Non-surgical cosmetic procedures regulation: consultation analysis - final report
Independent analysis of the responses to our consultation on the regulation of non-surgical cosmetic procedures, which ran in 2020.
Chapter 1 – Introduction
Introduction
1.1 This report provides an analysis of responses to the Scottish Government consultation on the licensing and regulation of non-surgical cosmetic procedures.
Background
1.2 As part of the Scottish Government's commitment to a safer Scotland, it wants to ensure that anyone providing non-surgical cosmetic procedures does so safely and is competent and trained to do so.
1.3 Non-surgical cosmetic procedures can include work such as dermal fillers, lip fillers and wrinkle smoothers. At this time there is no regulation to prevent or restrict individuals who are not healthcare professionals from carrying out such procedures. The number of these treatments being carried out across Scotland has risen considerably over the years, and there is some anecdotal evidence to suggest there is an increase in procedures going wrong and causing discomfort, harm or permanent damage in severe cases.

1.4 The Scottish Cosmetic Interventions Expert Group recommended a phased approach to regulation.
1.5 The first phase – from April 2016 - focused on the regulation of independent clinics run by certain healthcare professionals – including doctors, nurses, dentists, midwives or dental technicians. These independent clinics were brought within the regulation of Healthcare Improvement Scotland (HIS) from 1 April 2016. This means that the work of these qualified healthcare professionals in terms of non-surgical cosmetic procedures is now regulated and must take place within a clinic registered with HIS.
1.6 The second phase involved considering non-surgical cosmetic procedures that pierce or penetrate the skin and are provided by people who are not qualified healthcare professionals and who work from non-regulated premises. This could include beauty or hairdressing salons.
1.7 The Scottish Cosmetic Interventions Expert Group recommended that non-surgical cosmetic procedures should only be carried out by regulated healthcare professionals, or by people acting on behalf of a healthcare professional (with the professional then having overall responsibility for the quality of care delivered). However, while Scottish Government wishes to ensure the safety of these procedures, it is also keen to explore other options, to avoid undue financial difficulties for reputable small businesses and to ensure that unregulated providers are not driven underground. As a result, it is considering the option of a licensing system backed with rigorous implementation guidelines to ensure best practice. This would be a statutory system to ensure that providers of non-surgical cosmetic procedures that pierce or penetrate the skin are licensed and regulated to ensure both they and their premises meet required standards.
1.8 The Scottish Government also seeks to address a legislative gap which means that registered pharmacy professionals who are operating within an independent healthcare setting are not defined as independent healthcare services regulated by HIS. This is due to an increasing number of pharmacists entering the cosmetics procedure field and providing injectable procedures in premises that are not currently regulated. The Scottish Government is consulting on its proposals that these independent health care services provided by registered pharmacy professionals (and provided outwith a registered pharmacy and not under the terms of an NHS contract) are regulated by HIS in the same way as other independent healthcare services.
The consultation
1.9 The Scottish Government recently consulted on its proposals from 17 January to 30 June 2020 (extended from 30 April 2020). It explored questions around:
1. The need for further regulation of non-surgical cosmetic procedures.
2. Its proposal that those who are not qualified healthcare professionals and who provide higher risk non-surgical cosmetic procedures that pierce or penetrate the skin are regulated by licence under Part 1 of the Civic Government (Scotland) Act 1982.
3. Its proposal to regulate independent healthcare services provided by registered pharmacy professionals, including cosmetic procedures, within an independent healthcare setting where these professionals are not providing pharmaceutical services under an NHS contract. This would mean that these services would be regulated by HIS in the same way as other independent healthcare services.
1.10 The consultation asked 3 closed questions and 4 open questions.
Consultation responses
1.11 The consultation received 459 responses. Most responses were received through Citizen Space, the online platform for consultations.
1.12 The first stage of analysis involved reviewing the responses for duplicate, incomplete or campaign responses.
1.13 Six organisations submitted more than one response. Normally, organisational responses are expected to represent the views of the organisation, and only one response is expected per organisation. In each case, the responses were reviewed and the approach taken agreed with the Scottish Government:
- One organisation submitted six identical responses, plus another shorter, different response. One of the identical responses was retained for analysis and the others were removed. The shorter, different response was re-classified as an individual response – so that the views could still be represented within the consultation analysis.
- Three organisations submitted two responses, and one submitted three responses. In each case, the responses were merged to ensure one response per organisation.
- One organisation submitted a clear organisational response, as well as a shorter, different and less detailed response. The less detailed response was re-classified as an individual response – so that the views could still be represented within the consultation analysis.
- One response was submitted as an individual response, but it was on organisational headed paper and appeared to be an organisational response. Scottish Government checked with the respondent, and it was agreed that this should be re-classified as an organisational response.
1.14 This resulted in the removal of 10 organisational responses from the analysis, due to duplication, to ensure just one response per organisation. Two were re-classified from organisational responses to individual responses. One was re-classified from individual to organisational response.
1.15 In addition, 12 individuals submitted more than one individual response. In each case, duplicate responses from individuals were reviewed to assess which one should be kept. Where responses were identical, one was deleted. If responses were not identical, both were read in full and a decision was taken on which to keep. Generally, the most recent response was retained. In most cases this was more detailed. In a few cases it was clear that individual views strengthened between responses in light of the emerging public health situation at the time when cases of coronavirus were increasing.
1.16 These changes meant that a total of 22 responses were removed, and 437 responses were included for analysis. All amendments were approved by the Scottish Government, and a clear record of the changes made and the rationale for these was kept.
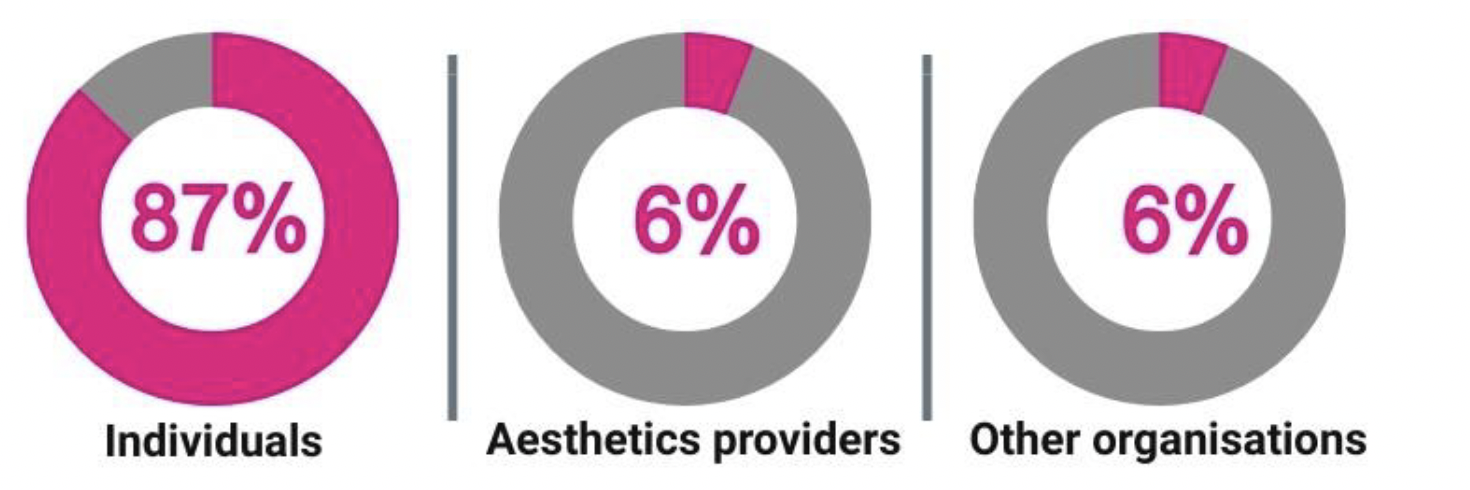
1.17 In total, 382 responses from individuals and 55 from organisations were analysed. The breakdown of responses is shown in the table below.
| Respondent type | Number of respondents | Percentage of responses |
|---|---|---|
| Individual responses | 382 | 87% |
| Breakdown of organisational responses | ||
| Aesthetics providers* | 27 | 6% |
| Membership/representative body | 11 | 3% |
| Public/Patient representation/Advisory | 7 | 2% |
| Local authority | 5 | 1% |
| Regulatory/standard setting body | 5 | 1% |
| Total responses | 437 | 100% |
*An organisation offering non-surgical cosmetic procedures
Quantitative analysis
1.18 For each question, a quantitative analysis of responses was undertaken. This was to help identify the balance of opinion. To undertake this analysis, broad groupings were used:
*Due to rounding, percentages may not always add up to 100%.
1.19 However, it is important to recognise that respondents to consultations are self-selecting and their views are not necessarily representative of these wider groupings.
1.20 Of the 27 aesthetics providers responding to the consultation, 18 were based in Scotland, almost all of which were registered with HIS, indicating that they were led by healthcare professionals. Nine aesthetics providers responding to the consultation appeared to be based in England.
Qualitative analysis
1.21 The bulk of the analysis was qualitative. Responses were sorted and coded, with themes, divergences and similarities in opinion reviewed by the respondent group. For the reviewing of themes, the following groups were used:
- Individuals (382)
- Aesthetics providers (27)
- Membership/Representative body (11)
- Public/Patient representation/Advisory (7)
- Local authority (5)
- Regulatory/Standard setting body (5).
1.22 The responses varied in length and style and not all respondents provided responses to every question. Some respondents provided lengthy responses to open questions, with supporting evidence and references. Open responses varied in terms of how closely they answered the consultation questions. In undertaking the qualitative analysis, it became clear that a small number of respondents had similar answers or used similar wording. It is likely that these respondents shared or discussed their response before submission. However, these have not been classified as a campaign as they are very small in number, and while there are similarities the responses were not identical.
1.23 When analysing the comments we have used a broad scale to indicate the proportion of respondents:
- a few (less than five respondents)
- some (between six and ten respondents)
- many (more than ten respondents).
1.24 The report summarises the range of opinions and does not include every point made by respondents. To demonstrate key points, direct quotes were used. Quotes were only used where respondents gave consent for their response to be published.
1.25 We note that many respondents presented their full opinions to the consultation within their response to the first questions. Thereafter, they repeated their response or referred to previous responses.
1.26 For access to the full responses submitted to the consultation please see: Published responses of the consultation on the regulation of non-surgical cosmetic procedures