Maternity and neonatal (perinatal) adverse event review process: guidance
Operational guidance to support health boards boards undertaking perinatal adverse event reviews incorporating the additional reporting required of maternity services.
Appendix B: National notification system for SAER
National Notification System for Category 1 Events
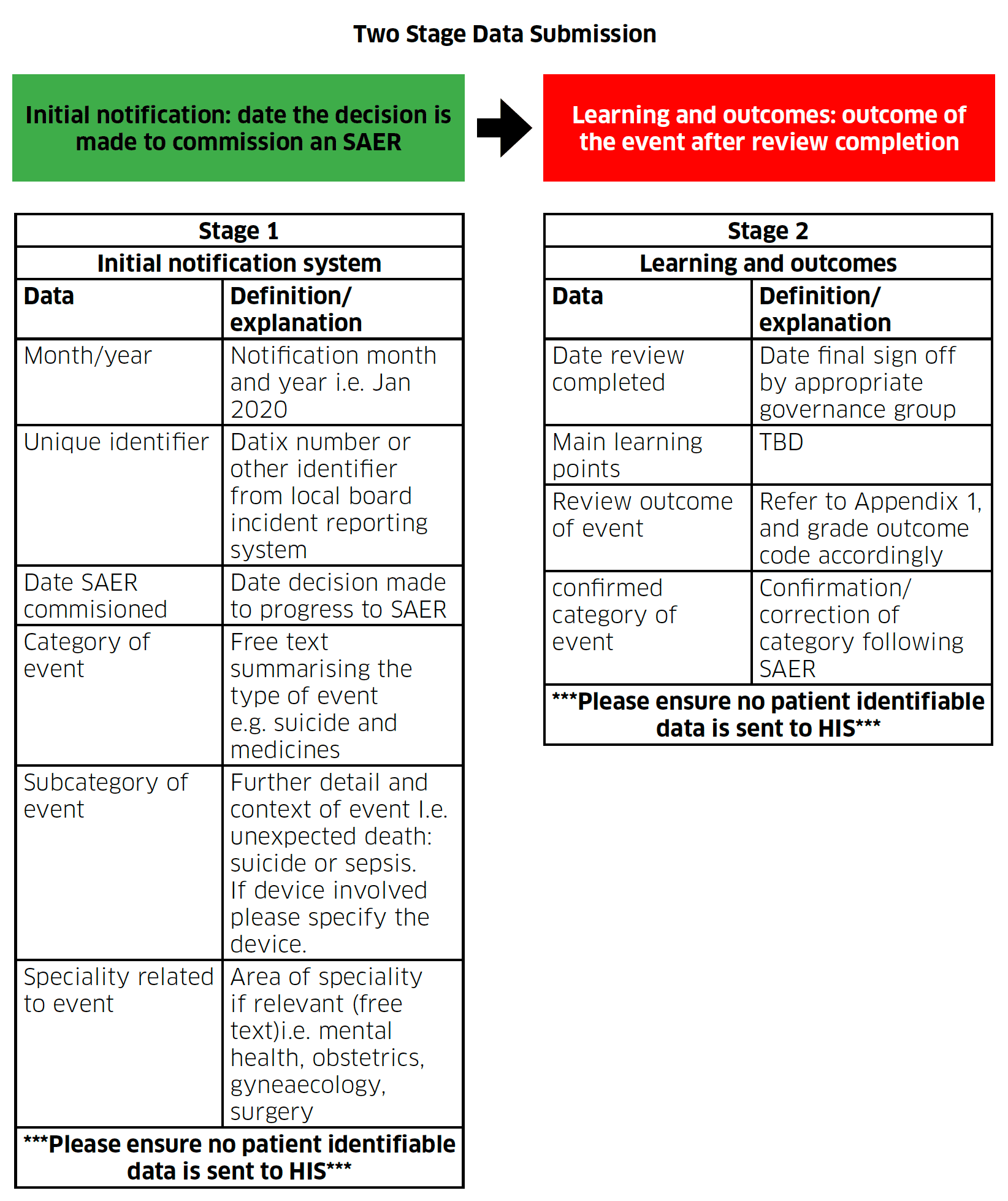
Infographic text below:
Two Stage Data Submission
Initial notification: date the decision is made to commission an SAER
| Stage 1 | |
Initial notification system |
|
Data |
Definition/explanation |
Month/year |
Notification month and year i.e. Jan 2020 |
Unique identifier |
Datix number or other identifier from local board incident reporting system |
Date SAER commisioned |
Date decision made to progress to SAER |
Category of event |
Free text summarising the type of event e.g. suicide and medicines |
Subcategory of event |
Further detail and context of event I.e. unexpected death: suicide or sepsis. If device involved please specify the device. |
Speciality related to event |
Area of speciality if relevant (free text)i.e. mental health, obstetrics, gyneaecology, surgery |
***Please ensure no patient identifiable data is sent to HIS*** |
|
Learning and outcomes: outcome of the event after review completion
Stage 2 |
|
Learning and outcomes |
|
Data |
Definition/explanation |
Date review completed |
Date final sign off by appropriate governance group |
Main learning points |
TBD |
Review outcome of event |
Refer to Appendix 1, and grade outcome code accordingly |
confirmed category of event |
Confirmation/correction of category following SAER |
***Please ensure no patient identifiable data is sent to HIS*** |
|
Contact
Email: thebeststart@gov.scot