationery Office) January 2011.
Desholm, M., Fox, A.D. and Beasley, P.D. 2004. Best practice guidance for the use of remote techniques for observing bird behaviour in relation to offshore wind farms. COWRIE - REMOTE-05-2004, 94 pp.
Drewitt AL & Langston RHW (2006). Assessing the impacts of wind farms on birds. Ibis 148, 29-42.
Dunnet, GM, Ollason, JC, & Anderson, A 1979 A 28-year study of breeding fulmars Fulmarus glacialis in Orkney. Ibis, 121: 293-300.
Dunnet, G.M. and Ollason, J.C. (1982) The feeding dispersal of Fulmars Fulmarus glacialis in the breeding season. Ibis 124:359-361.
Durinck, J., Skov, H., Jensen, F.P. and Pihl, S. (1994) Important Marine Areas for Wintering Birds in the Baltic Sea. Ornis Consult report to the European Commission. 110 pp.
Falk, K. and Møller, S. (1995) Satellite tracking of high-arctic Northern Fulmars. Polar Biol. 15:495-502.
Fenny, M. and Walls, R. 2009. UK Offshore Energy Strategic Environmental Assessment (SEA) Environmental Report Briefing Note. Prepared for SeaEnergy Renewables.
Fox, A.D. (1981) Red-throated Diver Gavia stellata pp.158-160 in: Fox, A.D. and Stroud, D.A. (eds) Report of the 1979 Expedition to Eqalungmiut Nunaat, west Greenland. Greenland White-fronted Goose Study, Aberystwyth
Furness, R.W. (1978) Kleptoparasitism by Great Skuas (Catharacta Skua Brunn.) and Arctic Skuas (Stercorarius parasiticus L.) at a Shetland seabird colony. Animal Behaviour 26:1167-1177.
Furness, R.W., 1997. A 1995 survey of the Rum Manx shearwater population. Scottish Natural Heritage Research, Survey and Monitoring Report, No. 73.
Furness, R.W. & Tasker, M.L. 2000. Seabird-fishery interactions: quantifying the sensitivity of seabirds to reductions in sandeel abundance, and identification of key areas for sensitive seabirds in the North Sea. Marine Ecology Progress Series 202: 253-264.
Furness, R.W., Wade, H.M., Robbins, A.M.C., and Masden, E.A. Assessing the sensitivity of seabird populations to adverse effects from tidal stream turbines and wave energy devices. ICES Journal of Marine Science. 69(8), 1466-1479.
Furness, R.W., Wade, H.M., & Masden E.A. 2013. Assessing vulnerability of marine bird populations to offshore wind farms. Journal of Environmental Management. 119; 56-66
Furness, R.W. 2015. Non-breeding season populations of seabirds in UK waters: Population sizes for Biologically Defined Minimum Population Scales (BDMPS). Natural England Commissioned Reports, Number 164
Gaston, A.J., & Jones, I. (1998). The auks. Oxford University Press, Oxford.
Glew, K. S. J., Wanless, S., Harris, M. P., Daunt, F., Erikstad, K. E., Strøm, H., & Trueman, C. N. (2018. Moult location and diet of auks in the North Sea inferred from coupled light-based and isotope-based geolocation. Marine Ecology Progress Series, 599, 239-251.
Goudie, R.I. and Ankney, C.D. (1988) Patterns of habitat use by sea ducks wintering in southeastern Newfoundland. Ornis Scandinavica 19:249-256.
Goudie, R.I., Robertson, G.J. and Reed, A. (2000) Common Eider (Somateria mollissima), The Birds of North America Online (A. Poole, Ed.). Ithaca: Cornell Lab of Ornithology; Retrieved from the Birds of North America Online: http://bna.birds.cornell.edu/bna/species/546 doi:10.2173/bna.546
Grecian W.J., Inger, R., Attrill, M.J., Bearhop, S., Godley, B.J., Witt, M.J., Votier, S.C. 2010. Potential Impacts of wave-powered marine renewable energy installations on marine birds. IBIS. 152, 683-697
Grémillet, D., Pichegru, L., Siorat, F., and Georges, J. (2006). Conservation implications of the apparent mismatch between population dynamics and foraging effort in French Northern Gannets from the English Channel. Mar Ecol Prog Ser 319: 15-25.
Griffin, L. 2010. Migration Routes of Birds in Relation to Current and Proposed UK Wind Farm Sites [online] Available at: http://www.microwavetelemetry.com/newsletters/spring09page5.pdf, accessed on 27/01/2020.
Griffin, L., Rees, E. & Hughes, B. 2010. The Migration of Whooper Swans in Relation to Offshore Wind Farms. WWT Final Report to COWRIE Ltd, WWT, Slimbridge. 69 pp.
Guilford, T.C., Meade, J., Freeman, R., Birdo, D., Evans, T., Bonadonna, F., Boyle, D., Roberts, S. and Perrins, C.M. (2008). GPS Tracking of the foraging movements of Manx Shearwaters Puffinus puffinus breeding on Skomer Island, Wales. Ibis 150(3):462-473.
Hamer, K.C., Monaghan, P., Uttley, J.D., Walton, P. and Burns, M.D. (1993) The influence of food supply on the breeding ecology of Kittiwakes Rissa tridactyla in Shetland. Ibis 135:255-263.
Hamer, K.C., Phillips, R.A., Wanless S., Harris M.P. and Wood, A.G. (2000). Foraging ranges, diets and feeding locations of Gannets Morus bassanus in the North Sea: evidence from radio tracking. Mar Ecol Prog Ser 200:257-264.
Hamer, K.C., Phillips, R.A., Hill, J.K., Wanless, S. and Wood, A.G. (2001). Contrasting foraging strategies of gannets Morus bassanus at two North Atlantic colonies: foraging trip duration and foraging area fidelity. Mar Ecol Prog Ser 224:283-290.
Hamer, K.C., Lewis, S., Wanless, S., Phillips, R.A., Sherratt, T.N., Humphreys, E.M., Hennicke, J., and Garthe, S. (2006). Use of gannets to monitor prey availability in the northeast Atlantic ocean: colony size, diet and foraging behaviour. Pp. 236-248 in: Boyd, I.L., Wanless, S. and Camphuysen, C.J. (Eds). Top Predators in Marine Systems. Cambridge University Press.
Hamer, K. C., Humphreys, E. M., Garthe, S., Hennicke, J., Peters, G., Grémillet, D., Phillips, R. A., Harris, M. P., & Wanless, S. (2007). Annual variation in diets, feeding locations and foraging behaviour of gannets in the North Sea: flexibility, consistency and constraint. Mar Ecol Prog Ser 338: 295-305.
Hamer, K.C., Humphreys, E.M., Magalhães, M.C., Garthe, S., Hennicke, J., Peters, G., Grémillet, D., Skov, H, and Wanless, S. (2009). Fine-scale foraging behaviour of a medium-ranging marine predator. Journal of Animal Ecology. doi: 10.1111/j.1365-2656.2009.01549.x
Harrison, N, Webb, A, Leaper, GM, & Steele, RD 1989 Seabird distributions and the movements of moulting auks west of Scotland and in the northern Irish Sea (late summer 1988). Nature Conservancy Council, CSD Report, No. 994.
Holden, P. and Cleeves, T., 2002. RSPB Handbook of British Birds. Publishers Ltd., London.
Humphreys, E.M., Wanless, S. and Bryant, D.M. (2006) Stage-dependent foraging in breeding Black-legged Kittiwakes Rissa tridactyla: distinguishing behavioural responses to intrinsic and extrinsic factors. J. Avian Biol. 37:436-446.
Kaiser, M.J. (2004) Predicting the displacement of common scoter Melanitta nigra from benthic feeding areas due to offshore wind farms. Centre for Applied Marine Sciences, School of Ocean Sciences, University of Wales, Bangor. Interim Report 2004 No 4.
Kaiser, M.J., Galanidi, M., Showler, D.A., Elliott, A.J., Caldow, R.W.G., Rees, E.I.S., Stillman, R.A. and Sutherland, W.J. (2006) Distribution and behaviour of Common Scoter Melanitta nigra relative to prey resources and environmental parameters. Ibis 148:110-128.
Köller J, Köppel J & Wolfgang P Eds (2006). Offshore wind energy research on environmental
impacts. Springer publications.
Kumari, E. (1979) Moult and moult migration of waterfowl in Estonia. Wildfowl 30:90-98.
Langston, R. 2010. Offshore wind farms and birds: Round 3 zones, extensions to Round 1 & Round 2 sites & Scottish Territorial Waters. RSPB Research Report No. 39
Langston, R. H., Teuten, E., & Butler, A. (2013). Foraging ranges of northern gannets Morus bassanus in relation to proposed offshore wind farms in the UK: 2010-2012. RSPB report to DECC.
Leaper, G.M., Webb, A., Benn, S., Prendergast, H.D.V., Tasker, M.L., and Schofield, R. (1988) Seabird studies around St Kilda, June 1987. Nature Conservancy Council, CSD Report No. 804.
Lewis, S., Sherratt, T.N., Hamer, K.C. and Wanless, S. (2001) Evidence of intraspecific competition for food in a pelagic seabird. Nature 412:816-819.
MacDonald, MA. 1977. An analysis of the recoveries of British-ringed fulmars. Bird Study, 24: 208-214.
Marine Scotland, 2010. Strategic Environmental Assessment (SEA) of Draft Plan for Offshore Wind Energy in Scottish Territorial Waters: Volume 1: Environmental Report
Marine Scotland, 2012. Vulnerability of Scottish Seabirds to Offshore Wind turbines. Prepared by Furness, R.W., and Wade, H.
Merkel, F.R. and Mosbech, A. (2008). Diurnal and nocturnal feeding strategies in Common Eiders. Waterbirds 31(4):580-586.
Merkel, F.R., Mosbech, A., Sonne, C., Flagstad, A., Falk, K. and Jamieson S.E. (2006) Local movements, home ranges and body condition of Common Eiders Somateria mollissima wintering in Southwest Greenland. Ardea 94:639-650.
Mitchell, P.I., Newton, S.F., Ratcliffe, N. & Dunn, T.E. (Eds). (2004) Seabird Populations of Britain and Ireland: results of the Seabird 2000 census (1998-2002). Published by T and A.D. Poyser, London.
Mudge, G.P. and Crooke, C.H. (1986) Seasonal changes in the numbers and distribution of seabirds in the Moray Firth, northeast Scotland. Proceedings of the Royal Society of Edinburgh B 91:81-104.
Nelson, J.B. (1978) The gannet. T & AD Poyser, Berkhamsted, Herts.
Nettleship, D.N. and Gaston, A.J. (1978) Patterns of pelagic distribution of seabirds in western Lancaster Sound and Barrow Strait, Northwest Territories, in August and September 1976. Canadian Wildlife Service Occasional Papers No. 39.
Ostrand, W.D., Drew, G.S., Suryan, R.M. and McDonald, L.L. (1998) Evaluation of radio-tracking and strip transect methods for determining foraging ranges of Black-legged Kittiwakes. Condor 100:709-718.
Owen, E. (2012) Connectivity between seabirds SPAs and windfarms: Overview for the FAME project. British Ornithologist’s Union Marine Renewables and Birds Conference 26 November 2012.
Pearson, T.H. (1968) The feeding ecology of sea-bird species breeding on the Farne Islands, Northumberland. Journal of Animal Ecology 37:521-552.
Perrow M. R. Skeate E. R., Lines P., Brown D. and Tomlinson M. L. 2006. Radio telemetry as a tool for assessing impacts of windfarms: the case of Little Terns Sterna albifrons at Scroby Sands, Norfolk, UK. Ibis 148:57-75.
Petersen I.K., Fox A.D. and Clausager I. (2003) Distribution and numbers of birds in Kattegat in relation to the proposed offshore wind farm south of Læsø -Ornithological impact assessment. Department of Wildlife Ecology and Biodiversity. National Environmental Research Institute, Ministry of Environment, Denmark.
Phillips, R.A. (1995) Population ecology of Arctic Skuas Stercorarius parasiticus on Foula. PhD thesis, University of Glasgow.
Pollock, C.M, Reid, J.B, Webb, A, and Tasker, M.L. 1997. The distribution of seabirds and cetaceans in the waters around Ireland. JNCC Report, No. 267.
Pollock, C.M., Mavor, R., Weir, C.R., Reid, A., White, R.W., Tasker, M.L., Webb and Reid, J.B. 2000. The distribution of seabirds and marine mammals in the Atlantic Frontier, north and west of Scotland. JNCC Report No 344.
Quinn, L. .2014 Intra-and inter-colony difference in non-breeding strategies in the northern fulmar, Fulmarus glacialis. University of Aberdeen.
Ratcliffe, N., Phillips, R. A., & Gubbay, S. 2000. Foraging ranges of UK seabirds from their breeding colonies and its implication for creating marine extensions to colony SPAs. Unpublished Report to BirdLife International, RSPB, Sandy.
Roos S, Humphreys L., Wernham C and Burton N. (2009) Informing Appropriate Assessment Of The Pentland Firth Strategic Area Leasing Round Ornithology Scoping Report. BTO report presented within the Pentland Firth Strategic Area Appropriate Assessment Information Report for The Crown Estate by ABPmer 2010
Soanes, L.M., Atkinson, P.W., Gauvain, R.D. and Green, J.A., (2013). Individual consistency in the foraging behaviour of Northern Gannets: Implications for interactions with offshore renewable energy developments. Marine Policy, 38, pp.507-514.
Scottish Executive, 2007. Scottish Marine Renewables: Strategic Environmental Assessment (SEA). Report prepared for the Scottish Executive by Faber Maunsell and Metoc PLC https://www2.gov.scot/Publications/2007/03/seawave
Smith, C. and Jones, T. (2005). Breeding ecology and diet of Great and Arctic Skuas on Handa Island 2005. Unpublished report to Scottish Wildlife Trust, Scottish Natural Heritage, The Seabird Group, Joint Nature Conservation Committee.
Stallcup, R. (1990) Ocean birds of the nearshore Pacific. Point Reyes Bird Observatory, Stinson Beach, CA.
Stone, C.J, Webb, A, & Tasker, M.L .1995. The distribution of auks and Procellariiformes in north-west European waters in relation to depth of sea. Bird Study, 42: 50-56.
Suryan, R.M., Irons, D.B. and Benson, J. (2000) Prey switching and variable foraging strategies of Black-legged Kittiwakes and the effect on reproductive success. The Condor 102:374-384.
Tasker, M.L., Jones, P.H., Blake, B.F. and Dixon, T.J. (1985a) The marine distribution of the gannet Sula bassana in the North Sea. Bird Study 32:82-90.
Tasker, M.L, Jones, P.H, Blake, B.F, and Dixon, T.J. 1985b. Distribution and feeding habits of the great skua Catharacta skua in the North Sea. Seabird, 8: 34-43.
Tasker, M.L, Webb,A, Murray, S, and Holt R. 1986. The dispersal of auks in the northern North Sea. Nature Conservancy Council, CSD Report, No. 627.
Tasker, M.L., Webb, A., Hall, A.J., Pienkowski, M.W. and Langslow, D.R. (1987) Seabirds in the North Sea. NCC, Peterborough.
Thaxter C.B., Daunt F., Hamer K.C., Watanuki Y., Harris M.P., Grémillet D., Peters G., Wanless S. (2009). Sex-specific food provisioning in a monomorphic seabird, the Common Guillemot Uria aalge: nest defence, foraging efficiency or parental effort? Journal of Avian biology 40:75-84
Thaxter, C.B., Wanless, S., Daunt, F., Harris, M.P., Benvenuti, S., Watanuki, Y., Grémillet, D., Hamer, K.C. (2010). Influence of wing loading on trade off between pursuit-diving and flight in Common Guillemots and razorbills. J.Exp. Biol. 213: 1018-1025
Thaxter, C.B., Lascelles, B., Sugar, K., Cook, A.S.C.P., Roos, S., Bolton, M., Langston, R.H.W., and Burton, N.H.K. 2012. Seabird foraging Ranges as a Preliminary Tool for Identifying Candidate Marine Protected Areas. Biological Conservation. doi: 10.1016/j.biocon.2011.12.009
Trevail, A.M., Green, J.A., Sharples, J., Polton, J.A., Arnould, J.P. and Patrick, S.C., 2019. Environmental heterogeneity amplifies behavioural response to a temporal cycle. Oikos, 128(4), pp.517-528.
Votier S.C, Bearhop S., Ratcliffe, N., Furness, R.W. (2004) Reproductive consequences for Great Skuas specializing as seabird predators. Condor 106:275-287.
Votier, S.C., Crane, J.E., Bearhop, S., de Leo, A., McSorley, C.A., Minguez, E., Mitchell, I.P., Parsons, M., Phillips, R.A., and Furness, R.W. (2006). Nocturnal foraging by Great Skuas Stercorarius Skua: implications for conservation of storm-petrel populations. J Ornithol. 147:405-413.
Wade, H. M., Masden, E. A., Jackson, A. C., Thaxter, C. B., Burton, N. H. K., Bouten, W., & Furness, R. W. (2014). Great skua (Stercorarius skua) movements at sea in relation to marine renewable energy developments. Marine environmental research, 101, 69-80.
Wakefield, E. D., Bodey, T. W., Bearhop, S., Blackburn, J., Colhoun, K., Davies, R& Jessopp, M. J. (2013). Space partitioning without territoriality in gannets. Science, 341(6141), 68-70.
Wakefield, E.D., Owen, E., Baer, J., Carroll, M.J., Daunt, F., Dodd, S.G., Green, J.A., Guilford, T., Mavor, R.A., Miller, P.I. and Newell, M.A., 2017. Breeding density, fine‐scale tracking, and large‐scale modeling reveal the regional distribution of four seabird species. Ecological Applications, 27(7), pp.2074-2091.
Wanless, S., Harris, M.P. and Greenstreet, S.R. (1998) Summer sandeel consumption by seabirds breeding in the Firth of Forth, southeast Scotland. ICES J. Marine Science 55:1141-1151.
Wanless, S., Harris, M.P. and Morris, J.A. (1990) A comparison of feeding areas used by individual common murres (Uria aalge), razorbills (Alca torda) and an Atlantic puffin (Fratercula arctica) during the breeding season. Colonial Waterbirds 13:16-24.
Wanless, S., Harris M.P., and Morris J.A. (1991). Foraging range and feeding locations of shags Phalacrocorax aristotelis during chick rearing. Ibis 133:30-36.
Wanless, S., Harris, M.P., Greenstreet, S.P.R. and Rindorf, A. (2000) Chapter 5: Avian Predators. Pp 5.1-5.93 In: Harwood, J. (Ed.) Final report to EU, ‘The effect of large-scale industrial fisheries on non-target species’.
Wanless, S., Daunt, F., Camphuysen, C.J., Humphreys, E., Scott, B. and Wanless, S. (2005) Setting the scene: seabird foraging behaviour, diet and breeding success. Pp 88-134 In: Camphuysen, C.J. (Ed.) Final report to EU, ‘Interactions between the Marine environment, Predators, and prey: implications for Sustainable Sandeel fisheries (IMPRESS)’
Webb, A., Tasker, M.L. and Greenstreet, S.P.R. (1985) The distribution of guillemots (Uria aalge), razorbills (Alca torda) and Puffins (Fratercula arctica) at sea around Flamborough Head, June 1984. Nature Conservancy Council, CSD Report No. 590.
Wernham, C. V., Toms, M. P., Marchant, J. H., Clark, J. A., Siriwardena, G. M., & Baillie, S. R. (eds). 2002. The Migration Atlas: movements of the birds of Britain and Ireland. T & A D Poyser, London.
Wildfowl and Wetland Trust (WWT), 2010. Svalbard Barnacle Goose Tracking Map 2009 [online] Available at: hhttps://monitoring.wwt.org.uk/our-work/goose-swan-monitoring-programme/species-accounts/svalbard-barnacle-goose/, accessed on 27/01/2020.
Wilson, L. J., McSorley, C. A., Gray, C. M., Dean, B. J., Dunn, T. E., Webb, A., & Reid, J. B. 2009. Radiotelemetry as a tool to define protected areas for seabirds in the marine environment. Biol. Cons. 142: 1808-1817.
Wood, K.A. (1989). Seasonal abundance, marine habitats and behaviour of Skuas off central New South Wales. Corella 13:97-104.
Wright, P.J. and Bailey, M.C. (1993) Biology of sandeels in the vicinity of seabird colonies at Shetland. SOAFD, Marine Laboratory, Aberdeen. Report No. 15/93.
Zalakevicius, M. and Jacoby, V. (1992) Radar and visual observations of the moult migration of Common Scoter within eastern Baltic. IWRB Seaduck Bull. 1:51.
Contact
Email: drew.milne@gov.scot