Chief Medical Officer's Annual Report 2014-15
The Chief Medical Officer Annual Report 2014 -2015 explores the challenges that face doctors today.
Chapter 6 Translation of Medical Research into Routine Clinical Practice
The translation of research findings into clinical practice has transformed healthcare. It is a cornerstone of modern evidence-based medicine and of an advanced healthcare system. However, the route to translation can be challenging; high costs, scarce funds, shortages in key research infrastructure, capacity or capabilities, slow and incomplete recruitment to trials are amongst the potential barriers to the progress of translational research studies. Medical research and development can follow ill-defined and circuitous paths before being taken up into improved patient care.
Over the last decade or so considerable attention has been given to understanding the process better, recognising it is often slow and the advantages of rapid translation of research are considerable. Hence, there is widespread interest in identifying opportunities to shorten translational pathways for the earlier realisation of research benefits to patients, the health service and wider society. Reaping the benefits of advancements in biomedical science including the development of precision medicine, and the care needs of an increasingly multi-morbid population are likely to drive changes to shorten translational pathways.
Translational Lag Time
Estimates, based on analyses of the later impact of medical research conducted during the 1970s to 1990s, suggest the average time for medical research to become embedded into clinical practice may have been around two decades. The considerable duration of this lag is not due only to the time to devise, undertake and report research studies or the nature of research, which often produces outcomes needing further investigation to be understood more fully. It can take time for a body of research evidence to accrue that is considered to be sufficiently well‑developed to be applied into clinical practice. Rarely, for example, are conclusions about the safety and clinical and cost effectiveness of an intervention based on a single study. There can also be other factors at play in the journey from invention to application.
Translational Lags and the Return on Investment in Medical Research
Economic analyses of UK medical research have found returns from investment in different disease areas to be substantial in terms of health gain and economic benefit.. In the area of cardiovascular disease (CVD), for example, it was estimated that UK public and charitable investments in research may produce an annual rate of return of around 39% (about 9% in health gain from new interventions and about 30% direct returns to the UK economy) or expressed differently: for every £1 of investment in CVD research, benefits equivalent to earning 39p per year in perpetuity may be produced. However, these analyses revealed such estimates were very sensitive to the lag between the time of research investment and when the eventual health benefits from research are accrued. The CVD estimate above was based on a lag of 17 years but when this was extended to 25 years the annual rate of return in terms of health gain, for example, dropped to 5.6% and conversely when the lag was shortened to 10 years it rose to 13.4%.
Translational Pathways
Understanding translational pathways to identifying potential opportunities to reduce avoidable time lags has become a major pre-occupation of health research funders, regulators, the life sciences industry and the research community in general.
Figure 4: A critical path for translation of medical research into clinical practice. Taken from Cooksey (2006) A review of UK health research funding.
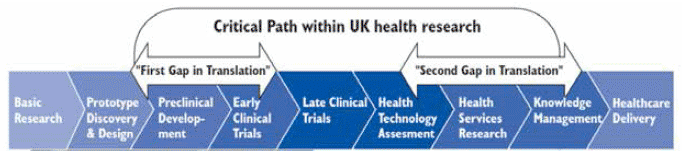
In the UK, the process of translation of medical research and associated lags were considered in detail in 2006 in the influential Cooksey review. This investigated how the potential of UK health research to benefit patients, the NHS and the wider healthcare economy could be optimised. The review defined a critical path for the process of translating medical research into healthcare improvement with a number of discrete stages. Beginning with basic research, through pre-clinical development and then clinical testing into health technology assessment, demonstration and finally implementation of research findings into practice (see Figure 4).
Cooksey also identified two "translational gaps" - points of failure along the critical path where moving promising research further forward into application can be impeded. The first gap related to a failure in the development of ideas from basic or early stage research that could be applied into clinical practice. The second gap related to a failure to implement into clinical practice new healthcare approaches or interventions developed from those ideas. These gaps were considered to be the result of cultural, institutional and financial barriers that could be addressed in part by: greater prioritisation of health research funding (particularly to support medicines and therapies for unmet health needs); greater coordination between research funders with funding directed to supporting promising leads across the translational gaps (for example to support health technology assessment); and a culture in the NHS more supportive of clinical research.
Research funders responded to address these gaps. In Scotland for example, the Chief Scientist Office (CSO) focused its research grants towards the "Cooksey gaps" and created NHS Research Scotland - a partnership between CSO and the Health Boards - to agree and implement national clinical research policies and provide a supportive and efficient environment in Scotland for clinical research in the NHS. Nevertheless, translation remains challenging and continues to receive considerable attention. More recent analysis has represented it as a series of key milestones along a number of different research-based and non‑research‑based tracks (see Figure 5). In an idealised drug development scenario, this pathway begins in the discovery track with a biological target and series of compounds that can act on that target identified, that are taken through a programme of pre-clinical testing to establish the mechanism of action with the most promising candidate(s) tested in animal models to examine the effect on disease and potential for toxicity. In the human research and review track, the safety and efficacy of the best candidate identified from the pre-clinical programme is assessed in a series of clinical trials which when the data are combined and synthesised provide robust evidence of safety and efficacy. In the non-research tracks, the synthesised evidence supports the market authorisation for use in patients by a medicine regulator and, together with economic evidence, a positive recommendation by guideline developers for adoption in the health service for the indicated group of patients. The drug is then supplied and used routinely in the health service.
However, progress along these tracks is often incremental and iterative. For example, in case studies of the route to translation along these tracks of the antihypertensive drug, amlodipine, and the antipsychotic drug, olanzapine, the translational timelines from discovery research to routine UK use of these drugs was judged to be 23 and 20 years duration, respectively. Inspection of the timelines, revealed potential avoidable lags of years between different clinical trial phases, between the completion of clinical trials and syntheses of the findings, and between regulatory approval of the drugs and policy statements on their use in the NHS and then their actual use in routine clinical practice. Thus, these timelines in these cases might have been appreciably shortened.
Figure 5: Research and non-research based tracks of development of a medical intervention with key milestones. The blue arrow represents the direction of the innovation process and the green arrow represents time. Based on Hanney et al.

Improving Translation
Refinements to current process around the discovery research, human research and research review tracks to generate greater efficiencies in translation have been suggested recently with five key areas where there is scope for improvement identified:
1. Better prioritisation of research by defining research questions of relevance and importance to users of research and for which there is an established need based on systematic reviews of the existing evidence.
2. Improved design, conduct and analysis of research studies so that they produce high quality, reproducible research results with a low likelihood of bias.
3. Research appraisal, regulation and management processes that support efficient and robust approval process to ensure high quality relevant research is funded with minimal delays in approval and administration times.
4. Better provision of information about studies underway and on all studies that have been completed, including those that produced negative results, so the complete existing evidence base can be established thereby enhancing evidence assessment and synthesis and reducing unnecessary replication of research.
5. Better reporting of research study methods and findings in order that the findings can be understood and used by others with confidence in an accurate and meaningful way.
While much of the responsibility for improvement in these areas lies with the research community and health research funders and regulators, the wider clinical and patient communities can play important and active roles. Patients, carers and clinicians can become actively involved in research prioritisation. Through initiatives such as the James Lind Alliance these groups can systematically identify and prioritise important uncertainties about treatments, that could be answered by research. In this way, the research agenda cannot be pushed forward by the research community and research funders but also pulled through by users who may be the eventual potential beneficiaries of research. Greater patient (and user) involvement in research design and conduct has been advocated and patient and public involvement in research funding decisions is now widespread.
Reducing avoidable lags in the later non-research tracks is also currently under much scrutiny, notably through the UK Government Accelerated Access Review. This aims to speed up patient access to innovative drugs, devices and diagnostics by looking at how processes may be accelerated in three key areas: assessment of safety and efficacy; health economic assessment and re-imbursement; and uptake by the NHS. The review is part-way through but an interim report published recently has set out a number of guiding principles for development including: greater patient involvement, early identification of emerging products offering the most patient benefit, and supporting innovation along the translational pathway and in the NHS.
Responding to These Challenges
The conventional translational model with average lags of two decades is unsustainable and the demand for new translational models favouring more rapid realisation of patient and health service benefits and economic returns is growing. With advances in genomics and informatics driving better understanding of the molecular basis of diseases, the research opportunities to develop new therapeutics (or repurpose existing ones) and diagnostics to stratify patients to guide treatment (precision medicine) will increase. Combined with innovative clinical trial designs that allow more flexible methods to accumulate safety and efficacy data, the challenge, as predicted by the Accelerated Access Review, will be systems that can evaluate, select and adopt effective and cost effective innovations more quickly.
Scotland has made great strides in addressing this challenge. In terms of study start-up, NHS Research Scotland (NRS) has significantly removed the bureaucracy associated with commencing multi‑centre studies. With generic study issues now being considered once for the whole country, as opposed to being revisited at every site as was previously the case, there has been a significant reduction in the time taken to approve studies. The introduction of a single cost and contract negotiation for the whole country has further increased efficiency and made Scotland a popular destination for commercial trials.
But addressing this part of the process in isolation will not deliver the improvements required for an efficient 21st century health service. For that reason CSO has been working with our Scottish universities with a view to joining up early stage innovative research with later stage clinical testing. Taking a holistic view of the product development pathway is essential if time wasting delays are to be avoided, and patients are to have earlier access to novel treatments.
A good example is the development of a Scottish precision medicine ecosystem, pulling together the academic excellence of our universities with the clinical expertise of NRS, creating a joined up portal through which a single contract - spanning early development work through to clinical evaluation - can be made.
This model developed for research has the scope for wider application. As reported above, the Interim Report of the Accelerated Access Review (AAR) also outlines the need for a coherent and joined-up innovation infrastructure from research to procurement. The Innovation Cluster approach being adopted in Scotland aims to facilitate this move towards a simpler and clearer pathway through the innovation landscape. Initiatives such as the Cluster's work on Open Innovation and a Small Business Research initiative (SBRI) call in diabetes (co-funded by Scottish Government and Scottish Enterprise) are good examples of work in this area.
Contact
Email: Diane Dempster