Recovery and redesign: cancer services - action plan
In response to the pandemic, we have adapted to new circumstances in which cancer services continue to operate. The plan details actions that will both redesign cancer services to benefit patients, and increase our services’ overall resilience to future rises in COVID-19 prevalence.
Treatment
Early detection and diagnosis improve outcomes for patients by ensuring the most effective options for treatment can be used. To improve cancer outcomes and survival, ensuring equitable access to the best treatment must also be prioritised. The Scottish Government is committed to ensuring access to treatments are equitable across Scotland, through a ‘Once for Scotland’ approach, regardless of where an individual lives.
An effective mobilisation of our NHS will lead to a renewed health and care system. This will be achieved in part through embedding innovations and digital approaches. Within cancer services, the use of innovative new approaches in treatment can be seen across each treatment type. The judicious use of new technologies can improve cancer treatments and increase capacity. Various clinical national guidance has been provided through our COVID-19 response and we will continue to support national clinical consensus on cancer treatment to ensure we continue to deliver best treatments to all patients, supported by up to £2.95 million of new investment.
Surgery
32. Specialist surgery offers specialist complex interventions for certain cancers, particularly rarer cancers, and can help improve both experience and outcomes for some patients. We will work with clinicians and Health Boards to assess new ways of improving equity of access across all specialist surgical pathways.
33. Robotic surgery has potential, for certain patients, to improve experience and for surgeons to treat more patients sooner. We will work with National Services Scotland through their Robotic Assisted Surgery Framework to assess what potential further investment may be required to make further improvements in this area.
34. The pandemic has highlighted the need to ensure consistent prioritisation and equitable access to surgery across Scotland. We will continue to promote the Framework for Recovery of Cancer Surgery, with regular reviews undertaken by governance groups to ensure the framework remains fit for purpose, in the event of fluctuating prevalence of COVID-19.
35. Improving a patient’s ability to recover after surgery leads to better patient outcomes. We will monitor and explore new initiatives and service changes to improve recovery after surgery, such as the Enhanced Recovery After Cancer Surgery (ERAS) programme. This programme aims to actively involve patients in their recovery and emphasises reducing physical stress of the operation and encouraging early mobilisation after surgery.
36. Losing weight, in individuals with excess body weight, can help improve patient outcomes and aid long-term recovery after treatment, especially surgery. We will support improving awareness of existing weight management services and referral pathways among cancer health professionals and patients.
Radiotherapy
37. One in four cancer patients in Scotland will receive radiotherapy. Over the duration of this plan, we will invest £45 million in our long established LINAC Procurement Programme, which will ensure the most up to date radiotherapy equipment across Scotland’s cancer centres.
38. We will work with the Scottish radiotherapy community to develop a national plan for the Scotland’s radiotherapy service, with a view to curing more cancers, and increasing access to the most modern treatments. The plan will also seek to embed access to clinical research trials across all centres.
39. The use of peer review in Radiotherapy has accelerated throughout the pandemic, as a result of the increased use of digital solutions, facilitating the best possible decisions about treatments. We will support the continued expansion of digital solutions to Radiotherapy peer review across Scotland.
Systemic Anti-Cancer Therapies (SACT)
40. Increased numbers of pre-treatment and follow-up tests have been provided in the community through COVID-19. For example, phlebotomy services have been provided through Community Treatment and Care (CTAC) services, in a much more coordinated manner. We will explore how this approach can be embedded across Scotland and link to ongoing work on CTAC services to support GP practices to maximise economies of scale.
41. There has been an increase in orally delivered SACT through COVID-19, offering patients better experience and in settings closer to home. Through our national governance we will support the continuation and expansion of this service development, where safe and appropriate.
42. Non-medical staff prescribing of SACT can make best use of the existing workforce and help patients receive safe and timely treatment. It can utilise clinical staff, such as chemotherapy nurses and oncology pharmacists, to increase capacity among other clinical groups for other tasks. We will develop a national approach to support the non-medical prescribing of SACT.
43. Community pharmacists can provide excellent local delivery of SACT. We will work with pharmacy leaders to optimise the potential of community pharmacy and build on the successful pilots of community pharmacy dispensing of SACT.
44. We want to ensure the guidance on the safe delivery of SACT accounts for recent innovations in medicines, digital transformation and COVID-19 service changes, such as the need for physical distancing in wards. The CEL 30 (2012) guidance on safe prescribing will be updated by October 2021 to reflect this.
45. The National Cancer Medicines Advisory Group, working alongside the Scottish Medicines Consortium and existing local systems for access to cancer medicines, will continue to approve new options for treatment, including maximising the use of off-label cancer medicines and use of new rapid decision making processes. We will support the continued work of this group, with up to an additional £400,000 in funding, which will provide more treatment options for cancer patients.
Acute Oncology
46. Where cancers have been detected at a later stage, access to high quality and well-resourced acute oncology services can help patient experience and outcomes. We will support nationally acute oncology services, where required, to meet increased demand.
Precision Medicine
47. We will look to the opportunities offered through Precision Medicine to optimise the provision of diagnosis, treatment and prevention of cancer in our approach to cancer services recovery and thereafter in how cancer services might be redesigned to implement precision medicine to further enhance person-centred, effective and sustainable service delivery.
48. Genetic testing has the potential to identify new medicines for cancer patients. We will work to improve the alignment of access to molecular pathology testing with new treatments, such as the expansion in Advanced Therapeutic Medicinal Products (ATMPs) which over the next few years offer the opportunity to personalise medicines for individual patients and provide long-term remission.
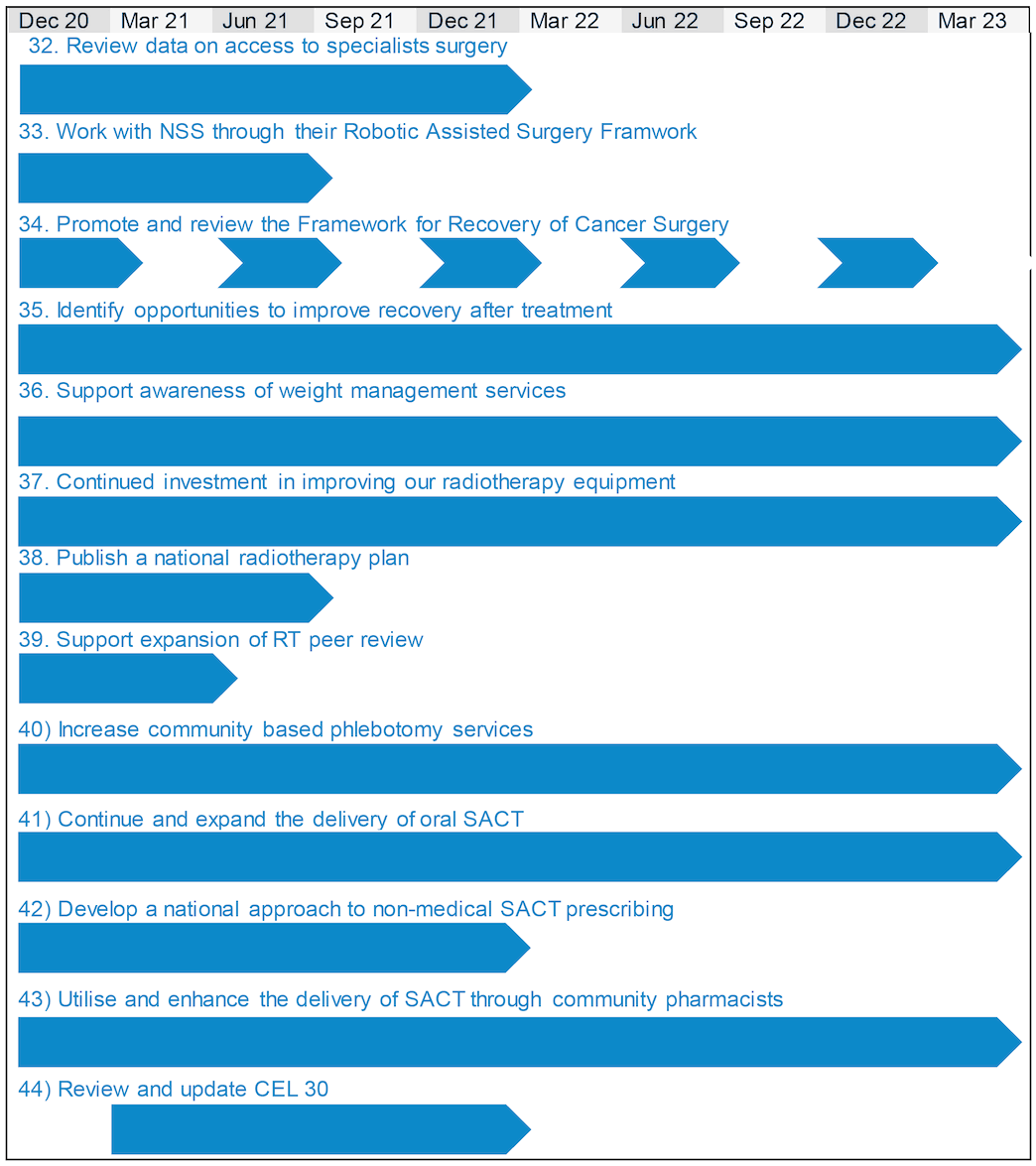

The graphic shows a timeline, displaying the end of each quarter and running from December 2020 to March 2023, and the expected completion times for action points 32 to 48, as described above. It shows that points 35, 36, 37, 40, 41, 43, 46, 47 and 48 are to be completed by March 2023. Action point 32 will aim to be completed by December 2021, while point 33 is shown to be completed by June 2021. Action point 34 shows that reviews will be undertaken every 6 months, and continue throughout the length of the plan, starting in December 2020.
Action point 38 is shown to be completed by the end of June 2021, and point 39 will be done by end of March 2021. The table also shows that both points 42 and 44 will be completed by December 2021, however the review for point 44 will not begin until after December 2020. Finally, point 45 will have system changes completed by December 2020, with long term changes established by the end of the plan in March 2023.
Contact
Email: CancerPolicyTeam@gov.scot