Standing Committee on Pandemic Preparedness: interim report - appendix
The appendix provides further information on the Committee’s initial discussions relating to pandemic preparedness, which have identified a number of areas and issues for further consideration by the Committee as they take forward their work and produce their final report.
Initial Areas of Discussion on Pandemic Preparedness
The Committee's initial discussion of pandemic preparedness identified a range of issues, which required consideration as they took their work forward. The areas and issues identified for further consideration by the Committee fall under the following four themes:
- Zoonoses and Surveillance (including the use of Data and Analytics)
- Behavioural Interventions and Community Engagement
- Governance and International Engagement
- Diagnostics, Vaccines, and Therapeutics
The following sections of this Appendix provide further detail of the issues discussed under these themes, and the areas of focus identified by the Committee for further consideration as they take forward this work and produce their final report.
I. Zoonoses and Surveillance (Including the Use of Data and Analytics)
The last 30 months have seen extraordinary investment in the ability to detect and characterise many public health threats. That investment built on a hugely expert scientific and research community in Scotland, which used data generated nationally in order to make significant contributions in the UK and global context. Those contributions have been seen for COVID-19, for example with EAVE II[13], COG-UK[14], virus and patient genomic sequencing, and the characterisation of SARS-CoV-2 variants. The tools and technologies harnessed for COVID-19 continue to support public health protection efforts, including the identification of monkeypox and non A-E hepatitis in children.
The levels of investment in science and research required to address COVID-19 could never be sustained indefinitely, but the Committee shares the view expressed in the SSAC's Report[15] recommendation that it should be a priority in the coming period to:
"Strengthen existing and identify innovative ways of stimulating collaboration across academia, life sciences industry and NHS Scotland science communities"
The Committee's view is that the priorities for action in this area are as follows.
Research and Response Synergies
Investments and efforts to develop improved structures for joined-up disease research and surveillance should draw on existing successful models. In Scotland, for example, the Centre of Expertise on Animal Disease Outbreaks (EPIC)[16], the National Creutzfeldt-Jakob Disease Research & Surveillance Unit[17], and the MRC-University of Glasgow Centre for Virus Research (CVR)[18], are examples of existing entities that provide joined-up research and disease surveillance approaches.
Drawing on the learnings from these collaborations, the creation of a unified body for public health intelligence would enable Scotland to maintain and build its research and response capacity. This type of collaboration would play a key role in joining-up academic and non-academic stakeholders for the investigation of unanswered questions. For example, during COVID-19 the work of EAVE II and the UKRI (UK Research and Innovation) supported National Core Studies led by Health Data Research UK and Office of National Statistics[19] has shown the value of converging NHS expertise with researchers and policymakers, with real-time analyses able to directly feed into policy development. Similar partnerships between CVR and Public Health Scotland (PHS) provided insights into the origin of the introductions of SARS-CoV-2 into Scotland and the emergence of variants as the pandemic progressed.
Case Study: The MRC-University of Glasgow Centre for Virus Research (CVR) and unexplained hepatitis in Scottish children
The CVR is the largest centre in the UK focusing entirely on the study of human viral diseases and viruses at the human-animal interface. Established in 2010 as a partnership between the University and the Medical Research Council (MRC), the CVR contributes to national virology capability and supports the global response to viruses and the diseases they cause.
The Centre has played a pivotal role in the UK and global response to the COVID-19 pandemic. The Centre's contributions have been wide-ranging, from fundamental discoveries on various aspects of virus-host interactions through to outputs with an immediate impact on the UK's response to the pandemic, reflecting the Centre's concentration of expertise and distinctive holistic approach. CVR researchers have also supported the development of protocols for the UK's Lighthouse Lab testing infrastructure, provided viral genomics sequencing training for NHS laboratory staff, and generated research outputs that have informed the formulation of public health measures and policy nationally.
New research methods, such as metagenomic and target enrichment sequencing have recently been used to investigate a cluster of cases of jaundice and acute severe hepatitis of unknown aetiology in young children. This investigation by the CVR has revealed the presence of two viruses: the virus adenovirus AdV41 and a second AAV2 virus coinfection (which can only occur in the presence of helper viruses like adenoviruses).
Metagenomic sequencing is a relatively new technology that requires expertise to implement and interpret. At present it is a research tool but there is huge potential for this to be implemented in future to diagnose many clinical syndromes in which a diagnosis is often not made, including fever in returning travellers (an important group to carry out surveillance on to reduce the impact of imported infection), meningitis (a pathogen currently only identified in around 1 in 4 cases) and other settings. There is great potential for the infrastructure developed for SARS-CoV-2 to be pivoted to this use.

As outlined in the main report, a key recommendation of this interim report is the creation of a Centre of Pandemic Preparedness Expertise, which could bring together, as a 'triple helix' collaboration between Scotland's public health, research, and academic communities, supplemented by input from the wider NHS, industry, and government. Part of the success of a structure such as this will be taking expertise and working relations that now exist and making these industrial in peacetime, with key partners working together around a common purpose and able to understand each other's operational realities.
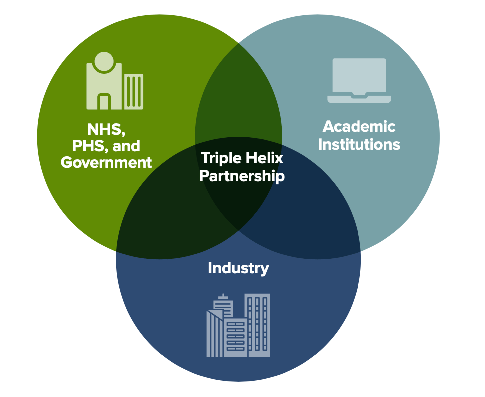
Such a Centre would not need to be a bricks and mortar entity but it would allow to build on Scotland's strengths in, for example, virus research, genomics, and epidemiology to be connected better to surveillance and pandemic response. Governance and structure to allow academics, clinicians, and policymakers to work together across organisation boundaries would be needed, as would infrastructure and resource to allow that to happen. In common with the SSAC, the challenge identified is how to sustain that sense of common purpose in periods between pandemics.
Data Linkages and Facilitating Ethical Approval of Research Proposals
Throughout the COVID-19 pandemic, the analysis of large datasets has been key to identifying signals and characterising the challenges posed by and response to the virus. As for COVID-19, this will be the case for a huge range of pathogenic (and other) hazards. Combining data from multiple datasets can enable the identification and investigation of rarer conditions, with a recent example being the identification of an increase in the cases of non A-E hepatitis in children. For future pandemic preparedness, a platform that is flexible and adaptable and where approvals have been provided in advance in order to be able to carry out rapid, real-time investigations of data is needed. In this sense, data should be considered as key infrastructure, similarly to specialised laboratory infrastructure. Committee members noted that data processes should allow for comparison of data across the Four Nations, and for data to be shared across sectors e.g. from human, animal, plant, food and environmental sources.
Committee members noted the importance of drawing on existing Scottish successes that should be championed in the report and which should be built on, as well as engaging with relevant stakeholders such as Research Data Scotland, NHS Research Scotland, PHS, and Health Data Research UK in order to build a well-governed data system which demonstrates trustworthiness and is secure, reliable, sustainable, and inter-operable. In times of acute need during pandemics, it needs to be scalable, and crucially rapidly accessible to those groups and organisations monitoring and responding to the pandemic. This forms the second key recommendation of the Committee for this interim report.
The work of EAVE II has shown a convergence of PHS expertise with researchers and policymakers. The ability to make near-real-time analyses and modelling with direct routes to policy has been invaluable during COVID-19 and has been instrumental to policy responses globally.
Data accessibility and in particular project delays due to existing information governance arrangements has been identified as a priority issue. Among the challenges noted, delays in existing information governance arrangements such as the Public Benefit and Privacy Panel (PBPP) for Health and Social Care have led to delays to projects such as linking vaccine effectiveness data with viral genomics – data which has been essential to the Scottish and UK governments' responses to the COVID-19 pandemic. There have also been challenges where ethical and information governance approval processes do not include representatives with expertise in a subject, for example in genomic technologies, which can lead to delays and challenges stemming from a lack of understanding of the desired application of data in genomics. Current processes should be reviewed to consider addressing these challenges, which can delay vital research during pandemics.
Case study: EAVE II
Scotland developed a national pandemic surveillance and evaluation platform – Early Assessment of Anti-Virals and Vaccine Effectiveness (EAVE) – in response to the H1N1 pandemic in 2009-10. This was then put into hibernation following the end of the H1N1 pandemic. Following the emergence of SARS-CoV-2, EAVE was brought out of hibernation to create EAVE II.
EAVE II brought together primary care, testing, sequencing, vaccination, hospitalization, intensive care unit, mortality, and a host of other datasets on 5.4m people (~99% of the Scottish population) into PHS.
EAVE II is one of the world's few national, end-to-end, near real-time COVID-19 data platforms. The EAVE II team have produced many major analyses and among these, their analysis was the first in the world to demonstrate the real-world effectiveness of first dose COVID-19 vaccines in preventing COVID-19 hospitalizations.
Bringing EAVE out of hibernation to create EAVE II was a time consuming process, thus largely reflecting how information governance procedures were not fit-for-purpose in the context of a pandemic. Other challenges encountered were the lack of trained data processors and analysts with permissions to access these data.
Going forward, it is crucial that the data infrastructure and associated capabilities developed are 'kept on the boil' to allow more rapid responses to any future pandemics.
Image Credit: University of Edinburgh – EAVE II
II. Behavioural Interventions and Community Engagement
Responses to future threats should be designed and delivered in such a way as to consider knowledge, capabilities, attitudes and behaviours, and the environments and circumstances in which people live and the groups most likely to be affected. No amount of pandemic planning will succeed if interventions are not understood, accepted, and supported by the public. At this stage, the Committee has identified: a) the most relevant issues for attention in this regard; and b) priorities for action for decision makers in Scotland to consider.
Issues for Attention
The Committee's initial discussions identified four broad sets of issues that are likely to be critical in any future pandemic.
Connections between authorities and the public. Pandemic measures should not be done to the public but with and for them[20]. Public engagement and inclusion are critical to the development of trust and the legitimacy of measures. Building trust depends upon an effective two-way process of communication. On the one hand, authorities need to be consistent, open, and transparent about their decisions. On the other, they need to be seen to listen and respond to the needs and concerns of the public. In addition, the legitimacy of specific decisions is bound up with a clear commitment to ethics and rights, both in terms of the content of measures and the processes by which they are developed.
Connections and relationships amongst the public. Throughout the current pandemic, we saw that a sense of community, social ties, and feelings of moral responsibility had a major influence on public contributions to the pandemic response[21] on multiple levels: adherence to pandemic measures; developing informal networks of support and care that sustained people in hard times; enabling social participation in educational and other activities; and sustaining mental and physical well-being.
Inequalities between groups. Pandemics and other crises result in deprived and marginalised groups being more exposed to harm, less able to deal with harm, and less able to engage or comply with measures designed to mitigate harm[22]. A successful pandemic response depends upon recognising these differences, understanding the barriers in different groups, building relationships with these groups, and ensuring that pandemic responses address the support necessary for all members of the public to avoid harm.
Capacity. The design and delivery of any interventions that involve behaviour change depends upon developing a strong and broad base of behavioural/social science capacity which can be accessed by decision makers and that can inform the design and delivery of interventions.
Priorities for Action
Structures of communication: Successful communication needs to be a two-way process in which the ability to communicate with the diverse publics in Scotland depends upon listening to these publics[23]. This depends on developing structures of public engagement that are fleet-footed enough to facilitate quick decisions in a fast-moving crisis, that allow genuine public input into the decision making process, and that includes those who traditionally have less voice. Successful communication also needs to complement trusted information with a challenge to dis – and mis-information that undermines the pandemic response. This requires the development of rapid-response structures that identify and correct false information. In both areas, we need to draw on experiences in Scotland[24] and international best practice[25] to develop structures and methods that are best suited for meeting the needs of both longer-term preparedness and emergency response.
Assessing inequalities and barriers to adherence: Pandemics, and measures to control pandemics fall on groups in different ways, with marginalised, minoritised, and deprived groups generally both hardest hit and least able to respond. It is therefore essential to develop a methodology that allows a rapid assessment both of (a) how different groups are affected by a pandemic (and hence what response measures are needed); (b) the different barriers to engagement/adherence in different groups and how these can be overcome.
Building connectedness in the community: There are two levels at which the issue of connectedness needs to be addressed. The first has to do with the structures of community resilience, both established (such as community anchor groups) and emergent during crises (such as the 'mutual aid' groups that played such a pivotal role during the COVID-19 pandemic[26]). The existence of these is fragile, so how can they be supported and resourced to function more effectively? The second level is virtual/digital. Lack of online connectivity severely affected already marginalised communities in terms of having contact with others, access to information, and ability to learn[27]. How can we address digital inequality both in terms of provision of equipment, connectivity and digital skills? Both of these issues extend well beyond pandemic preparedness, and the Scottish Government has put forward a digital strategy[28] for Scotland, but we should consider how our approach to pandemic preparedness will support existing initiatives to address these issues, and vice-versa.
Building and harnessing expert capacity: We must develop behavioural/social science expertise in Scotland, identify and draw on existing experts (e.g. via RSE), embed such experts with policy makers and practitioners (e.g. ensure that expertise about social influence is employed in the design, production and evaluation of behavioural interventions and communication), create structures for rapid input and response during emergencies, and also ensure that adequate expertise is represented at all levels of the advisory process.
III. Governance and International Engagement
Scientific advice has played a central role in the Scottish Government's response to COVID-19. There are a number of different channels, formal and informal, internal and external, through which advice fed into Scottish Government policy.
Future pathogenic threats are likely to originate abroad rather than in Scotland as for example, the risk of a pandemic starting is considered greater in other regions of the world than in Scotland. There is therefore a high value on ensuring Scotland's surveillance and advisory structures are well connected formally and informally to UK and international bodies. That is true both for the best gathering of intelligence and data, but also in terms of ensuring Scottish interests are taken into account in setting the questions and work programmes of key advisory bodies.
The Scottish Government benefitted hugely from participation in the Scientific Advisory Group for Emergencies (SAGE) and access to SAGE and its subgroups' papers and material from groups such as the Joint Committee on Vaccination and Immunisation (JCVI) and the New and Emerging Respiratory Virus Threats Advisory Group (NERVTAG). The connections between the UK's four Chief Medical Officers (CMOs), and between the Chief Scientific Adviser (CSA) for Scotland and the wider CSA Network all were and are of huge value in allowing for the dissemination of the best clinical and scientific expertise across the Four Nations. It is however hugely valuable to supplement that approach with Scottish-focussed groups. The Scottish Government COVID-19 Advisory group, and its subgroups, were cited as an example of a structure that allowed scientific data to be interpreted in a way that was sensitive to the specifics of Scottish society.
It is important that Scotland is well connected and able to leverage networks that will allow its public health agency and academic community to access essential information on emerging threats. Much of that is already achieved through participation in UK and global scientific structures, however the Committee agreed that there is a need for these to be supplemented by structures which brings together leadership in research, operational, and policy expertise in Scotland and which therefore allows Ministers and policy advisors to have swift and direct access to advice in the face of an emerging threat.
Enhancing Scotland's Links with International Partners
The Committee agrees the importance of clear information flows and reciprocity of access to scientific advice between the Scottish Government and UK-level and international advisory forums group and supports the recommendation of the SSAC[29] that there is a need to "formalise the network of scientific advisers within the Scottish Government and its role in accessing integrated scientific knowledge from outside government[30]".
Part of that recommendation describes the need to map out and develop a greater understanding of the existing advisory ecosystem. The Committee has therefore commissioned the Scottish Government to produce a high-level mapping of flows of scientific advice and information to and from the Scottish Government. The Committee will use this mapping as it takes work forward to analyse information flows and access to scientific advice by the Scottish Government with the aim of identifying where gaps exist and where existing connections could be strengthened to increase the resilience of these arrangements. The aim should be to create a clear pathway for Scotland to draw on international best practices and learn from the experiences of other countries. As noted in the third key recommendation, where links with Scottish, UK, and international scientific advisory structures, networks, and agencies (including WHO, World Organisation for Animal Health, and European Centre for Disease Control) are not through formal membership, good working relationships should be fostered in order to strengthen information flows from these and ensure Scotland has access to the most up-to-date evidence and practice in the face of future threats.
Following the publication of this Interim Report, the Committee will also engage with a wide range of stakeholders to develop a greater understanding of the current flow of information from the Scottish Government to its domestic partners and where there may be opportunities to reinforce this. These workshops will also provide an opportunity to engage with learned groups such as the RSE and the SSAC. For the RSE, this ties to the recommendation in its 'Coming Out of Covid-19: Reimagining Scotland'[31] report, that the "RSE should build on its existing international connections with national academies and other key institutions overseas to facilitate the sharing of learning between countries on shared challenges[32]."
During the COVID-19 response, the creation of the Scottish Government COVID-19 Advisory Group provided the Scottish Government with a forum where information from bodies such as SAGE could be considered in order to inform the development of policy for Scotland. The Committee noted the role of this group and other public sector collaborations. The Committee noted that arrangements required to establish collaborations of this type and allow their redeployment redeployment during national emergencies should be reviewed across the public sector, in order to facilitate the rapid formation and/or redeployment of these in future pandemics, and endorsed the recommendation of the RSE report, which highlights the need to identify "how best to support financial resilience and how to build resilience into physical and administrative structures[33]."
IV. Diagnostics, Vaccines, and Therapeutics
In any future pandemic, Scotland must be able to utilise rapid diagnostic testing at large scale, provide evidence-based therapies, and access the development and delivery of safe and effective vaccines. These aims are challenging, as a future infectious agent with pandemic potential may be entirely novel, and the scale of response required could not be maintained at such a level in the absence of a realistic threat. Scotland's experience and expertise in clinical trials is a strength which can be built on and future research may lead to completely novel methods of diagnosis or vaccine delivery which may require very different responses to those developed for COVID-19.
Development of novel diagnostics, vaccines, and therapeutics will be an international effort, but the Committee noted the successful partnerships between academia, the health service, and industry, which were vital contributors to a comprehensive and effective response against COVID-19. The fourth key recommendation of this Interim Report focusses on the importance of continued support for innovation in life science and public health research, for the development of diagnostics, vaccines, and therapeutics to provide the capability to respond to novel threats when required. Part of this is will be to ensure Scotland has adequate capacity in human resource to meet future pandemic challenges, and to cement the inter-sectoral collaborations that were of such value for COVID-19.
The challenge is to formulate structures that can function usefully in the absence of a pandemic, which can then be rapidly pivoted (and expanded if required) to meet the urgent needs of a novel infectious threat. The Committee's view was that this could be best met by providing technologies and capabilities that can be utilised within Scotland under 'normal' conditions, which could then be adapted to contribute to the immediate demands of a pandemic response. Routine contracts with industrial partners should also be created, for useful, routine, diagnostic testing which can be adapted to provide the capability to respond to novel threats when required. The same approach should be considered for vaccines, where possible.
Scaling Diagnostic Testing
Providing rapid, sensitive, and specific testing at scale is an essential part of a pandemic response. There may also be opportunities to harness new technology for initial screening and point of care testing, such as has been done in community drug clinics. Existing nucleic acid amplification testing will likely remain important, but newer technologies, in particular rapid and/or point of care tests will also be required in the face of a new infectious threat. Although in the early stages academic/UKHSA/reference labs will most likely develop a novel assay, as has happened for pandemic flu, COVID-19, and monkeypox, industry is best placed to develop and to deliver such technologies at scale.
Linking Diagnostics with Genomics, and Training in Partnership
Pathogen genome sequencing is also an essential part of a pandemic response, as it can link diagnosis with clinical outcomes, response to therapeutics and vaccination status, as well as tracking the emergence of potential novel variants and helping to identify and understand the potentially problematic characteristics of variants, including resistance to particular therapeutics. This expertise was invaluable during the COVID-19 pandemic and would be similarly important in responding to potential future pandemics and other outbreaks/incidents that might occur (e.g. the recent non A-E hepatitis cases in children and previous legionella community outbreaks) by repurposing current COVID-19 capacity to other areas of significance in healthcare. Such areas include monitoring antibiotic resistance in bacteria, detecting potential transmission of environmental organisms into patients within secondary care, monitoring of wastewater for viruses, and tracking blood borne viruses to monitor micro-elimination of both human immunodeficiency virus (HIV) and hepatitis C virus lineages.
Many academic organisations either conduct sequencing in-house or contract out sequencing of multiple infectious agents for research and development purposes to commercial providers. A publically underpinned facility might be able to attract sufficient income from multiple users across Scotland to cover recurrent costs in inter-pandemic periods. Consideration should also be given to developing emerging sequencing technologies such as metagenomics, as well as improving turnaround times, which would allow more agile public health actions and adjustment of clinical management.
Collaborations between academic institutions, PHS, and industry were very successful in the response to COVID-19 and it is vital to maintain a workforce that can adapt to the needs of a pandemic, and that strength in designing and performing clinical trials is maintained. Sufficient capacity in terms of expertise in trained staff in bioinformatics and sequencing capability, should be maintained to respond to potential future pandemics and other outbreaks/incidents, alongside the formalising of collaborations between academia, PHS, and industry, including joint PhD programmes, clinical secondments, and industrial placements. Using flexibility in staffing would help take forward these ideas, working with Scottish Government and Scottish Enterprise towards offering apprenticeships that will provide technician as well as graduate jobs. The Committee also discussed the potential of establishing a core retired team with relevant skills, such as in bioinformatics and sequencing, which would have access to yearly updated training, ensuring they were ready to be deployed on a voluntary basis should this be needed.
Funding Clinical Research
The Committee noted that funding of clinical research by the Chief Scientist Office (CSO) has remained static since 2012 and appears significantly less than the funding in England provided by the National Institute for Health Research (NIHR)[34]. This should be reviewed in order to provide the optimal infrastructure necessary for Scotland's future pandemic responses.
Ensuring a Sustainable Manufacturing Base for Diagnostic, Therapeutic, and Vaccine Production and Delivery
We need to ensure that novel therapies, diagnostic reagents, and vaccines are available in Scotland in the face of a pandemic threat. 'Stockpiling' of key therapeutics or vaccines was felt by the Committee to have inherent problems in terms of potentially wasted investment on agents that were not required or became unusable over time.
The Committee noted the importance of close dialogue with the UKHSA, the Department of Health and Social Care and other relevant UK institutions to ensure Scotland is a stakeholder in decisions over any procurement on a Four Nations basis, including around stockpiles.
In terms of manufacturing capacity for vaccines, as considered above in relation to diagnostics, establishing local production of routine vaccines (which may utilise novel technologies in the future) would allow the demand of a pandemic threat to be met at speed.
Data Flow and Diagnostic Centres
Linking patient data to diagnostic test results and vaccination status is an essential part of a pandemic response. Inconsistency in information technology systems and data sharing between health boards hinders this response. The Committee noted the importance of a National Laboratories Information Management System being completed as soon as possible so that all health boards are using the same system. Consideration should also be given to a national diagnostic strategy, which includes specific consideration of pandemic preparedness.
Many of the 50 or so diagnostic laboratories in Scotland have precarious funding – a smaller number with sufficient throughput to be confident of maintaining expertise could be more sustainable. Consolidating laboratories would also enable them to be more flexible in responding to future threats. Beyond NHS diagnostic laboratories the Committee suggest that the critical role of Reference laboratory services and separately non-NHS laboratories (academic, private and partner one health laboratory services) are considered as part of a national diagnostic strategy to respond to a future pandemic threat. The Committee noted that a review of available laboratories across Scotland relevant to pandemic preparedness is currently being conducted. In the context of pandemic preparedness, a clear National Laboratory Network which encompasses both Reference laboratory requirements and wider One Health activities would be desirable outcomes. Linking data outputs across the Network would also form an essential element. The Committee will consider the current laboratory review further as it takes its work forward, before reaching conclusions.
Contact
Email: scopp@gov.scot