New Medicines Reviews 2013
Reports on the role of SMC & Review of IPTR & ADTC
A Way Forward
6 - Recommendations
In our view, SMC has efficient, robust, independent and timely HTA processes. It is important that these attributes can be clearly observed by those who may be affected by the process and by the advice given to NHS Scotland by SMC. The principle of "procedural justice" adopted by NICE emphasises the importance of ensuring that the processes by which decisions are reached are transparent, and that the reasons for these decisions are explicit (National Institute of Health and Clinical Excellence 2008). Thus in order to further increase the transparency of the process for patients, patient groups, the public, by health professionals and by pharmaceutical manufacturers,
Recommendation 1. SMC should meet in public so that members of the public, patients, patient group representatives, other health professionals and members of the pharmaceutical industry can attend to observe the appraisal process.
This will allow all these groups the opportunity to hear the discussions leading up to the decision-making process. Patients and patient group representatives can observe how the patient voice provided by their submissions contributes to the discussion, and those working in the NHS or pharmaceutical industry can observe the rigour of the scrutiny being applied to the sources of evidence. One HTA body (AWMSG in Wales) has met in public since its inaugural meeting in 2002, and NICE has now conducted its appraisal meetings in public for several years. After the public documentation, any further deliberations can be held in private before the committee' view is ascertained and announced to the audience. This may be by general assent (as is most commonly the case in SMC and NICE) or by confidential simple majority vote (as occurs at AWMSG). Because of the "commercial and in-confidence" nature of deliberations around medicines associated with a patient access scheme (PAS), part or all these particular appraisals will still need to be held in private at the present time.
Recommendation 2. SMC should invite the manufacturer of the new medicine under consideration to give evidence at their main SMC appraisal meeting, in order to address any outstanding questions that SMC members have and highlight any outstanding issues of which they believe SMC should be aware prior to its advice being published.
This ensures that manufacturers can correct factual errors, highlight any criticisms of the process, and clarify any outstanding issues for committee members before they make a judgement. It thus also allows the manufacturer to be assured that no relevant issues have been overlooked, and that the subsequent committee decision is made based on a sound and complete evidence-base. It also allows them to give assurances to the Committee that they believe the process to have been fair and transparent.
It is disappointing when manufacturers decide not to engage with a HTA process. Non-engagement can result in uncertainty within the NHS regarding the clinical effectiveness and cost effectiveness of the medicine for that particular indication. While some of these non-engagements may be for indications for which alternative treatments are already available, some may be for innovative medicines, which may have potentially significant benefits for patients.
It is essential to ensure that the medicines appraisal process continues to remain comprehensive, and provides timely advice to NHS Scotland on all relevant new medicines. To address the challenge associated with non-engagement of manufacturers with the SMC process (this was 30% of all new medicines in 2012) and guided by the views of Health Boards, ADTCs and clinicians:
Recommendation 3. SMC should be able to appraise any new medicines which the NHS in Scotland considers potentially of major importance to patient care, but which have not been submitted to SMC by the manufacturer within 3 months of launch. If necessary this appraisal may be conducted using such data as is already available in the public domain.
This helps to reinforce the centrality of the patient in the HTA process, as well as the importance of the views of clinicians, who are striving to do the best they can for their patients. While a "not recommended" notice may be issued when non-engagement occurs, clinicians may still remain uncertain of the clinical effectiveness of the medicine and its potential place in treatment. They can therefore benefit from access to a rigorous and independent appraisal of the evidence drawn together from available sources already in the public domain. Information on these medicines can be obtained from SMC's well-developed horizon scanning process, as well as by regular dialogue with the appropriate clinicians and patient groups in Scotland and with the ADTCs.
In order to further improve the responsiveness of the HTA process, it is vital that there is close dialogue between the HTA body and the manufacturer (e.g. in scoping, choice of comparators etc.) from the beginning of the process. To further facilitate this interaction,
Recommendation 4. SMC should be able to have a temporary pause in the appraisal process at any stage in order to permit further dialogue with manufacturers on issues that would be likely to be central to the subsequent decision-making process.
While it is possible to conduct a reappraisal, this process can be expensive and result in duplication of preparation of evidence and wasted committee time, resulting in unnecessary expense for all parties. It may also result in the issuing of several notes of advice for the same medicine in a particular indication. Using this option when SMC considered it appropriate should help to reduce the resubmission rate, and while the advice might be slightly delayed by such consultation, it would improve efficiency, clarity of recommendation, and ensure that the SMC process was even more responsive than is the case already.
In order to further address the specific challenges associated with "ultra-orphan" medicines (those medicines licensed for the treatment of diseases with a UK prevalence of less than 1 in 50,000)
Recommendation 5. SMC should develop a policy specifically relating to ultra-orphan medicines to guide the process of consideration of all available evidence relevant to its advice on these medicines.
Elsewhere, method to assess the views of the publics such as a "Citizen's Councils" or "Citizen's Jury" have been successfully used to explore specific societal issues in relation to the availability of new medicines. To further improve SMC's engagement with patients and the general public,
Recommendation 6. SMC, with the appropriate resource, and in partnership with other relevant bodies in Scotland, should be encouraged to set up an engagement process such as a "Citizen's Council" or "Citizen's Jury" to explore views around specific societal issues of importance to the people of Scotland in relation to the availability of new medicines, and the impact of these views on the existing processes for ensuring access to medicines.
This work can be led by the public partners and the Patient and Public Involvement Group (PAPIG), who are ideally placed to help to build on SMC's existing strong commitment to further improving transparency of the HTA process.
Recommendation 7. SMC should explore other innovative approaches to increasing patient and public awareness of its role in ensuring timely access to clinically effective and cost-effective medicines in Scotland. Consideration should also be given to expansion of its role to support other aspects of safe, effective and cost-effective prescribing. SMC should produce a publicly available annual report of progress in this regard detailing its important contributions to this objective.
Value Based Pricing (VBP) is intended to ensure that the prices of new individual branded medicines reflect their 'clinical and therapeutic value to patients and the broader NHS. It is designed to link the pricing of a medicine to the benefit it has been shown to deliver, so that access to new medicines can be further improved. The Office of Fair Trading (OFT) noted that 'in the interests of patients, it is vital that NHS resources be used cost effectively. Since their creation, NICE, SMC and AWMSG have made a significant contribution to achieving this aim". The OFT report on The Pharmaceutical Price Regulation Scheme: An OFT market study also stated that "NICE, SMC and AWMSG are natural candidates for conducting the cost effectiveness analysis required to implement value-based pricing" (OFT2007b). These skills within SMC could be harnessed to support the development and delivery of VBP in the future.
Recommendation 8. NHS Scotland should explore ways in which the expertise available within SMC could be used to support the process of Value Based Pricing (VBP).
When a medicine is not recommended, it is important that The Individual Patient Treatment Request (IPTR) process possess the same qualities (transparency, timeliness, relevance, in-depth robustness, and usability) as an ideal HTA process. For these reasons,
Recommendation 9. A register of IPTR decisions in all Health Boards, suitably anonymised to protect patient confidentiality should be kept, and supporting information related to IPTRs shared between Health Boards.
Recommendation 10. There should be regular sharing of experiences between the IPTR panels and members of IPTR panel members across Scotland should meet at least annually for feedback and training.
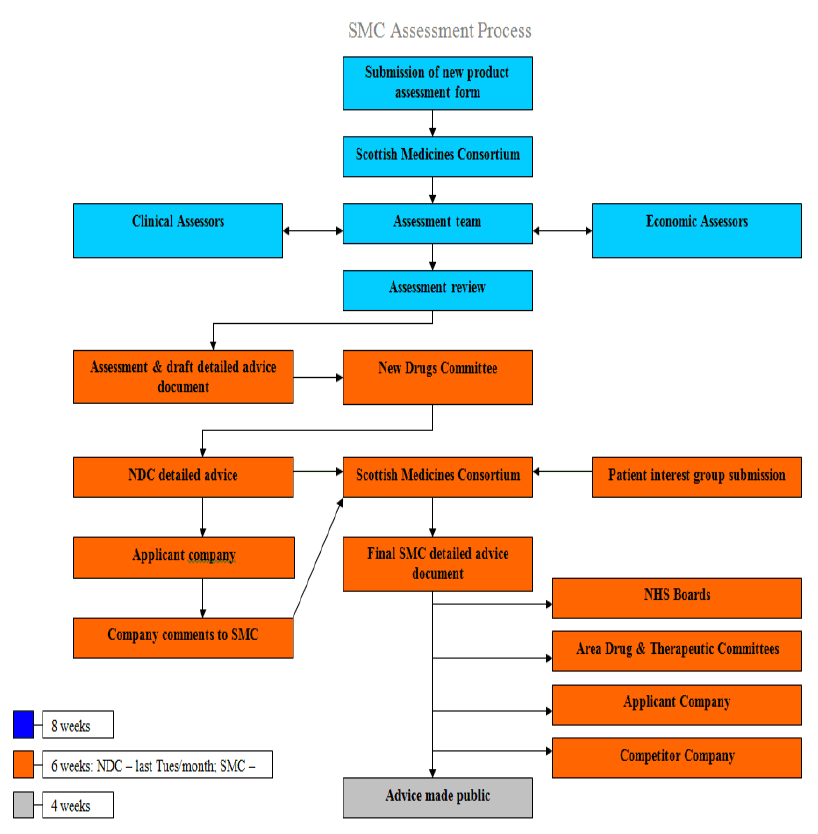
Figure 1. The process used by SMC for Health Technology Appraisal
Contact
Email: VERONICA MOFFAT