Scotland's place in Europe: science and research
Our latest analysis of the implications for Scotland’s science and research if the UK exits the European Union.
Scotland's Participation in European Research
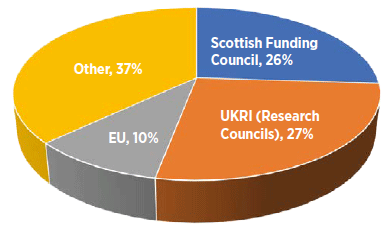
Research and innovation at Scottish universities remains cutting edge in global terms. The Scottish Government provides around £500 million annually for science and research in Scotland at universities, research institutes and public bodies, including NHS Scotland, to support maintaining this status. Scottish universities (which play the biggest role in research) receive around 10% of their research income from the EU, to the value of around £105 million per year. EU funding for other research organisations and companies in Scotland is in addition to that secured by universities.
Figure: Sources of university research funding in Scotland 2016-17 (Percentage of £1.05 billion)
Participation in European research programmes gives Scottish universities and other organisations access not only to EU funding but also to collaborative networks, expertise, facilities and data that add immense value to their work. The case study shown on the right is a striking example of that.
The Scottish Government fully expects that any assessment of value for money regarding UK participation in European programmes needs to consider these non-financial factors.
Horizon 2020 Case Study: European Prevention of Alzheimer’s Dementia (EPAD)
Project – an interdisciplinary research programme under Horizon 2020 involving public and private sector organisations across Europe, including the University of Edinburgh. This is a major pan-European initiative to create a novel environment for testing numerous measures targeted at the prevention of Alzheimer’s dementia.
Aim – to understand better the early stages of Alzheimer’s disease and prevent dementia before symptoms occur as part of a global effort in the fight against Alzheimer’s disease.
Benefit – preventing the development of dementia in people at risk would be a fantastic step forward in our fight against Alzheimer’s disease. The EPAD and its novel approach to trials should help speed up the drug discovery progress and bring us closer to this ambitious aim.
Impact – this project has numerous advantages over current approaches. These include an excellent pre-trial description of research participants to inform selection and reduce screen failure, the establishment of the highest possible quality study sites across Europe, the rapid decision making on the likely success of a drug (or combination of drugs) in subsequent confirmatory trials as well as access to a placebo group shared across Europe.
Contact
Email: Pieter van de Graaf