Scottish Marine and Freshwater Science Volume 3 Number 3: Clyde Ecosystem Review
A review of previously published information and knowledge concerning the Firth of Clyde ecosystem and suggestions for further studies in the Clyde required in order to support sustainable use of this active Scottish marine ecosystem
4. BIODIVERSITY
The Clyde Sea area has a wide range of habitats which support a large diversity of plant and animal species. There is some concern that human activities may have already had effects on those species and habitats, through overexploitation, habitat destruction and environmental change. Historically the Clyde has supported large populations of herring and whitefish, which have in recent years undergone a dramatic decline, and commercial fishing is now largely dependent on shellfish species. The fauna of the Clyde does show some anomalies when compared with that of the west coast, particularly the Clyde algal fauna, with some species only found in the Clyde (Maggs, 1986). An extensive amount of marine faunal lists have been published for the Clyde Sea over the past century from general accounts by Chumley (1918) to more specific accounts of specific groups, Polychaeta (Clark, 1960), Mollusca (Allen, 1962), fish (Bagenal, 1965), Amphipoda (Moore, 1984) and Echinodermata (Wilkie, 1989). The seas of the Clyde also support populations of megafauna such as seals, dolphins, whales and basking sharks. Many of theses species and habitats of the Clyde are recognised as being of national or international importance and have been afforded some level of protection under legislation to protect marine biodiversity.
Figure 4.1 Location of species hotspots (shown in red) in the Clyde Sea Area identified by Langmead et al. (2008). The five areas were Northern Loch Fyne ( HS2) and Loch Shira ( HS1); Irvine Bay ( HS8-11); East of Dunoon in the upper Firth of Clyde ( HS4); East of Rothesay, Bute ( HS7); and The Kyles of Bute ( HS6) and Loch Striven ( HS3 and 5).
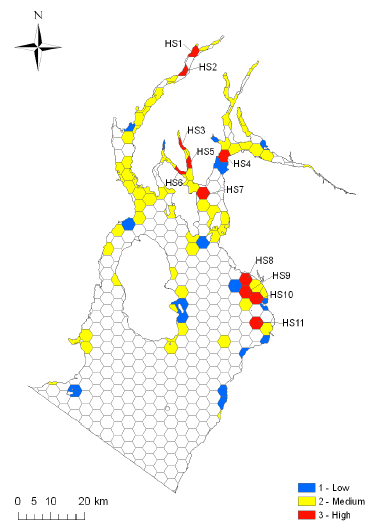
A recent review of the biodiversity of the Firth of Clyde (Langmead et al., 2008) identified and collated c.133,000 species and habitat data records for the area. The review also analysed patterns of biodiversity and determined the location of 'hotspots' defined as areas of high species and habitat richness that include representative, rare and threatened features (Ross et al., 2009). The locations of the species hotspots identified by this study are shown in Figure 4.1. It is the purpose of the following section to briefly describe the rich biodiversity of the area, discuss current threats and to describe some of the conservation and management measures that are already in place to protect biodiversity in the Clyde.
In general, the Firth of Clyde benthic populations have been given selective attention according to the habitat type, location and research interests in relation to specific environmental problems. As a result much of the work on the benthic fauna of the Clyde has focussed on the shallower coastal sediments of the rocky shores and sandy bays. A widespread littoral survey was undertaken in 1979 by Paisley College of Technology who examined 82 sites of both rocky and sedimentary types of the lochs and open coasts of the Firth (Connor, 1991). The survey suggested that the richest sediment shores were concentrated in the more sheltered parts of the Clyde such as Loch Fyne, Striven and Gareloch. The shores on the more exposed coasts of Ayrshire, the Kintyre peninsula and the coast of Arran tend to have fewer individuals and less species diversity. The intertidal infauna is dominated by the bivalves Angulus tenuis and Cerastoderma edule, the amphipod bathyporeia sp. and polychaetes Scoloplos armiger, Pygospio elegans, Arenicola marina and N. cirrosa. The intertidal fauna of sandy beaches in the Clyde have also been studied by Eleftheriou and McIntyre (1976) examining shores which are moderately exposed or sheltered. The exposed shores were dominated by crustaceans and polychaetes and the bivalve Angulus tenuis. On the sheltered beaches crustaceans were generally less common and bivalves represented the greatest biomass. Areas of particularly high benthic infaunal biomass are concentrated in Upper Loch Long, Loch Striven, the Kyles of Bute, the central Kilbrannan Sound and the Irvine and Ayr Bays.
The benthos of the deeper sediments has also been investigated centred on the influence of the Garroch Head dumping site. Life in the deep muds of the Clyde is mainly buried in the soft, fine sediment. The most conspicuous species of the Clyde deep muds are the heart urchin Brissopsis lyrifera, the brittle star Amphiura chiajei, the bivalves' Nucula tenuis and N. sulcata, the polychaetes Glycera alba, G. rouxii and Lumbrinereis hibernica. In the shallower sandier muds A. filiformis tends to replace A. chiajei, Echinocardium cordatum becomes the dominant heart urchin and the most numerous bivalves in addition to N. tenuis are Thyasira spp., Alba spp. and Mysella bidentata. Sediments with significant admixtures of gravel in the Clyde tend to have the brittle stars Ophiothrix fragilis, Ophiopholis aculeata and Amphipholis squamata. The surfaces of the mud in the lochs of the Clyde Sea are also the habitat of the spectacular burrowing anemone, the fireworks anemone ( Pachycerianthus multiplicatus).
Wireweed ( Sargassum muticum) was first found in Loch Ryan in 2004 and since then has been recorded in a number of locations throughout the Firth of Clyde. It is a fast growing large olive-brown seaweed that competes with native seaweeds through overgrowing, shading and abrasion and can alter the ecology of the habitat. In harbours and shallow water it is considered a nuisance where large floating masses can become a hazard to vessels through the entanglement of propellers and blocking of engine cooling systems (Ashton et al., 2006).
The gaping file shell ( Limaria hians) is a small bivalve, distinguished by long orange filamentous tentacles, which has been recorded along the west coast of Scotland around Kintyre and in the Clyde Sea (Hughes et al., 2009). In a study by Hall-Spencer and Moore (2000) it was concluded that the gaping file shell has disappeared from regions where it was once common including Ayrshire, the Isle of Bute and Stravanan Bay. The decline of file shell reefs in this area was attributed, by Hall-Spencer (2006), to the destructive effects of scallop dredging. The file shell has a thin delicate shell and damaged individuals dislodged from their nests are rapidly consumed by scavengers. The file shell reefs also have ecological importance as they support a rich fauna of small invertebrates and provide attachment surfaces for algae and larger sessile organisms.
Sea pens are colonial cnidarians found on sandy and muddy sediments around the British Isle and in many Scottish sea lochs. They are typically found in areas of high salinity, highly sheltered areas where tidal streams are negligible. Some areas of the Clyde Sea may provide ideal habitats for sea pens. There are three sea pen species which occur in Scottish waters tall sea pens Funiculina quadrangularis, Virgularia mirabilis and Pennatula phosphorea. The two human activities which are most likely to affect this biotope are Nephrops trawling and organic pollution. Of the three sea pen species Funiculina quadrangularis is likely to be the most vulnerable to trawl damage due to its brittle stalk and inability to retract into the sediment. Heavy organic pollution can exclude sea pens from an area, with severe oxygen depletion probably having damaging consequence to sea pens.
Maerl beds are living sediments characterised by accumulations of unattached calcareous red algae (Corallinales, Rhodophyta), also known as rhodolith beds (Barbera et al., 2003; Steller and Foster, 1995). They occur in many environments including tropical, temperate and polar environments. In Europe they are patchily distributed occurring throughout the Mediterranean and along the Western Atlantic coast.
Maerl beds appear as patchy, poorly sorted and complex sediment that differ greatly from more uniform habitats such as mud. The interlocking of the branching thalli creates a highly complex habitat that provides numerous microhabitats for macrofaunal organisms (Steller et al., 2003). They have high ecological importance as they support a wide range of species, some of which may be unique to this habitat (Keegan, 1974). Many studies have reported rare and unusual species living in association with maerl beds, they create areas of high biodiversity and hence the beds are of international conservation significance ( BIOMAERL, 2003). In addition, maerl are slow growing and an ecologically fragile habitat considered to be a non-renewable resource. A study by Grall et al. (2006) demonstrated the high productivity of the maerl bed environment as shown by the co-dominance of endobenthic polychaetes ( Eupolymnia nebulosa, N. latericeus), bivalves ( V. verrucosa, Venerupis aureus) and Sipunculids with epibenthic crustaceans ( Melitid amphipods, P. longicornis, etc.), Molluscs (chitons or gastropods) and sponges.
Maerl beds can also be of importance to fisheries, providing nursery grounds for commercial species of fish and shellfish (Hall-Spencer et al., 2003). In the Clyde Sea area, the maerl beds coincide with many of the productive fishing grounds for scallops and infaunal bivalves and are under threat from dredging and bottom trawling in spite of their ecological and economic importance (Barbera et al., 2003). Maerl beds are known to be highly sensitive to physical disturbance and have a very low regenerative capacity and thus may require protection through appropriate conservation management strategies.
It is thought that the UK, and particularly Scotland, is home to many of the most extensive maerl beds in Europe. It occurs on exposed west coasts, such as those in Scotland, Ireland and Brittany, but it is absent from large areas of European waters, such as most of the North Sea, the Baltic, the Irish Sea and the eastern English Channel (Birkett et al., 1998). However, studies which quantify the areas covered by maerl beds in anything other than very local studies, are rare.
Maerl bed habitats in the Clyde
There have been several observations of maerl beds, both dead and living, in the Clyde. However, a first question is what habitats in the Clyde could potentially be suitable for maerl?
The physical environment needed by living maerl has been difficult to determine, as it's slow growth (~1mm per year) makes experimentation difficult.
In terms of sea bed type, Birkett et al. (1998) suggest that Maerl beds can be found in association with a range of different sediments, varying in size from fine mud to coarse gravel and pebbles.
Maerl can not withstand exposure, thus only forms below the low-water level (Birkett et al., 1998). As maerl is an algae, it requires light to grow. Thus light availability, and hence water turbidity, are parameters which will limit where maerl can live. These, in turn, will limit the depth-distribution of maerl as in clearer waters, maerl will be able to live at deeper depths compared to more turbid waters. A comprehensive study of the depth range within which maerl beds are found in the Clyde has not been performed, although Hall-Spencer (1995) suggested a range of 6-18m (Birkett et al., 1998). Maerl has not been found extensively below 30m in UK waters and is generally found shallower than 10m (Birkett et al., 1998).
A further requirement is in terms of currents near the sea bed. Maerl requires a moderate flow of water, but can not withstand intense current or wave action (Birkett et al., 1998). Therefore it will not be found in either water that has limited movement, or extreme sea bed currents from either tide or waves. This implies there are minimum and maximum sea bed current tolerances of living maerl, although these have not been quantitatively determined.
Wilson et al. (2004) suggest that maerl is not as susceptible to changes in temperature, salinity or heavy metal pollution as was previously thought. However, a critical factor was smothering by fine sediment. Thus maerl will not be found where there are natural sources of such sediment, for example near river mouths, where there is natural deposition of fine sediment, for example within persistent gyres, or where there is frequent resuspension of fine sediments by tide or wave action. Sedimentation from human activity, including trawling, dredging, sediment extraction, sewage discharges or fish farming will kill maerl (Wilson et al. 2004).
Within the Clyde studies have found living (Kamenos et al., 2004) and dead (Hall-Spencer, 1995) maerl beds, as well as living maerl beds, but heavily impacted by the effects of fishing, primarily scallop dredging (Hall-Spencer and Moore, 2000).
Surveys for maerl could include those by diver, direct remote physical methods from vessels such as grabs or towed dredges, direct visual methods from vessels such as lowered cameras or towed video, and indirect remote sensing from vessels using acoustic methods including ground determination acoustics ( e.g. RoxAnn).
Studies such as the BIOMAERL programme are advancing our overall understanding of maerl and will hopefully allow management of maerl to have a firmed scientific basis.
Seabirds are species of birds which have adapted to exploit the potential food available from the marine environment. They fall into two broad categories; those which specialise in feeding from the air picking up food from the vicinity of the sea surface, such as terns and those which dive for food such as gannets which may swim underwater in pursuit of their prey (Monaghan and Zonfrillo, 1986). However, all seabirds are dependent upon the land to breed and their distributions are governed by the constraints of this environment, where there is a safe place to nest with sufficient food resources available in the surrounding waters. Although the Firth is home to three quarters of the UKs breeding seabirds, the Clyde coast does not support a major proportion of most species of the Scottish breeding population. This is largely due to the low-lying topography of the coast providing accessibility of the Clyde coast to the large human population which reside in central Scotland (Monaghan and Zonfrillo, 1986).
The primary foods of seabirds are densely schooling lipid-rich pelagic fishes, crustaceans and cephalopods in the upper-mid water column (Hunt et al., 1996). Thus changes in the availability of pelagic fish may affect the breeding biology of these birds. Herring are especially important to the kittiwake and the decline in herring stocks may in part be responsible for the decline in kittiwakes in some areas.
Ailsa Craig, the cliffs of the Ayrshire coast and Sanda Island located off the southeast coast of Kintyre, are the most notable seabird sites of the Firth of Clyde. Ailsa Craig and Sanda Island are both designated Sites of Special Scientific Interest ( SSSIs) set up for the protection of the breeding seabirds. Fishing activity in the Clyde is known to benefit seabirds by providing an easily exploitable food source in the form of discards. Regular scavengers include the fulmar ( Fulmarus glacialis), the great black-backed gull ( Larus marinus) and the kittiwake ( Rissa tridactyla) which obtain a large proportion of their food from discards, the herring gull ( Larus argentatus), great skua ( Stercorarius skua) and gannet ( Morus bassanus) also commonly scavenge behind fishing vessels.
The basking shark ( Cetorhinus maximus) is the world's second largest fish and is regularly observed off the western British and Irish coast, mostly between April and October. It is entirely planktivorous and in Scotland basking sharks are regularly sighted around the outer Firth of Clyde where waters are plankton rich (Speedie et al., 2009). In particular the coastline of the Isle of Arran is a 'hotspot' for basking shark sightings. The spatio-temporal distribution of basking sharks can be directly attributed to changes in zooplankton abundances and this can have important implications for the management and conservation of the species (Thom et al., 1999).
Historically the basking shark was exploited for the high oil content of its liver, which was used in the steel, medical, and cosmetic industries, as well as as a fuel for lighting. A fishery remained in the Clyde until the 1980s, when declining numbers and reduced public support meant the fishery was no longer viable. The basking shark is now protected within the 12 nautical miles limit off Scotland under the Wildlife and Countryside Act 1981. Potential threats to basking shark populations include by-catch in fishing nets and disturbance or impact with vessels. The basking sharks slow growth rate and late sexual maturity coupled with overfishing has resulted in the shark becoming rare in areas where it was once common. As a result of the decline in numbers the species qualifies as vulnerable on a global level and the British population is classified as 'endangered' on the IUCN Red List.
In the waters of the Clyde, fifteen species of cetaceans have been recorded since 1980. However, there is a lack of data available on the abundance of cetaceans in the Clyde which limits the ability to determine trends and current status. Any reliable scientific data is restricted to observations of distribution rather than determining numerical abundance.
Some of the most significant non-fishing threats to biodiversity are outlined in the following section. The effects of fishing are discussed in Sections 6 and 7.
Non-native invasive species are considered by the UN to pose a great threat to biodiversity. Recently there has been an increase in the incidence of non-native marine species in coastal areas around the UK with potentially damaging impacts on native flora and fauna including commercial species. These effects can include competition with native species for food and space, alteration of habitats, changes in water quality and transmission of disease (Donnelly et al. 2010). Recent introductions into the marine environment of the Clyde include the cord-grass Spartina anglica which colonises shallow mudflats and competes with the seagrass Zostera noltii. Two of the most important vectors of invasive species are shipping and the aquaculture industry.
In the last 60 years climate change has altered the distribution and abundance of many marine species (MarClim, 2006) and there is now increasing concern of the impacts of climate change on the conservation and management of marine biodiversity. As temperatures increase a general shift in species distributions is expected as species respond to the changes of suitable 'climate space' available to them (Fields et al., 1993; MarClim, 2006). Individual species are likely to respond to temperature increases at different rates due to differences in their metabolism and physiological processes (Sims et al., 2004). In addition, climate change could cause local extinction of species that are unable to adapt to fluctuations in their physical environment.
4.7 Protection of Biodiversity
In order to effectively manage habitats and/or species of conservation importance, monitoring and regulation of those human activities likely to damage the areas of interest is essential.
Criteria for assessing the conservation importance of a habitat or species are a matter of continued debate. Such criteria are especially difficult to establish in the marine environment where basic knowledge of ecosystem function is still at a relatively low level (Hughes, 1998). The UK already has obligations to protect internationally important species and habitats that are listed in a variety of directives and conventions. Scotland has been active in establishing closed areas to protect biodiversity, for example, through the establishment of a No Take Zone ( NTZ) at Lamlash Bay, Arran. The Clyde Sea area also contains a wide range of protected sites of national and international importance for both biological and ornithological reasons. There are three types of protected areas related to the marine environment in the Clyde; Sites of Special Scientific Interest ( SSSI), Special Protection Areas ( SPA) and Marine Consultation Areas ( MCA).
SSSI's are areas of land or water that Scottish Natural Heritage ( SNH) considers to best represent our natural heritage - its diversity of plants, animals and habitats especially those of greatest value to wildlife conservation. They are designated under the Nature Conservation (Scotland) Act 1981 and are intended to form the essential building blocks of Scotland's protected areas for nature conservation (SNH website, 2011). There are currently 1,450 SSSIs in Scotland of which there are 14 that protect some aspect of the marine environment ( e.g. Seabirds, saltmarsh) of the Clyde Sea Area (see Table 4.1).
SPA's are designated areas of the terrestrial and marine environment in response to the Wild Birds Directive which aims to protect the habitats of migratory and threatened bird species. In the UK, SPAs must first be designated as an SSSI before gaining the protection as an SPA. The Clyde has one designated SPA in the area - the island of Ailsa Craig (see Table 4.1).
The Nature Conservancy Council has identified MCAs which deserve particular distinction in relation to the quality and sensitivity of their marine environment. The sites have no statutory status but are known to bodies such as SNH for marine conservation issues. The MCAs located in the Clyde include Loch Fyne and Loch Ryan due to the presence of important species such as the fireworks anemone and gaping file shell reefs.
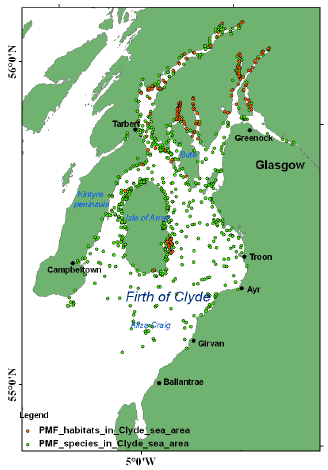
Implementing marine protected areas ( MPAs) is a priority for Scotland as it has international commitments to establish an ecologically coherent network of MPAs under the OSPAR agreement and so alongside other management practices, establishing MPAs will underpin the future use of the seas around Scotland. Currently SNH, the Joint Nature Conservation Committee ( JNCC) and Historic Scotland are working closely with Marine Scotland and stakeholders to provide advice to the Scottish Government on where MPAs should be designated. To help target biodiversity conservation action SNH and JNCC have put together a focused list of habitats and species of importance in Scottish waters - Priority Marine Features ( PMFs) which will underpin the selection of MPAs ( Figure 4.2). The Clyde sealochs have been identified as areas which represent major priorities for the establishment of MPAs due to the presence of many PMFs such as sea pens, native oysters and flame shell beds (Moore and James, 2011).
4.8 Suggestion for Further Work
There is much concern over inshore benthic habitats, especially those associated with maerl beds. It is estimated here that 10% of the sea bed of the Clyde might be able to support living maerl, although the figure may be smaller owing to constraints such as water flow and exposure (see Section 9). Most of these potential sites are within the 1 nm zone from the coast. In this zone the existing data sets of sea bed type, and also water depth, may be most in error. A comprehensive ground-determining acoustic survey, with associated validation using direct and indirect sampling methods, is needed of the Clyde 1 nm zone and waters out to the 20m depth contour.
Table 4.1 Sites of Special Scientific Interest located in the Firth of Clyde.
| Site of Special Scientific Interest ( SSSI) | ||||
|---|---|---|---|---|
| SSSI | Protection | Area (ha) | National Grid Reference | |
| 1 | Sanda Islands | Birds Maritime Cliffs |
81.45 | NR 725037 |
| 2 | Ballantrae Shingle Beach | Shingle | 32.74 | NX 080818 |
| 3 | Turnberry Dunes | Beetle Assemblage | 55.71 | NS 199060 |
| 4 | Maidens to Doonfoot | Shingle Maritime Cliffs |
224.5 | NS 316194 - NS 265177 |
| 5 | Troon Golf Links and Foreshore | Sand dune | 150.11 | NS 335287 |
| 6 | Western Gailes | Sand dune Invertebrate assemblage |
92.58 | NS 320358 |
| 7 | Portencross Coast | Mudflats Birds (non-breeding) |
477.9 | NS 190521 |
| 8 | Kames Bay | Sandflats | 4.6 | NS 171550 |
| 9 | Ballochmartin Bay | Sandflats | 18.9 | NS 182570 |
| 10 | Inner Clyde | Saltmarsh Birds |
1824.92 | NS 312811- NS494698 |
| 11 | Ruel Estuary | Saltmarsh | 332.78 | NS 010800 |
| 12 | Ailsa Craig | Birds Seabird colony |
99.94 | NX 020998 |
| 13 | South Coast of Arran | Shingle Maritime Cliff |
218.3 | NR 042213 |
| 14 | Clauchlands Point - Corrygills | Saltmarsh Maritime Cliff |
46.18 | NS 048338 |
Figure 4.2 The location of Priority Marine Features determined by SNH survey of the Firth of Clyde 2010.