New Medicines Reviews 2013
Reports on the role of SMC & Review of IPTR & ADTC
3 Results
The findings below detail the figures from the three key questions.
Twelve boards responded with complete data. The two NHS boards that did not provide data follow the decisions of NHS Grampian's ADTC; therefore their data duplicates NHS Grampian's.
For the purposes of this report, the data from the 12 NHS boards that responded will be used in the analysis.
Also included is information on the accessibility of information of new medicines from each NHS board's website.
3.1 What was the decision taken on each medicine by each NHS board?
All 12 boards made a decision on all 23 medicines.
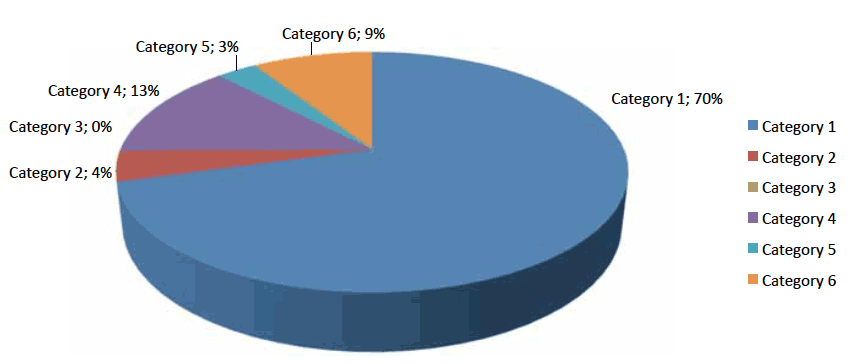
The average percentage uptake of SMC accepted medicines on to the 12 NHS boards' formularies was 74% (17 out of 23) (decision categories 1 and 2). One of the 12 NHS boards included all 23 medicines on to their formulary.
CMO(2012)1 detailed the following categories for formulary decisions. Ten out of the 12 NHS boards used the categories below to indicate their ADTC decisions.
- Included on the NHS board formulary (to include specialist, approved, additional lists) for the indication in question.
- Included pending protocol.
- Not included* from the NHS board formulary because the NHS board decision is that the medicine does not represent sufficient added benefit to other comparator medicines to treat the condition in question which are already available in the formulary.
- Not included* from the NHS board formulary because clinicians do not support the formulary inclusion.
- Not included* from the NHS board formulary because clinicians have not responded to an invitation to apply for formulary inclusion for this medicine.
- Not included* pending protocol.
* Where a medicine has not been included in the formulary, there will be a link to the formulary in order that the comparator medicines can be viewed.
Appendix 3 details the breakdown of each NHS board's decisions on the 23 medicines.
Figure 1 below illustrates the breakdown of ADTC decision categories used by the NHS boards for all 23 medicines.
Figure 1: Breakdown of NHS boards' use of ADTC decision categories
3.2 Was the decision taken by the ADTC within 90 days?
The NHS boards made an average of 87% (20 out of 23) of their decisions on all 23 newly licensed medicines within 90 days (see Appendix 5). Four boards made 100% (23 out of 23) of their decisions on all 23 newly licensed medicines within 90 days. One board made 96% (22 out of 23) and two boards made 91% (21 out of 23) of their decisions on the 23 medicines within 90 days. The remaining five boards made between 57% (13 out of 23) and 78% (18 out of 23) of their decisions within 90 days.
Four medicines in particular did not have their decisions made on them within 90 days, across five boards.
3.3 Was the decision published on the NHS boards' website?
On average, 70% (16 out of 23) of ADTC decisions were published by the boards, on their websites, within 14 days of their ADTC decisions being made.
Four NHS boards published decisions of all 23 medicines on their board website within 14 days of the ADTC decisions being made. One board published 91% (21 out of 23) of their ADTC decisions on to their board website within 14 days. Six boards published between 26% (6 out of 23) and 83% (19 out of 23) of their ADTC decisions on their website within 14 days of their ADTC decisions being made. One board did not provide dates of publication for their ADTC decisions (see Appendix 5).
3.4 How accessible was this information to patients and the public?
We viewed all 14 NHS boards' websites to determine accessibility for patients and the public on the decisions made by their own board's ADTC, using the search terms detailed in Appendix 2.
CMO(2012)1 guidance outlines the type of list that NHS boards are expected to maintain on their website (see Appendix 6), in relation to SMC medicines and formulary decisions. NHS boards are expected to publish updated lists of SMC accepted medicines included and excluded from their formularies, together with the rationale for such decisions.
It was found that seven boards had easy access for the public to see ADTC decisions and patient information, and allowed access in a logical and intuitive way to the boards' formularies. The information was not always presented in accordance with CMO(2012)1.
For three boards, their access for the public was somewhat limited. For example, one board had an external formulary website that gave excellent information on local formulary decisions, but nothing linking the main board website to the external site. This means patients or members of the public would only be able to find the information if searched for directly in a search engine, such as Google.
The remaining four boards had no information relating to their formularies and had no information available regarding ADTC decisions. When conducting a search, using the search terms detailed in Appendix 2, none of these boards provided a link to the their formularies, and in the case of two boards, did not have a search function on their websites.
Contact
Email: VERONICA MOFFAT