National cancer plan: progress report - August 2022
Scotland's national cancer plan comes to completion in March 2023. This report uses insights from the available evidence to update on progress against all 68 actions in the plan as at 31 August 2022.
Cancer Plan Progress Updates: Treatment
Data on Treatment
Systemic Anti-Cancer Treatment
Systemic Anti-Cancer Treatments (SACT) is a collective term for drugs that are used in the treatment of cancer. The main type of drugs are cytotoxic chemotherapy drugs but there are other treatments such as targeted agents and immunotherapies.
Weekly and monthly activity reports are generated from the SACT National MVP Data Platform held by Public Health Scotland. Monthly patient numbers are a good indication for long-term trends in SACT activity. Understanding the impact of the COVID-19 pandemic on SACT delivery is important for a full appreciation of the short- and long-term consequences for patients and for planning future cancer care.
Figure 11 shows the volume of patients treated with SACT drugs between January 2020 and July 2022. After a decrease in activity at the start of pandemic, the numbers of patients receiving treatment have increased to levels of activity above those reported in January 2020.
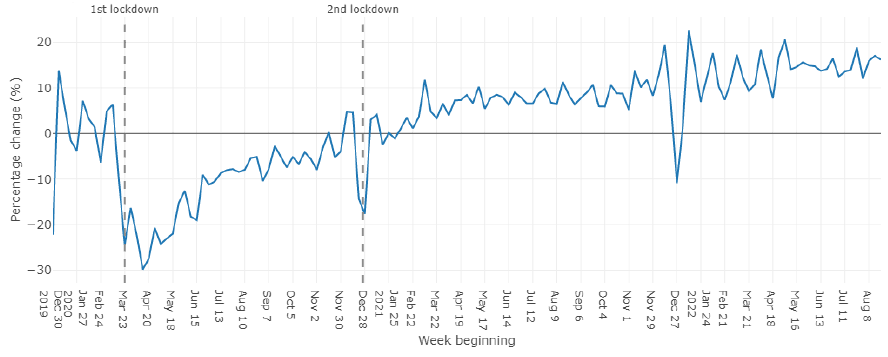
The percentage change of weekly appointments shows weekly activity against an average week in the pre-COVID period. The average week (also called reference week) is defined as the average number of appointments occurring during the weeks beginning 22/01/2020 up to the week beginning 26/02/2020. The percentage change is a good measure to demonstrate the effect of COVID-19 on SACT activity and how cancer services are recovering from the impact.
As shown in Figure 12 below, appointments fell to below average levels between the first and second lockdowns, from March to December 2020, and during December 2021. Falls in appointments during December in 2020 and 2021 may be impacted by Christmas/New Year public holidays, reflecting a similar trend in December 2019. Since the marked fall in activity at the start of the pandemic (-30% at April 2020), there has been an increasing trend in activity over time, with weekly appointments increasing above the reference week level (+14% at June 2022).
Actions for Surgery
Action 32 – Review data on access to specialist surgery
Commitment: We will work with clinicians and Health Boards to assess new ways of improving equity of access across all specialist surgical pathways.
Progress: This work was led by the National Cancer Treatment Group, however this Group was paused in December 2021 due to clinical pressures over winter.
Conversations around access to specialist surgery continue to take place at a regional and local level. The Integrated Planned Care Programme Board is overseeing overall surgical capacity, including the work of the new National Electives Coordination Unit.
Action 33 – Work with National Services Scotland through their Robotic Assisted Surgery Framework
Commitment: We will work with National Services Scotland’s National Planning Board through their Robotic Assisted Surgery (RAS) Framework to assess what potential further investment may be required to make further improvements in this area.
Progress: The Scottish Government has contributed over £20 million to the national framework allowing surgical robots to be purchased for NHS Scotland. Prior to this investment, there were 5 RAS Systems in place in Scotland, equating to 1.1 per million population. As of August 2022, there are now 15 RAS systems in Scotland, which is 3.3 per million population. This number is now comparable to other developed nations.
Investment has also been made in the training of new RAS surgeons with over 35 new RAS surgeons commencing training across Scotland in colorectal, gynaecologic oncology surgery and thoracic surgery. There is a defined process for training RAS surgical teams designed by Intuitive Surgical including practice and online training.
This work has been guided by the RAS Oversight Group and RAS Clinical Reference Group, who report back to the National Planning Board. Recommendations for any future purchase of robots are required to be approved by the National Planning Board before Scottish Government will provide capital investment in RAS.
Data being collected will inform future decisions on purchase of additional new robots or replacements of existing systems.
Action 34 – Promote and review the Framework for Recovery of Cancer Surgery
Commitment: We will continue to promote the Framework for Recovery of Cancer Surgery with regular reviews by governance groups to ensure the Framework remains fit for purpose in the event of fluctuating prevalence of COVID-19.
Progress: The Framework for Recovery of Cancer Surgery was initially published in June 2020 with the Maintenance of Cancer Surgery: Framework being reviewed, updated and a new version published and shared with Health Boards for immediate use in December 2021. This Framework continues to be referenced and promoted at a national, regional and local level. Updates via the Federation of Surgical Specialty Associations are regularly monitored to ensure the Framework remains relevant, and will be considered by the National Cancer Recovery Group.
Action 35 – Identify opportunities to improve recovery after treatment
Commitment: We will monitor and explore new initiatives and service changes to improve recovery after surgery, such as the Enhanced Recovery After Cancer Surgery (ERAS) programme.
Progress: The Centre for Sustainable Delivery has over-arching responsibility for this action. The National Colorectal Enhanced Recovery Initiative continues to support Health Boards in maintaining standards against the national pathway with routine data collection and NHS Scotland discussions. Work is underway to set up similar programmes within Ear Nose and Throat (ENT), Urology and Gynaecology. It is linked to and will benefit from work to progress the Prehabilitation Actions (24-27).
Action 36 – Support awareness of weight management services
Commitment: We will support improving awareness of existing weight management services and referral pathways among cancer health professionals and patients.
Progress: This action is linked to and will benefit from work to progress the Prehabilitation actions (specifically Action 26, addressing nutritional cancer care).
Actions for Radiotherapy
Action 37 – Continued investment in improving out radiotherapy equipment
Commitment: We will invest £45 million in the Linear Accelerator (LINAC) Procurement Programme to ensure access to the most up to date radiotherapy equipment across Scotland’s cancer centres.
Progress: Work is ongoing as part of a rolling infrastructure replacement programme and we continue to invest in the LINAC Procurement Programme. There is some risk around Health Boards finding it difficult to locate adequate bunker capacity for future LINACs. A technical Specification Group who provide oversight of this work meet regularly to discuss and address issues.
Action 38 – Develop a national plan for Scotland’s radiotherapy service
Commitment: We will work with the Scottish radiotherapy community to develop a national plan for Scotland’s radiotherapy service, with a view to curing more cancers, and increasing access to the most modern treatments. The plan will also seek to embed access to clinical research trials across all centres.
Progress: The National Radiotherapy Plan for Scotland was published on 2 March 2022 following a delay of six months from the original timescale for publication, due to service pressures and limited available clinical input. Ongoing delivery of the National Radiotherapy Plan’s objectives will continue throughout 2022 and beyond the term of the National Cancer Plan (due to end in 2023).
The National Radiotherapy Programme Board has met quarterly since 2021 to support delivery of the National Radiotherapy Plan and monitor progress.
Action 39 – Support expansion of radiotherapy peer review
Commitment: We will support the continued expansion of digital solutions to Radiotherapy peer review across Scotland.
Progress: This action has been incorporated into the National Radiotherapy Plan for Scotland published on 2 March 2022 (see Action 38).
As for action 38, the National Radiotherapy Programme Board has met quarterly since 2021 to support delivery of the Plan and will monitor progress in relation to this action throughout 2022 and beyond the term of the National Cancer Plan (due to end in 2023).
Actions for Systemic Anti-Cancer Therapies (SACT)
Action 40 – Increase community-based phlebotomy services
Commitment: We will explore how an approach that increases numbers of pre-treatment and follow-up tests can be embedded across Scotland. We will investigate how the coordinated provision of services through Community Treatment and Care (CTAC) services can support GP practices and maximise economies of scale.
Progress: This is an ongoing action with the Centre for Sustainable Delivery (CfSD) now taking forward exploratory work to scope out models and approaches that CTAC services are using for wider community phlebotomy services. The aim is to create a baseline and highlight lessons learned in order to develop models of community phlebotomy for SACT patients.
Action 41 – Continue and expand the delivery of oral Systemic Anti-Cancer Therapies
Commitment: We will support the continuation and expansion of oral Systemic Anti-Cancer Therapies development, where safe and appropriate through national governance.
Progress: An interim group was established to approve interim treatment options for clinicians during the COVID-19 pandemic. The work of this group has now been superseded by the National Cancer Medicines Advisory Group Programme (NCMAP), hosted by Health Improvement Scotland. The programme was formally convened at the end of January 2022. A governance and operational framework is now in place. The group is accepting and considering new proposals for off-label medicine use, with the first piece of advice issued in July 2022. The number of new/new use of oral SACT treatments is being monitored to evaluate the delivery of oral treatment.
Action 42 – Develop a national approach to non-medical Systemic Anti-Cancer Therapies prescribing
Commitment: We will develop a national approach to support the non-medical prescribing of SACT, to make best use of the existing workforce and help patients receive safe and timely treatment.
Progress: A short-life working group on Non-Medical Prescribing (NMP) convened in December 2021 to identify a set of nationally applicable principles, drawing on current regional frameworks. The SACT Programme Board approved the proposed approach of one national NMP framework. Progress has been slower than anticipated due to pandemic pressures limiting clinicians’ time to meet. Work is ongoing to agree definitions, models of working and required governance. A National Non-Medical Prescribing framework is currently being drafted and anticipated to be complete ahead of March 2023.
Action 43 – Utilise and enhance the delivery of Systemic Anti-Cancer Therapies through community pharmacists
Commitment: We will work with pharmacy leaders to optimise the potential of community pharmacy and build on the successful pilots of community pharmacy dispensing of Systemic Anti-Cancer Therapies (SACT).
Progress: This is an ongoing action. Changes to community pharmacy infrastructure are required before enhanced delivery of SACT through community pharmacists can be considered.
Action 44 – Review and update CEL 30
Commitment: The CEL 30 (2012) guidance on safe prescribing will be updated by October 2021 to ensure guidance on the safe delivery of SACT accounts for recent innovations in medicines, digital transformation and COVID-19 service changes, such as the need for physical distancing in wards.
Progress: A Short Life Working Group was formed to take forward the review and update of CEL 30 guidance in June 2021. A survey was issued to members with a request for updates to the Chief Executive Letter 30 (2012) and guidance for the safe delivery of SACT. Updates have been made to the draft guidance based on the survey responses.
This work was paused from December 2021 until March 2022 due to winter pressures since it is dependent on clinical time. Since restarting, pharmacy and nursing colleagues have reviewed the revised guidance to suggest changes and it was agreed in March 2022 that this work will be taken forward by the SACT Programme Board.
The update is expected to be completed by Autumn 2022.
Action 45 – Embed long-term rapid decision making for off-label medicines
Commitment: We will support the National Cancer Medicines Advisory Group with an additional £400,000 in funding to provide more treatment options for cancer patients, including maximising the use of off-label cancer medicines.
Progress: The National Cancer Medicines Advisory Group Programme, hosted by Health Improvement Scotland, has convened and have agreed terms of reference. Funding of £400,000 has been released. As outlined under Action 41, a governance and operational framework is now in place. The group is accepting and considering new proposals for off-label medicine use, with the first piece of advice issued in July 2022.
Action for Acute Oncology
Action 46 – Support nationally acute oncology services to cope with potential increased demand
Commitment: We will support nationally acute oncology services, where required, to meet increased demand.
Progress: A short life working group was established to look at the optimum model for a national approach to acute oncology. This group has developed a framework for management of acute oncology, which is being published on the Scottish Government’s web pages.
Actions for Precision Medicine
Action 47 – Optimise the provision of diagnosis, treatment, and prevention through precision medicine
Commitment: We will look at the opportunities presented by precision medicine to optimise cancer services, and ways to implement precision medicine to enhance person-centred service delivery.
Progress: The Scottish Strategic Network for Genomic Medicine (SSNGM) was established in May 2022. The SSNGM has 13 ‘facets’ or resource banks of individuals with expertise and interest in specific areas of genomic medicine, including one dedicated to precision medicine. These facets can be called upon for both expert advice and for membership within working groups convened to help achieve SSNGM objectives.
Initial priorities for the Network, which will have subsequent working groups established, are strategy development, workforce, demand optimisation and data standardisation. A working group will also be established to accelerate the implementation of a number of cancer pathways including DPYD testing which looks for dihydropyrimidine dehydrogenase (DPD) deficiency. Having a deficiency in the DPD enzyme could worsen the side effects of certain chemotherapy drugs. This pharmacogenomics test helps to predict patients who may have excess toxicity if given full doses of chemotherapy, and allow planned dose reductions.
Action 48 – Improve alignment of access to molecular pathology testing with new treatments
Commitment: We will work to improve the alignment of access to molecular pathology testing with new treatments, such as the expansion in Advanced Therapeutic Medicinal Products (ATMPs) which over the next few years will offer the opportunity to personalise medicines for individual patients and provide long-term remission.
Progress: The Scottish Strategic Network for Genomic Medicine (SSNGM) replaced the Scottish Genomics Leadership Group in May 2022 as well as the many individual genomics groups to come under a single umbrella. The SSNGM provides strategic leadership for genomics and acts as a front door for engagement with clinicians, academics, industry and others to deliver on a genomics health service for Scotland. The SSNGM Oversight Board has been convened and held their first meeting on 17th August 2022. Lead Clinicians for both Germline/Rare Diseases and for somatic cancer were appointed in September 2022.
With funding from the Scottish Government, the NHS National Services Division (NSD) has put in place a Genomics Transformation Team. This team will support the SSNGM and develop an action plan in response to the major service review completed by NSD in March 2022. It carried out a gap analysis in September 2022 and identified as high priority the development of a process to review Scottish Medicine Consortium (SMC) Horizon Scanning Forward Look Reports and identify tests required for new approved cancer and germline medicines. This will enable the SSNGN to consider the service impact of new and emerging treatments and medicines. It will ensure alignment between SMC approval and the capacity and infrastructure in place in laboratories to make them available to patients.
Contact
Email: CancerPolicyTeam@gov.scot