The microsegmentation of the autism spectrum: research project
Economic research on autism and implications for Scotland, including how the economic cost of autism can inform strategy and planning.
4 Study 1: The Prevalence Of Autism Spectrum Disorders
Introduction
4.1 An accurate estimate of prevalence is crucial to any economic analysis of autism, and it is the principal factor on which the earlier studies by Knapp and colleagues have depended. It was therefore a fundamental requirement of this study that it should analyse the basis on which the figures of Knapp and colleagues were derived, provide the most reliable figures possible and apply these figures to the Scottish context.
4.2 Ultimately, the question of the economic cost of autism is a question of numbers, and even a small variation in the figure selected can have enormous economic significance. It is for this reason that the most accurate prevalence figures are essential. The figure of one percent used by Knapp and colleagues in their later study (Knapp et al., 2009) was slightly lower than the figures indicated in the study by Baird and her colleagues in one of the most widely cited prevalence studies (Baird et al., 2006), but still somewhat higher than in other prevalence studies. If the slightly higher figure of 1.16 percent had been used as in the study by Baird and her colleagues, the total annual cost UK would have risen by around £5 billion. If, on the other hand, a lower figure of 0.7 percent had been used (see, for example, the review by Fombonne, 2009), the annual cost would have been reduced by around £8 billion.
4.3 However, there are two key problems here. First, the prevalence of ASD in Scotland is not known. Second, the even more fundamental question of the general prevalence of ASD in terms of the worldwide literature cannot be relied upon.
4.4 Regarding the prevalence of ASD in Scotland there are no reliable figures. The reason for this – and it is a reality that affects prevalence studies in general – is that it is a vast exercise both practically and economically to carry out a robust population study. Studies which rely on clinical samples of cases already identified or on figures generated from official records have no reliability for academic purposes. They only answer questions relating to how many individuals have been identified in any given area, and that is highly dependent on the nature and extent of diagnostic services and on the artefacts of local record-keeping practices.
4.5 This may be illustrated by reference to three data sources relevant to Scotland at national level. First, a study of educational provision for children with autism in Scotland published in 1996 (of which the first author of the current report was chair of the Government’s steering group) (Jordan & Jones, 1996) arose from a research proposal in which a key issue was to establish prevalence figures. For practical and economic reasons this could only be done by gathering data across the country for identified cases. The total number of cases identified suggested a prevalence rate of just over 6/10,000, or about one tenth even of the relatively modest 60/10,000 being cited as the best available prevalence figure at that time. This was in face of the fact that a very inclusive approach was taken in which cases were included if ASD was suspected even if it had not been diagnosed.
4.6 A second effort to establish Scottish prevalence on a case study basis was made in a Government audit in 2004 (Scottish Executive, 2004). This was based on gathering information from all Health Board areas in Scotland. Again it produced unrealistically low prevalence rates of 35/10,000 for children and 2/10,000 for adults.
4.7 The third potential data source is the Scottish Census 2011. While it was seen as a significant step forward that a question relating to autism was inserted in the census, it was not possible for those campaigning for this insertion to have it relate exclusively to ASD. In the section which asks about disabilities the insertion is for ‘developmental disorder’, for which it includes the examples: ‘eg Autism Spectrum Disorder or Asperger’s Syndrome’. However, that would not meet a specification for answering a prevalence question because, first, developmental disorders are wider than ASD and indeed the way they are defined in the diagnostic classifications is significantly wider and, second, the question relies totally on self-report at the simplest level (a tick in a single check box). Regarding self-report at this level there are known issues. There tends to be inclusion of those who believe they are on the spectrum on the basis of ‘self-diagnosis’, those who have been told by professionals such as teachers that they may be on the spectrum but who have had no diagnostic investigation of this and those who have been assessed and not diagnosed, but who assert that the assessment is wrong.
4.8 In addition to these national sources of prevalence data, a more systematic study was conducted at local level in Lothian by Harrison, O’Hare, Campbell, Adamson and McNeillage (2006). They found a prevalence of 33/10,000 based on children age 15 years and under known to local autism services. After adjusting this figure to estimate for other diagnosed cases not identified through these services their overall estimate was 44/10,000. Again this study had the limitations found in all referral-based estimates of prevalence in markedly under-estimating actual prevalence levels.
4.9 While it is clear that we therefore have no reliable information on ASD prevalence that is specific to the Scottish population, it was nevertheless our view that there is no sustainable argument, either economically or clinically, to support the recommendation of a Scottish prevalence study. In economic terms, the costs would be very high and would not be in accord with the overall recommendations in the Scottish Strategy for Autism. In clinical terms, a Scottish prevalence study would only be justified on one of two grounds. The first would be the availability of convincing evidence that the prevalence of autism from an international perspective does not show a relatively stable underlying pattern. The second would be the presence of any reasonable uncertainty as to whether Scottish autism prevalence might represent a special case that did not fit the underlying international pattern as identified in the best prevalence studies. It is our view that there is no such convincing evidence or reasonable uncertainty.
4.10 With regard to the relative stability of underlying patterns of autism prevalence internationally, it may be stated that there are examples of variations, and of apparent variations, in prevalence across some specific cultural contexts. For example, Barnevik-Olsson, Gillberg and Fernell (2010) studied the medical records of all children with autism in combination with intellectual disability born from 1999 to 2003 to Somali immigrants living in Stockholm. They reported prevalence some four to five times higher than for those not of Somali origin. Other than specific exceptions of this nature, the highest quality prevalence studies, when controlled for moderating factors such as age, show a general underlying pattern of prevalence which has an acceptable level of homogeneity.
4.11 With regard to autism in Scotland, we have no basis on which to view its presentation and prevalence as representing a special case. It is likely that there will be local variations for a variety of reasons. For example, there may be particular circumstances that would lead to variations in prevalence in small populations, such as island communities. However, there is nothing to indicate that the Scottish ASD population as a whole differs from the underlying pattern identified in the highest quality international prevalence studies. In addition, as reported later in this chapter, the studies selected for our meta-analysis of prevalence broadly reflected UK and Scandinavian populations, and there is no reasonable basis for asserting that Scotland represents a different case by comparison with these populations.
4.12 Regarding the more fundamental question of the general prevalence of ASD in terms of the worldwide literature, the position at first glance points to what seems like a fairly consistent pattern in which the main moderator is time of study, in relation to which, in turn, the most important factor for population studies is changing diagnostic criteria; to this can be added increased recognition of ASD and more widely available diagnosis in the case of referral-based studies. In terms of changing diagnostic criteria, the basis for a diagnosis of autism or an autism spectrum disorder expanded systematically from the early prevalence studies of Kanner’s Syndrome or classical autism in the 1960s, to the increasing recognition of autism in the context of intellectual disability, to wider acceptance of autism as a context defined behaviourally and applicable to those with other conditions such as tuberous sclerosis and Down’s Syndrome, to the Camberwell study by Wing and Gould (1979) and the emergence of what would be the autism spectrum, to the entry of Asperger’s Syndrome into the classification systems from 1992 onwards (American Psychiatric Association, 1994; World Health Organization, 1992) with its own significant prevalence rates.
4.13 This pattern was, inevitably, one of constantly increasing prevalence, with the most commonly cited studies progressing from estimates of just over 4/10,000 in the 1960s for Kanner’s autism (Lotter, 1967), to 21/10,000 in the late 1970s (Wing & Gould, 1979) for a much wider interpretation of the syndrome based on the triad of impairments, to 36/10,000 for Asperger’s Syndrome alone in the 1990s (Ehlers & Gillberg, 1993), the latter two figures commonly being combined to give an overall prevalence of about 60/10,000 for ASD in the 2000s (see, for example, Public Health Institute of Scotland, 2001). The South Thames study by Baird and her colleagues (Baird et al., 2006) provided an increasingly cited benchmark of a little over 100/10,000. That study was not in itself a reflection of changing criteria, as these had not been revised since the 1990s, but there was still no indication of any real increase in prevalence in clinical terms.
4.14 However, any examination of the clinical and methodological basis on which these figures have been reached, not only in the Baird et al. (2006) study but in other studies consistent with it, raises some fundamental issues. For example, in the Baird et al. study there were few who met the diagnostic for Asperger’s Syndrome – a syndrome for which the prevalence estimates published in peer reviewed journals are so diverse that they range from 0.3/10,000 (Sponheim & Skjeldal, 1998) to 48.4/10,000 (Kadesjo, Gillberg, & Hagberg, 1999). Some of the difficulties associated with estimating prevalence may be illustrated by the Ehlers and Gillberg (1993) study and the basis on which a prevalence figure of 36/10,000 for Asperger’s Syndrome was established. The methodology suggests that somewhat stricter inclusion criteria were probably applied than would be used in clinical practice by many diagnosticians. If we add their ‘likely’ cases to the ones they considered to be definite then it virtually doubles the prevalence figure they quoted. A further variable is the age of the population surveyed, as a pre-school population would under-represent those who would subsequently be identified in an older age-group with Asperger’s Syndrome, which is associated with a later age of diagnosis (Howlin & Asgharian, 2007).
4.15 Overall, the published studies of ASD prevalence in recent years range from 1.4/10,000 (Al-Farsi, Al-Sharbati, Al-Farsi, Al-Shafaee, & Brooks, 2011), through 264/10,000 (Kim et al., 2011) with almost every data point across that range, other than towards the extremes, represented by one or more studies. The studies at the extreme highlight many of the issues arising in prevalence studies in general, albeit in a more prominent way.
4.16 In relation to the above, the Al-Farsi et al. (2011) study was not a population study but was based on children in the Sultanate of Oman who had received a diagnosis of autism. Thus, it was totally dependent on the availability of ASD diagnostic services in an area where, as the authors noted, there was a single child psychiatry unit based in the capital and servicing the entire nation. The Kim et al. (2011) study is harder to assess as it was a large sample prevalence study which used a robust methodology. However, this is a study that derived its figures, not from the actual prevalence identified (0.36% for total ASD, 0.18% for autism and 0.18% for all other ASDs). Statistical adjustments were then made to account for non-responses and a final prevalence rate was calculated at 2.64% for total ASD, 0.95% for autism and 1.70% for all other ASDs.
4.17 In setting out the extremes of prevalence estimates we have not made reference to a study by Dillenburger, Jordan, McKerr and Keenan (2015) which estimated prevalence at 3.5%. This, however, is not a study which makes a serious contribution to the question of prevalence, as it derived its figure simply from asking parents of 11-year-old children if a doctor or health professional had ever ‘told them their child had autism/Asperger’s Syndrome’ (p.41). Reliance was placed on this self-report alone, with no proof of any diagnosis being sought.
4.18 In addition to the above, the methodology of studies which are not at the extremes but are commensurate with the results reported in this meta-analysis and with commonly held views of prevalence must also be examined rigorously to ascertain whether the contribution they make to the prevalence literature is a reliable one. For example, the prevalence studies are replete with classification issues, with studies variously covering autism, autism spectrum disorder or pervasive developmental disorder. They are very diverse in terms of their methodology, of the types of sample used and of diagnostic practice in the areas or the countries where the studies were conducted.
Method
4.19 Our study procedure was as follows. The online journal databases 'Medline', 'PsycInfo' and 'PsycArticles' were searched for English-language, peer-reviewed papers, published since 31 December 2002 which investigated, or commented upon, the prevalence of ASD. The search terms are summarised in Table 4.1 and the results of this search are summarised in Figure 4.1. The initial search (Stage 1) returned 40,713 results (598 from Medline and 40,115 from PsycInfo, with no unique articles identified from PsycArticles). However, the majority of these (n = 40,648) were removed from further analysis as they did not specifically measure the prevalence of ASD or PDD, or were review studies which did not report primary data (Stage 2). The literature reviews and reference lists of the remaining 65 papers were searched for mentions of previously unidentified studies, adding a further 27 studies, resulting in a total of 92 papers at this stage (Stage 3).
Table 4.1 Summary of search strategies
| Journal Database | Search Terms | Search Location | ||
|---|---|---|---|---|
| MedLine | “MESH.EXACT.EXPLODE (“Child Development Disorders, Pervasive”)” * | AND | “prevalence” OR “epidemiology” |
Abstract |
| PsycInfo | “Pervasive Developmental Disorder” ** | “prevalence” OR “epidemiology” |
Abstract | |
| PsycArticles | “Pervasive Developmental Disorder” ** | “prevalence” OR “epidemiology” |
Abstract | |
Notes: * This is a composite term which when used will return papers including the terms Autistic Disorder, Asperger’s Syndrome and Autism Spectrum Disorder, as well as any variations/alternatives to these terms, and any terms which are more broadly related (e.g. ‘repetitive behaviour’). ** In terms of the type of papers it returns, this term is almost identical to that used for the Medline search.
Figure 4.1 Flowchart of prevalence study selection process
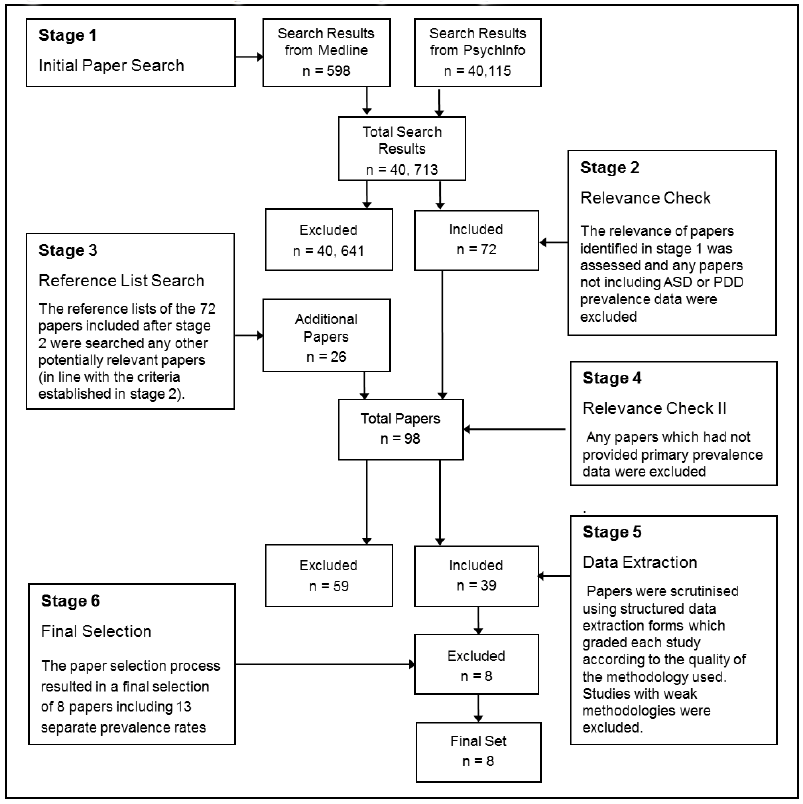
4.20 All 92 papers were subject to a full appraisal for relevance. Any paper not reporting useful prevalence information (including reviews not reporting any primary data) was removed from the analysis. Papers with serious methodological flaws, including those which based prevalence rates upon clinically unconfirmed diagnoses or unrepresentative populations, such as the study by Mandell et al. (2012), which drew its sample from a state psychiatric hospital, were also removed. In total, 57 papers were removed from the analysis at this stage (Stage 4). The justification for the removal of each is detailed in Appendix A.1.
4.21 The remaining 35 papers varied in the method they used to arrive at their prevalence estimate. To ensure that these methodological differences had no bearing on our findings the methodology of each paper was scrutinised using an 11-point data extraction form developed by the researchers (Stage 5). The content of the data extraction form was informed by the SIGN guidelines on ASD diagnosis (Scottish Intercollegiate Guidelines Network, 2016) and also by Stage 2 of the literature review, throughout which the authors adjusted the content of the form according to the variance in methodology and overall quality observed within the papers analysed. Quality assessments of the studies were based on the grading of five key points on the data extraction form which concerned the level of detail studies had provided about the population that had informed the prevalence estimate, the diagnostic criteria used, the tools and professionals involved in diagnosis and the overall quality of the methodology followed. Those not meeting the pre-determined level of quality in relation to each of these factors were subsequently removed. The data extraction form and the grading criteria used to assess these aspects of a study’s methodology have been included in Appendix A.2.
4.22 A cross-validation process was used to ensure reliability of coding in the data extraction process. Following training on a random set of six papers from the 35 identified as meeting the criteria for a detailed Stage 4 quality assessment, two of the researchers independently coded a further random sample of 6 papers (17% of the total). There was 83% overall agreement (five out of six papers) about whether the paper should be included or excluded from further analysis and the correlation between the independently coded quality of evidence scores (score range 0-20) for these 6 papers was 0.90 (with Means ( SD) of 14.83 (4.07) and 14.83 (3.71) for Coders 1 and 2 respectively). Disagreement on the remaining paper was resolved through discussion.
4.23 A further 27 papers were excluded at this final stage (see Appendix A.3). Eight papers in total were selected for final inclusion in the meta-analysis as they met all of the criteria. These were further assessed using a refined quality of evidence score. This was on a scale of 0-10, and in addition to previous criteria regarding sample size, diagnostic criteria used, and nature of the diagnostic process we also assessed quality of recruitment strategy (for example, whether whole population, stratified sample, or high quality record
Table 4.2 Summary of final set of prevalence studies
| Study | Population sampled Target / screened | Age | Prevalence Per 10,000 (95% CI) | Comments |
|---|---|---|---|---|
| <6 years Chakrabarti & Fombonne (2005) | 10,903/10, 903 |
4-6 |
22 (14.1-32.7) |
Analysis of a high quality surveillance system in the English Midlands. |
| Idring et al. (2012a) | 589,114 | 4-6 | 65 (59-71) | High quality record review covering over 99% of the Stockholm population. Prevalence estimates reported separately for four age groups. |
| Nygren et al. (2012a) | 4,871/ 4,871 | 2-3 | 80 (57-109) | Estimates of ASD rates in Gothenberg over a 10-year period for 3 different groups. 2012a and b refer to a 2000 cohort; 2012c to a 2010 cohort. |
| Nygren et al. (2012b) | 5,220/5,220 | 2-3 | 4 (1-14) | |
| Nygren et al. (2012c) | 6,220/5,007 | 2-3 | 18 (8-35) | |
| 6-12 years Baird et al. (2006) | 56,946/1,170 |
9-10 |
116.1 (90.4-141.8) |
From SNAP special needs birth cohort in London |
| Baron Cohen et al. (2009) | 11,700/ 3,373 | 5-9 | 94 (75-116) | ASD prevalence (unadjusted raw point estimate with no weighting for non-responses) in mainstream and special schools in Cambridgeshire. |
| Mattila et al. (2011) | 5,484/4,414 | 8 | 84 (61-115) | School based study of 80% of all 8 year old children in Finland. |
| Idring et al. (2012b) | 589,114 | 7-12 | 120 (114-126) | High quality review of records covering over 99% of the Stockholm population. Prevalence estimates reported separately for four age groups. |
| > 12 years Brugha et al. (2011) | 14,532/ 7,403 |
16+ |
98 (30-165) |
Data from the English National Adult Psychiatric Morbidity Survey. |
| Idring et al. (2012c) | 589,114 | 13-17 | 146 (140-153) | High quality record review covering over 99% of the Stockholm population. Prevalence estimates reported separately for four age groups. |
| Idring et al. (2012d) | 589,114 | 18-23 | 105 (99-110) | |
| Kočovská et al. (2012) | 7,128/ 7,128 | 15-24 | 94 (73-119) | Follow-up of young adults in the Faroe Islands. |
4.24 review), and participation level. Final coding of the elements of these 10 papers for inclusion in the meta-analysis was carried out jointly by both coders to ensure uniformity in final data extraction.
4.25 These papers included prevalence estimates relating to 13 different samples (described in Table 4.2) drawn from the final eight studies. The majority of these estimates (n = 10) were based upon population studies; that is, they screened an entire population (for example, individuals aged 15 to 24 living in the Faroe Islands) before individually diagnosing those identified as more at risk. The study by Idring et al. (2012), which contributed three estimates to the final set, was the only study to base its prevalence figures on the results of a record review of medical records (that is, the investigators were not specifically involved in any of the diagnoses to which their figures related). However, the detail this paper provided about the diagnostic process and the manner in which cases in their target area were identified (through a surveillance system covering over 99% of the population) meant that it was very comparable to the other studies in this final set and met the required inclusion criteria.
4.26 The investigations were carried out in four countries, with seven of the estimates relating to a Swedish sample, four to English samples, and the final two to samples from the Faroe Islands and Finland. The size of the samples targeted ranged between 4,871 and 56,946 (m = 13667.11, SD = 16580.21), though the number of individuals actually screened ranged between 4,414 and 10, 903 (m = 5498.78, SD = 2753.03). The studies covered individuals from pre-school to young adulthood. Of the 13 prevalence estimates, five related to children under the age of six years (coded as a pre-school sub-group), four related primarily to participants aged between six and 12 years (coded as a primary school-age sub-group), and a further four to children and young people aged 12 years and above (a post-primary school-age sub-group). The mean quality assessment scores for the 13 datasets from the 10 studies included in the analysis used to rate quality of evidence ranged from 6-10, with a mean of 7.46 ( SD 1.45) and a median of 7. Studies were further graded as ‘2’ (above the median), or ‘1’ (below the median) for analysis of the effects of quality of evidence.
4.27 The studies obtained their samples from a series of different sources: five studies, including the review of records by Idring et al. (2012), obtained them from hospitals, three from schools, one from information acquired as part of a national mental health survey and one from a previously constructed special needs sample. All studies, with the exception of Idring et al. (2012), included a screening stage in their investigation. However, Chakrabarti and Fombonne (2005) screened their population by clinical interview while the remaining studies used an established screening measure with three using the ASSQ, three using M-CHAT, and the others using the AQ, the SCQ or the CAST. Again with the exception of Idring et al. (2012), all studies used either the ADI or the ADOS (generally considered the highest quality interview and assessment tools in the autism literature) in making their final diagnosis. In total, nine used the ADOS, four used ADI or ADI-R and four used DISCO to support the ADI or ADOS.
4.28 The diagnostic criteria used in the studies varied: two studies used ICD-10, five used DSM-IV/ DSM-IV-TR, one used a combination of ICD-10 and DSM-IV, one used the same combination to confirm old diagnoses using earlier versions of these criteria and one used ADOS-4 scores (which can be related to DSM-IV criteria). Though all studies included ASD prevalence estimates, some reported a breakdown of the prevalence estimates associated with the individual conditions: six provided estimates for childhood autism/autistic disorder, two for Asperger’s Syndrome/Asperger’s Disorder and three for atypical autism.
Results
4.29 Raw estimates of prevalence of ASD per 10,000 and the standard errors from each of the 13 datasets from the 10 included studies were entered into the Comprehensive Meta-Analysis v. 3.3.070 software. Meta-analyses were conducted on weighted logit-transformed prevalence estimates with age-group added as a between-group variable. There were no missing data.
4.30 Meta-analysis may be carried out using ‘fixed effects’ or ‘random effects’ models (Borenstein, Hedges, Higgins, & Rothstein, 2009), as well as ‘mixed effects’ models which combine fixed and random effects in one analysis. Fixed effects models assume that studies are sampled from a single population, with one source of error, ‘within-studies’ sampling error, and that there is an underlying ‘true’ effect size for all of the studies. In contrast, random effects models assume that studies are randomly sampled from a ‘universe’ of within-studies variance. Thus, instead of assuming one underlying ‘true’ effect size, random effects models assume a distribution of such ‘true’ effect sizes. As a result, random effects models have two sources of error: ‘within-studies’ sampling error and ‘between-studies’ sampling error, which is an estimate of the population variance (Borenstein et al., 2009). Random effects models thus yield pooled estimates with larger confidence intervals due to the additional source of error, but estimates which are more warranted when comparing data from studies carried out by different investigators. (Borenstein et al., 2009). We carried out random effects meta-analyses using the method of moments ( MM) (DerSimonian & Laird, 1986), an approach which makes no assumptions regarding the distribution of effects.
4.31 Table 4.3 provides a summary of the random effects meta-analysis of the prevalence estimates of ASD from the 13 data sets reported in the 10 included studies, with their associated forest plots – a plot of the point estimate of prevalence with a 95% confidence interval, which shows the level of
Table 4.3 Summary of random-effects meta-analysis of prevalence estimates from 13 ASD samples included in 8 studies
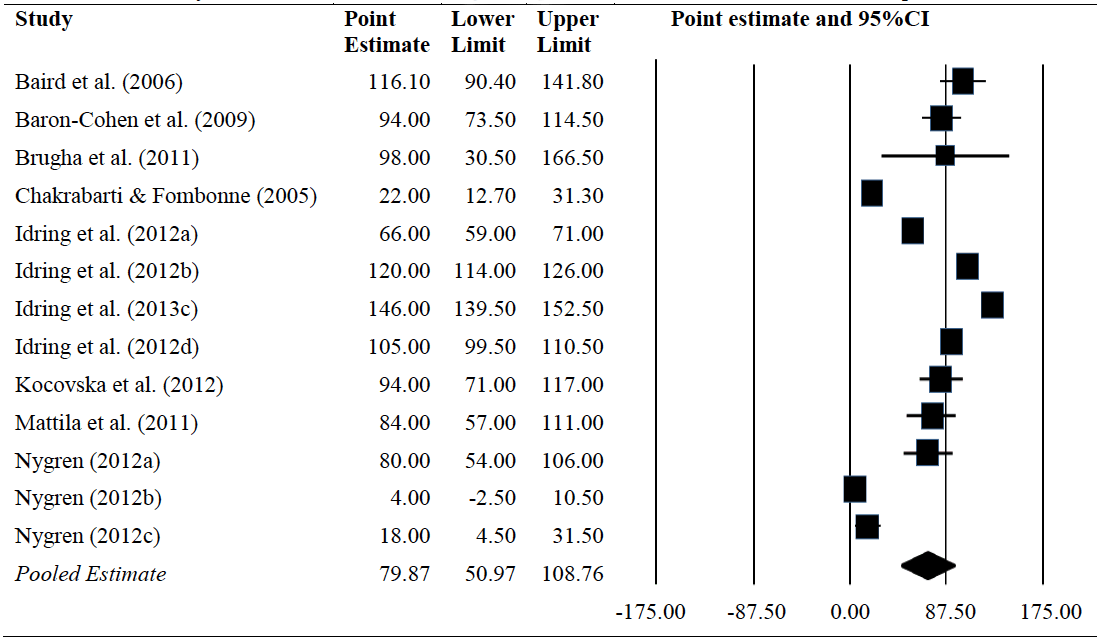
variability in the estimate. We note that in five cases the 95% CI based upon the standard error in the figure differs from those reported in the original papers. This is due to the fact that the authors of the papers reported asymmetric CI, which can arise as an artefact of the use of log/antilog transformations in their analyses. Our use here of point estimates of prevalence and their standard errors allows these studies to be included in the meta-analysis. The random effects model meta-analysis reveals an overall pooled prevalence estimate of 79.87 per 10,000 (95% CI 50.97 - 108.76). However, there were highly significant levels of heterogeneity (Q = 1,433.429, df=12, p=.000, I 2=99.16%, tau 2=2,689.96) indicating that the point prevalence estimates were not all from the same type of population.
4.32 To explore this, a funnel plot of prevalence estimate by standard error is shown in Figure 4.2. The funnel plot is used here to provide information about statistical outliers which contribute to the heterogeneity that has to be explained or accounted for in the meta-analysis. The funnel plot here consists of a graph of the point estimate of prevalence in the X axis plotted against the standard error of the prevalence estimate (which reflects study size) with the 95% confidence intervals ( CI) shown (Borenstein et al., 2009). Estimates outwith the 95% CI indicate possible statistical outliers.
Figure 4.2 Funnel plot of standard error by point estimate of prevalence of ASD from a random effects model showing 95% confidence intervals
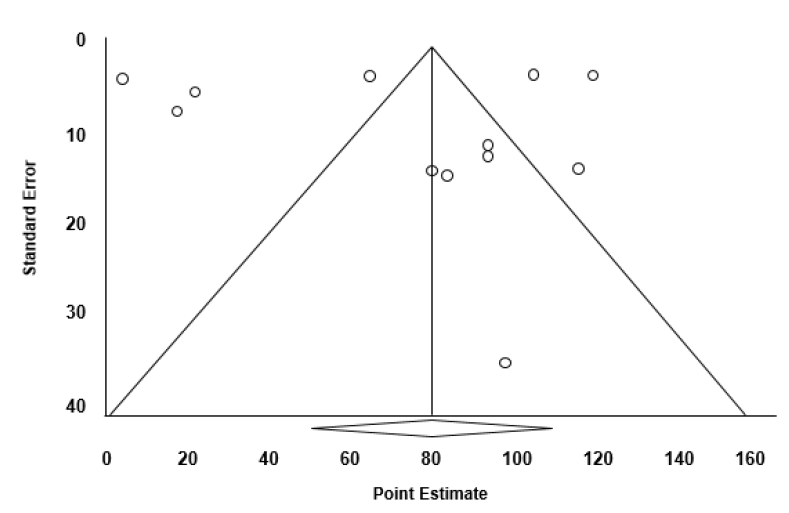
4.33 The funnel plot revealed considerable heterogeneity, that is, variability to be accounted for, with seven studies outwith the 95% CI. We investigated the observed heterogeneity using a mixed effects analysis (Borenstein et al., 2009), with age as a between-group (independent) variable, as shown in Table 4.4. In a mixed effects analysis, we use a random effects model to compute the average effect size for each sub-group within an independent variable, and a fixed effect model to compute the overall effect size across sub-groups.
4.34 Age groups were unevenly distributed across the date-sets, which was problematic for the use of age as a continuous variable in a meta-regression. Studies thus were categorised in terms of their focus on either pre-school participants (i.e. < 6 years), primary-school age participants (i.e. 6-12 years), or post-primary school age (i.e. > 12 years). Five of the data sets related to pre-school participants, and 4 each to primary and secondary school/post school-age participants. The mixed effects sub-group analysis taking age into account (see Table 4.4) showed a significant effect of age-group upon prevalence (Q between groups = 16.36, 2 df, p < .0001), with a pooled estimate for the < 6 years age-group of 36.66 per 10,000 (95% CI 9.72 – 63.59) compared with 104.16 per 10,000 (95% CI 73.02 – 135.31) and 113.54 per 10,000 (95% CI 81.14 – 145.93) for the 6-12 years and > 12 years age-groups respectively. I 2 values (the percentage of observed between-study variance which cannot be accounted for by sampling error) and the τ 2 measure of between-study variance (used to compute the weights for the random effects model) are also reported in Table 4.4, together with the Q-values for heterogeneity, which are calculated from a fixed effect analysis. These all reveal marked heterogeneity in the prevalence estimates for all three age-groups.
Table 4.4 Summary of random-effects meta-analysis of prevalence estimates from 13 ASD samples included in 8 studies, with age group as a between-group variable
| |
|
Effect size and 95% CI | Heterogeneity | |
|||||
|---|---|---|---|---|---|---|---|---|---|
| Group | No. of Studies | Point Estimate per 10,000 | Lower limit | Upper limit | Q-value | df (Q) | P-value | I 2 | τ 2 |
| Fixed effect analysis | |
||||||||
| < 6 years | 5 | 33.89 | 30.11 | 37.67 | 208.23 | 4 | .000 | 98.08 | 1094.45 |
| 6-12 years | 4 | 116.46 | 110.95 | 121.96 | 11.50 | 3 | .009 | 73.91 | 235.55 |
| > 12 years | 4 | 121.12 | 116.99 | 125.24 | 95.09 | 3 | .000 | 96.84 | 781.26 |
| Total within | |
|
|
|
314.82 | 10 | .000 | |
|
| Total between | |
|
|
|
1118.61 | 2 | .000 | |
|
| Overall | 13 | 82.46 | 79.97 | 84.94 | 1433.43 | 12 | .000 | 99.16 | 2689.96 |
| Random effects analysis | |
||||||||
| < 6 years | 5 | 36.66 | 9.72 | 63.59 | |
|
|
|
|
| 6-12 years | 4 | 104.16 | 73.02 | 135.31 | |
|
|
|
|
| > 12 years | 4 | 113.54 | 81.14 | 145.93 | |
|
|
|
|
| Total between | |
|
|
|
16.36 | 2 | .000 | |
|
| Overall | 13 | 79.15 | 61.90 | 96.39 | |
|
|
|
|
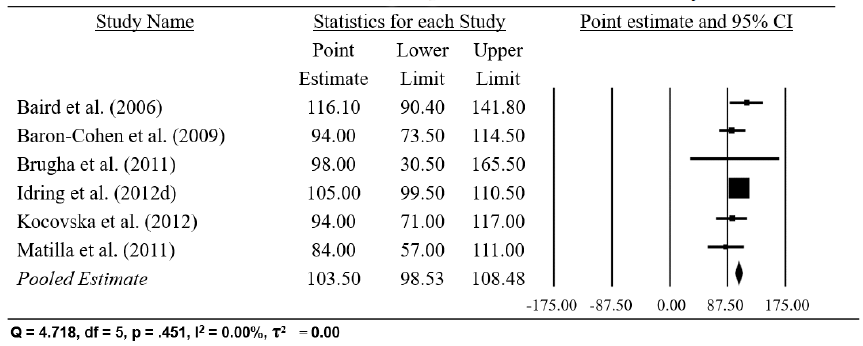
4.35 An overall pooled prevalence estimate of 109.83 per 10,000 (95% CI 93.88 – 125.77) was observed from an analysis of the combined > 6 years data sets (n = 8). This removed five of the outliers identified by the funnel plot. Three of the studies of the children aged > 6 years had quality assessment scores of > 7 (i.e. above the median), and 5 had scores below the median. There was no significant effect of study quality upon the prevalence estimates from this combined data set (Q between groups = 0.72, 1 df, p = .399, n.s.). A further analysis revealed no significant effect of age-group (6-12 years versus > 12 years) (Q between groups = 0.24, 1 df, p = .626, n.s.). However, it should be noted that as before there was evidence of marked heterogeneity of prevalence estimates in this dataset (I 2 = 93.45, τ 2 = 403.72). Sensitivity analysis (Borenstein et al., 2009) revealed the presence of two outliers indicating possible sampling error, the Idring (2012b) and (2012c) data sets, large-scale data sets with prevalence of 120 and 140 per 10,000 respectively, compared with the lower prevalence of 105 per 10,000 from the older age-group in the Idring (2012d) data set. A re-analysis with these two Idring data sets removed yielded a prevalence estimate of 103.50 per 10,000 (95% CI 98.53 – 108.48), with no significant heterogeneity (I 2 = 0, τ 2 = 0), as shown in Table 4.5. A parametric Maximum Likelihood model ( ML) (Kelley & Kelley, 2012) yielded identical estimates.
Table 4.5 Final random effects meta-analysis prevalence estimates for six years and above
4.36 We carried out sensitivity analyses to explore further this estimate and its related CI. Diagnostic checks on the model revealed that the Idring (2012d) study was highly ‘influential’ (Cook’s Distance = 6.99; DF Fits = 2.85). This means that although in this case the study is not an outlier, its exclusion would lead to changes in the model. We note that the fact that a study is influential does not mean in itself that it is invalid. Rather, it indicates that it requires further examination (Viechtbauer & Cheung, 2010). We note further that Idring (2012d) is a population study, with a much larger sample size than the other included studies, and is weighted accordingly in the meta-analysis. We would thus expect it to be influential.
4.37 Fitting a parametric Restricted Maximum Likelihood ( REML) random effects model (which assumes a normal distribution of the random effects and results in a less biased, but less precise estimate than an ML model) substantially reduced the influence of the Idring study (Cook’s Distance = 2.20; DF Fits = 1.51), yielding a prevalence rate estimate for the 6 years and above age-range of 102.20 per 10,000 (95% CI 95.11 – 109.30, Q = 4.72, df =5, p=.451, n.s., I 2 = 0.00%, τ 2 = 12.05) which is of the same order of magnitude as that from the more robust non-parametric MM model and indeed, the ML model.
4.38 Finally, we carried out a sensitivity analysis comparing the pooled prevalence rate for six years and above from the three UK studies with that from the three other countries in Table 4.5 using a non-parametric MM random effects model. The findings revealed no significant difference (Qbetween groups = 0.02, 1 df, p = .886, n.s.), indicating that no effect upon the overall pooled prevalence rate of any differences in clinical guidelines or perception of autistic people between the UK and the three other countries included in the analysis. The small number of studies should again be noted.
Discussion
4.39 The results from the meta-analyses revealed that significantly lower prevalence estimates were observed in the case of studies focussing upon children aged below six years. This is to be expected. Howlin and Asgharian (1999), in noting the later age at which more able children with ASD are diagnosed, highlighted the fact that it is much more difficult to receive an early diagnosis when there are no delays in language and cognition and other difficulties may be relatively subtle. Studies of older children thus provide the most accurate estimates and a more suitable basis for planning services. We note that there was no significant effect of age upon prevalence estimates from the 6-12 years and 13-24 years age groups in the studies included here, permitting the combination of these two age groups to provide a pooled estimate based upon 23,488 children and young people screened.
4.40 Prevalence studies of autism spectrum disorders are marked by very significant levels of variability in the estimates they have proposed. This meta-analysis has sought to establish a reliable prevalence estimate for ASD using a rigorous selection procedure in which the methodology of all relevant studies has been interrogated in detail. Our final figure for the population aged six years and above was 103.50/10,000, with a 95% confidence limit of 98.53/10,000 to 108.48/10,000. We propose, therefore, that the most reliable prevalence estimate for ASD is 1.04% (95% CI 0.99%-0.108%). This figure will be recognisable as being within the range of figures which currently have come to be most generally accepted as a basis for resource planning, aggregate cost estimates and other purposes (see, for example, Buescher et al., 2014; Knapp et al., 2009).
4.41 We would treat with caution the view that the ‘true’ prevalence of ASD varies internationally from one country or area to another. We reviewed very many studies indicating low rates in a wide range of countries, but in all cases we found that this arose either from sampling only known cases, often reflecting limited diagnostic provision, or from other artefacts or inadequacies of methodology. Likewise, we treat with considerable caution the finding by Kim et al. (2011) of atypically high rates of ASD in a South Korean community (2.64%), a study which we excluded on the basis of methodological issues relating to sampling and level of participation. At the same time we note the series of reports regarding a higher prevalence of autism in association with intellectual disability among children of Somali origin in Sweden, reported at 0.98% compared with 0.21% for those of non-Somali origin (see Barnevik-Olsson, Gillberg, & Fernell, 2010). Overall, however, we have not found evidence in general in the studies we have reviewed for differential geographical prevalence of ASD.
4.42 We were unable to establish prevalence estimates for diagnostic sub-groups within the autism spectrum. While a number of studies reported data separately for autism or for Asperger’s Syndrome this information was not of the quality or extent to allow meta-analysis. A particularly difficult issue arises in relation to atypical autism, which did not feature in some studies but accounted for a high proportion of cases in other studies. Approaches to this sub-group have varied widely among diagnosticians. While some have used the category sparingly within the original spirit of ICD-10 for presentations of autism occurring ‘most often in profoundly retarded individuals’ with ‘very low level of functioning’ (World Health Organization 1992, p.255), others have treated it as a catch-all for a wide diversity of cases with sub-threshold symptomatology – ‘not, or not quite, autism’ (Klin, Volkmar, & Sparrow, 2000). The matter of prevalence within sub-groups will, of course, be overtaken, following these being subsumed under autism spectrum disorder in DSM 5 in 2013 (American Psychiatric Association, 2013), with a similar approach being taken in the current Beta Draft of ICD-11 (World Health Organization, 2016).
4.43 There were several limitations to this study. First, we limited our search to three databases. It is possible that relevant studies may not have been included in these databases. However, it is unlikely that any study meeting the selection criteria of this study would not have appeared in these databases among the more than 40,000 articles screened in the initial trawl. In addition, we searched the literature reviews and reference lists of all relevant review papers we identified. Second, our search was limited to English language, peer reviewed papers. It is possible that relevant work has been published in other languages, or that usable data would have been found in non-peer reviewed sources such as Master’s or Doctoral theses. Third, our final analysis included only studies from England, Sweden, Finland and Faroe Islands. Nevertheless, as noted, we found no convincing evidence in the course of our investigation to support the view of systematic geographical variations in terms of prevalence, other than allowing for the possibility of very specific exceptions. Fourth, we operated very rigorous selection criteria in terms of study methodology. While this had the benefit of ensuring only very high quality studies in the final analysis, it is possible that some excluded studies may have contained relevant data. Fifth, by using such rigorous selection methods we found only a small number of studies left for our meta-analysis, having 13 samples derived from eight studies.
4.44 It would have been possible in this study to have taken a less exacting approach to selection, and to have included many of the 27 studies we excluded at the final stage of selection. However, this would have resulted in figures on which we would have been less able to rely. The studies we analysed met the strictest standards in terms of diagnostic criteria, diagnostic procedures, sample size and representativeness, statistical analysis, and all other relevant aspects of methodology. In terms of representativeness, for example, the three samples from Idring et al. (2012) covering the age range above six years comprised 99.8% of the population of Sweden, a country with a universal system for surveillance and screening for ASD and with well-established protocols for diagnosis and for maintenance of comprehensive records. We trust that the results of this meta-analysis will provide researchers, service providers and economic planners with a confident basis within which to view the prevalence of autism spectrum disorders.