Coronavirus (COVID-19) vaccine certification: evidence paper update
This paper summarises the range of evidence available on vaccination certification schemes. Evidence is drawn from clinical and scientific literature, from public opinion and from international experience.
7. Extending the vaccine certification scheme
The current Covid-19 vaccination certification scheme in Scotland aims to[113] [114] [115] [116]
- Reduce the risk of transmission (harm 1);
- Reduce the risk of serious illness and death and in doing so alleviate current and future pressure on the healthcare system (harm 1);
- Allow higher risk settings to continue to operate as an alternative to closure or more restrictive measures (harms 3 & 4); and
- Increase vaccine uptake (harm 1)
Looking at the approaches adopted in other countries and advice from SAGE[117], the potential approaches to extending the current Scottish scheme would be to:
- Extend the range of settings
- Include testing, either as an option or alongside vaccination
- Include recovery instead of testing or vaccination
- Timestamp for boosters to allow for waning.
There are no real life studies directly comparing the effect on transmission for certification schemes based on testing only, vaccination (or previous infection) only, or both. As outlined above in the international section of the paper different countries have adopted different approaches and have changed their approach over time.
7.1 Extending the range of settings
EMG/SPI-M/SPI-B have noted that certification based on vaccination status or prior infection would indirectly reduce the likelihood of an infected person being present as they would demonstrate some level of immunity[118]. They note that certification based on negative test results could reduce the likelihood of an infected person being present. However this would depend on the quality of the test and when the test was taken in relation to the event. They note that the practical and ethical issues need to be considered[119].
They state that increasing the range of applicable settings, time-limiting certificates based on last vaccination date, and including a requirement for proof of a negative test, could all be considered to increase the potential impact on transmission and vaccine uptake [120].
Extending the range of settings would increase the possibility of reducing transmission and would potentially encourage a wider range of people to take up vaccination as was noted above.[121]
7.2 Testing
Successfully including testing as an alternative depends on the accuracy of the tests and how they will be used by the public.
Two main testing methods exist for detection of SARS-CoV-2; LFT (lateral flow tests or devices) or RT-PCR. RT_PCR is the recommended testing method if you have COVID-19 symptoms while LFT is recommended only for people who do not have symptoms[122]. RT-PCR is a highly sensitive and specific technique to detect SARS-CoV-2 and is a recommended diagnostic testing method by the WHO[123]. Specificity and sensitivity levels of >95% have been reported by SAGE for RT-PCR testing[124].
LFD testing is effective at identifying people with the virus when they are at their most infectious and have high viral loads[125]. A peer-reviewed study on sensitivity of the LFDs carried out by the University College London found that LFDs are more than 80% effective at detecting any level of COVID-19 infection therefore can be an effective tool in reducing transmission[126]. Another study showed that LFDs are 95% effective and 89.1% specific at detecting COVID-19 when used at the onset of symptoms[127].
SAGE endorsed the benefits that rapid antigen testing could have on reducing transmission when discussing the UK Government Plan B options; 'Other measures are available which, if introduced, could also make Plan B (or more stringent measures) less likely (and could potentially offer better efficiency or effectiveness) for example encouraging wider use of rapid antigen testing in workplaces and the community, and ensuring self-isolation of those who test positive by providing sufficient support'[128].
Clinical evaluations of LFTs by Public Health England (PHE) and University of Oxford reported a slight reduction in the LFT test sensitivity with nasal-only swabbing compared with the throat and nose swab test (88% vs 92% respectively). The sensitivity of the nasal-only swabbing was unaffected by the experience level of the operator[129]. Therefore LFT testing can be an effective measure in reducing transmission.
The optimal testing strategy in order to gain access to a high risk setting would be to take the test as close as practically possible before entry into the setting. This will have the best chance of detecting infectious individuals. RT-PCR samples need to be sent to a laboratory for processing and testing using expensive equipment, therefore the reporting of a result can take between 24 to 72 hours. In contrast a LFT is a rapid test, producing a result within 30 minutes. There are trade-offs between the lower sensitivity of lateral flow tests and the greater likelihood that more people will use them as they can be carried out at home quickly. Whether these trade-offs lead to a net beneficial or detrimental effect is unclear[130].
Outside of testing for certification purposes the guidance for children and teachers is to currently test twice a week. Around 1 in 3 people with coronavirus do not show symptoms. Regular lateral flow testing is encouraged to find infectious, asymptomatic positive cases[131]. Evidence suggests that serial antigen testing every three days or twice a week will almost always identify SARS-CoV-2 during early stages of infection, and therefore significantly reduce transmission[132]. A longitudinal assessment of diagnostic test performance of RT-qPCR and LFT assays throughout an acute SARS-CoV-2 infection was studied. Recently infected adults (n=43) with mild or asymptomatic SARS-CoV-2 infections were tested daily using nasal and saliva samples. Both RT-qPCR and LFT peaked in sensitivity when live virus was detected in nasal swabs, but sensitivity of RT-qPCR tests was greater than LFT at detecting virus prior to the infectious period. All tests showed >98% sensitivity if used at least every 3 days and daily screening using antigen tests can achieve approximately 90% sensitivity for identifying infected individuals while they are viral culture positive[133]. It would therefore be advantageous to routinely self-test multiple times per week to maximise early detection of virus and reduce transmission.
There is some evidence that COVID certification based on test results as well as vaccination status can be open to manipulation. The Netherlands implemented Covid-19 certification for nightclub entry on 26 June, 2021. Despite attempts to prevent unauthorised access unvaccinated and untested individuals were still able to gain entry due to end-user errors and oversight[134]. Subsequent rises in cases led to the closure of nightclubs again[135]. However, the government is currently developing support measures for nightclubs and discos to try to reopen[136].
Media reports increasingly note the problem of fake certificates[137] [138] [139]. Some countries have implemented fines and prosecution for individuals found using counterfeit certificates and for businesses found not to be checking certificates[140] [141].
7.3 Vaccine effectiveness
An expert consensus view of vaccines effectiveness against symptomatic disease in fully vaccinated people is between 45-95%, depending on the vaccine and when it was given[142] [143]. Evidence is not available on which type of certification scheme (testing or vaccination) would be most effective at reducing the likelihood of infected people being present at an event.
Vaccination reduces the chance of getting infected and uninfected people cannot transmit the virus, therefore, the vaccines are also effective at reducing transmission[144]. However, in breakthrough cases, where a vaccinated person becomes infected they may still pass the virus on. There is some evidence that at the peak of infection vaccinated and unvaccinated people have similar viral loads and a vaccinated person who is infected may be as likely to transmit virus to others as an unvaccinated person (High confidence)[145] . A UK cohort study found that the secondary attack rates (SAR) in household contacts exposed to the Delta variant was 25% (95% CI 18–33) for fully vaccinated individuals compared with 38% (24–53) in unvaccinated individuals. This indicates that fully vaccinated people with breakthrough infections can efficiently transmit infection in household settings, including to fully vaccinated contacts. However infected vaccinated people were slightly less likely to pass the virus on than infected unvaccinated people[146]. Analysis from the ONS Community Infection Survey concluded that vaccination reduces transmission of Delta, but by less than the Alpha variant and the impact of vaccination decreased over time[147].
7.4 Vaccine waning
The effectiveness of vaccination at reducing infections, symptomatic disease, severe disease, hospitalisation and death reduces with time after the second dose of the vaccine. Waning occurs from around 10 weeks post second dose and is most evident in older groups for symptomatic disease. Waning protection of the vaccine against hospitalisation and death is most evident in clinical risk groups [148].
Waning has a significant impact on the efficacy of certification as a useful measure to reduce the risk of transmission, symptomatic disease and hospitalisation.
Two real world studies from Israel found an increased rate of infection and severe disease for early vaccinated adults compared to those who were vaccinated later in the year. The first study analysed data from adults (n= 4,791,398 people) and found that among those fully vaccinated, rate ratios of infection two months after receiving the second dose of vaccine compared to the months when first fully vaccinated were: 1.6 for ≥60 years old, 1.7 for 40-59 years old's and 1.6 for 16-39 years old's. The rate ratios for severe disease were: 1.8 for ≥ 60, 2.2 for 40-59, insufficient data for 16-39[149]. The second study compared breakthrough infections in people ≥ 16 years old (n= 1,352,44 individuals) who received the second dose of the vaccine between January and April 2021. Researchers reported a significant increase in risk of infection among individual that were early vaccinated compared to those who were vaccinated later (1.51-fold, 95% CI, 1.38-1.66). The increase was similar across all age groups[150]. Results from both studies indicate that immunity against the Delta variant of SARS-CoV-2 waned in all age groups a few months after the receipt of the second dose of vaccine.
A recent, pre-published UK study looking at antibodies targeting the spike protein (anti-S antibodies) and breakthrough infections reported that those with anti-S levels of < 500 units (U)/ml following the second dose were nearly twice as likely to have a breakthrough infection compared to those with higher levels. Researchers estimated that antibody levels decline to this threshold after about 3 months in individuals that received a second dose of the Oxford-AstraZeneca vaccine and after about 7 months for people that received a second dose of the Pfizer/BioNTech vaccine, making people who received two doses of Oxford-AstraZeneca more at risk of a breakthrough infection compared to those doubly vaccinated with Pfizer/BioNTech (OR: 1.43, 95% Cis: 1.18-1.73, p<0.001)[151].
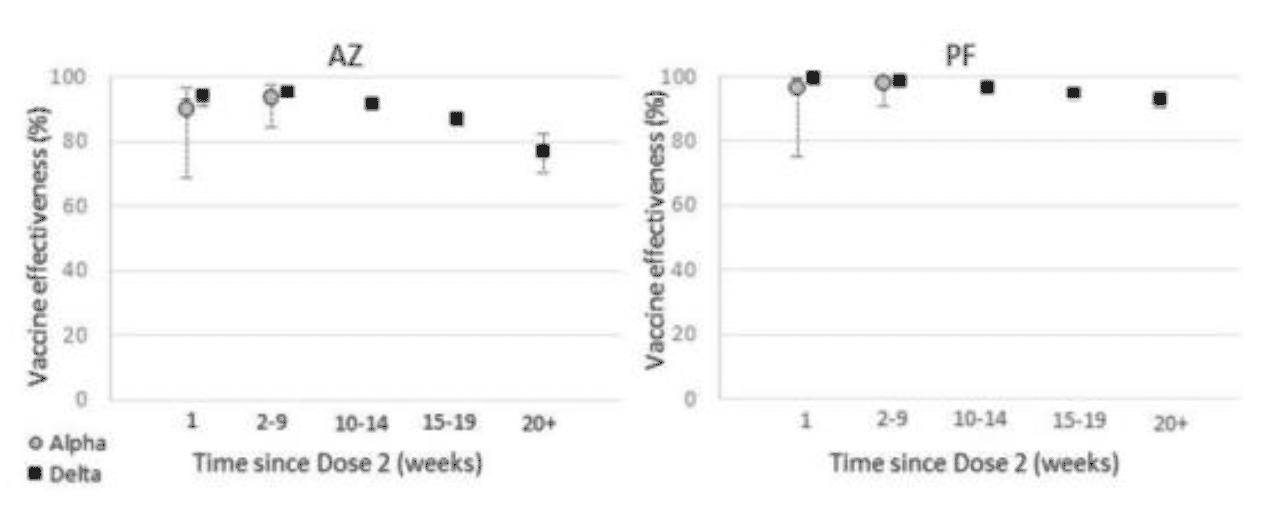
The latest Covid-19 vaccine surveillance report (week 45) published by the UK Health Security Agency (UKHSA) states that "There is some evidence of waning of protection against infection and symptomatic disease over time, though protection against severe disease remains high in most groups at least 5 months after the second dose" see figures 11 and 12[152].
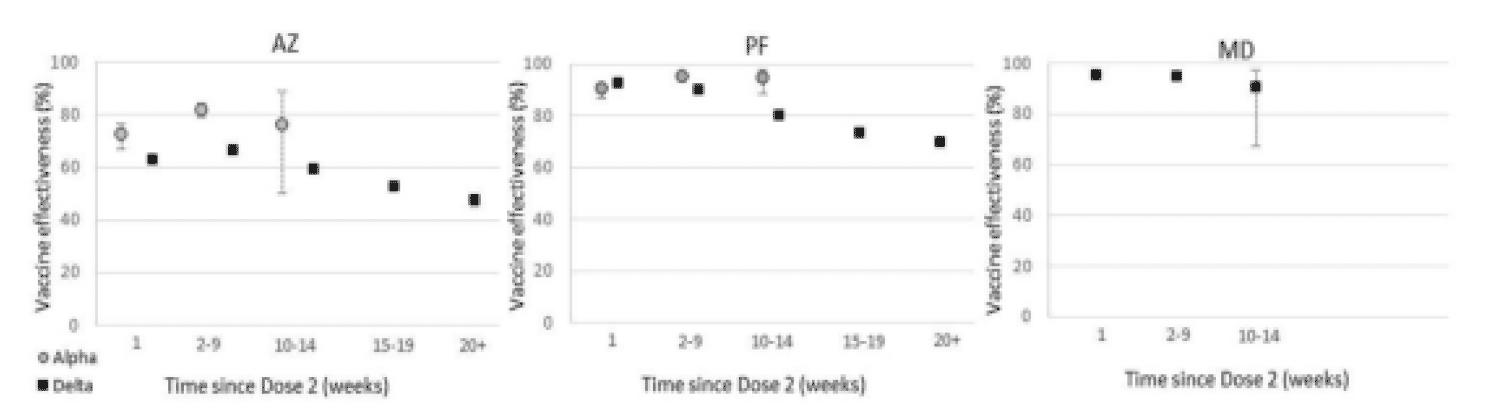
A study conducted on the resident population in Qatar looked at waning of Pfizer/BioNTech vaccine protection against infection (n= 907,763, people with two doses of vaccine). The study reported that the protective effect reached a peak in the first month after the second dose (77.5%), but then effectiveness started declining gradually and it accelerated after four months reaching approximately 20% between month 5-7 after the second dose. Effectiveness against severe, critical or fatal disease reached 96% or higher in the first two months after the second dose and it persisted at approximately this level for 6 months[153].
A pre-print paper detailing a UK real world data study demonstrated that vaccine effectiveness against symptomatic disease peaked in the early weeks after the second dose and then fell to 47.3 (95% CI 45 to 49.6) and 69.7 (95% CI 68.7 to 70.5) by 20+ weeks against the Delta variant for Oxford-AstraZeneca and Pfizer/BioNTech respectively. Waning of vaccine effectiveness was greater in older adults (65+ years old) compared to people 40-64 years old and amongst clinically extremely vulnerable groups. A smaller reduction in vaccine effectiveness against hospitalisation was reported, decreasing to 77.0 (70.3 to 82.3) and 92.7 (90.3 to 94.6) beyond 20 weeks post-vaccination for Oxford-AstraZeneca and Pfizer/BioNTech respectively. Vaccine effectiveness against death was 78.7 (95% CI 52.7 to 90.4) for Oxford-AstraZeneca and 90.4 (95% CI 85.1 to 93.8) for Pfizer/BioNTech[154].
Given the evidence around vaccine waning it points to the need for boosters to ensure that certification remains effective as a mitigation for transmission. The effectiveness of certification over the next 4-6 weeks and across the festive season on transmission therefore depends on boosters being rolled out quickly enough especially for those who received AZ.
7.5 The impact of boosters
If boosters are rolled out and take up is high, certification will retain its effectiveness against transmission until there is more waning.
Results from the first UK real-world study (n=271,747 people) showed significantly increased protection against symptomatic disease from a booster dose of the Pfizer-BioNTech vaccine in those aged 50 years and older. The absolute VE from 14 days after the booster, using the unvaccinated baseline, was 93.1% in those with Oxford-AstraZeneca as their primary course and 94.0% in those with Pfizer/BioNTech as their primary course[155].
A large observational study conducted using nationwide mass vaccination data in Israel (n= 728 321 individuals), estimated that a third dose of the Pfizer/BioNTech mRNA COVID-19 vaccine is effective in preventing severe COVID-19-related outcomes. Compared with two doses of the vaccine administered at least 5 months before, adding a third dose was estimated to be 93% effective in preventing COVID-19 related admission to hospital, 92% in preventing severe disease, and 81% effective in preventing COVID-19-related death, as of 7 or more days after the third dose[156]. This was an observational study with several limitations, however these early findings suggest that a third dose of mRNA vaccine is effective in reducing severe COVID-19-related outcomes for patients who have received two doses at least 5 months before.
Results from the Phase 3 booster dose efficiency trial announced in October by Pfizer/BioNTech (n=10,000 participants 16 years of age and over) showed that a booster dose restored vaccine protection against COVID-19 to the high levels achieved post-second dose with relative vaccine efficacy of 95.6% compared to those who did not receive a booster. The trial was carried out during a period when Delta was the prevalent variant[157].
The effectiveness of certification will change through time based on waving and timeliness of boosters, suggesting that a time limited certificate dependent on a recent second dose vaccine or booster may be useful to ensure the effectiveness of the vaccine remains high.
This is an approach already being introduced elsewhere, for example in Israel and France.