Review of Access to New Medicines
An independent review to assess the impact of the new approach introduced in 2014 by Scottish Medicines Consortium (SMC).
6.7 How the new approach has had an impact on reliance on access to medicines on an individual patient basis (through individual patient treatment requests and peer approved clinical system)
6.7.1 The systems in place in Scotland have always been based on SMC deciding on whether a medicine should be available for use in NHSScotland whereas IPTR has placed the responsibility with Health Boards to decide whether individual patients should get access to medicines which have not been recommended for use by SMC. This onus does not change with PACS.
6.7.2 Given the increase in the number of end-of-life, orphan and ultra-orphan medicines accepted for use by SMC, it might have been expected that the number of clinicians and patients accessing medicines via IPTRs would have decreased, and further, a smaller proportion of those IPTRs would have been supported. This has not been the case.
6.7.3 This is in part due to the impact of the letter on Transitional Arrangements For Processing Individual Patient Treatment Requests sent to Health Boards by the CMO in November 2013 (SGHD/CMO(2013)20) 2. In tandem with the introduction of SMC's new approach this guidance served to increase the number of positive IPTRs decisions from Boards. There has also been a striking increase in the number of IPTRs submitted by clinicians to Boards which is less easy to explain. It may represent greater levels of awareness of new medicines and higher levels of expectation in relation to access even when SMC has reached a not recommended decision. In other words, there appears to be a greater reluctance on the part of patients, patient groups and clinicians to accept decisions by SMC not to recommend certain medicines.
6.7.4 Because of the way that IPTR data has been collected and the change in definitions the ability to analyse the data at any level other than total number of submissions and decisions is limited. As stated above the figures show a significant rise in the number of IPTRs submitted to Boards and an increase in the proportion of submissions approved.
6.7.5 It appears that clinicians and patients are increasing their use of the IPTR systems to successfully access medicines given not recommended status by SMC. Between 2012 and 2016 the number of IPTRs submitted rose from 389 in 2012-13, peaking at 918 in 2014-15 and dropping to 696 in 2015-16. In the same periods the approval rates were 69%, 82%, 90% and 87% respectively. (Table 4)
Table 4
| Total IPTRs | Approved | Rate | Not Approved | Rate | |
|---|---|---|---|---|---|
| 2012 - 2013 | 389 | 268 | 69% | 121 | 31% |
| 2013 - 2014 | 426 | 351 | 82% | 75 | 18% |
| 2014 - 2015 | 918 | 829 | 90% | 89 | 10% |
| 2015 - 2016 | 696 | 608 | 87% | 88 | 12% |
6.7.6 It is not possible with the same degree of confidence as applies to SMC data ( Paragraph 6.4.9) to differentiate end-of-life, orphan and ultra-orphan medicines within IPTR data but it is possible to make broad assumptions in relation to the available data. On the basis of this it does appear that the proportion of IPTRs that are for end-of-life, orphan and ultra-orphan medicines has changed being around 30% in 2012-13 and rising to around 60% in 2014-15 and 2015-16.
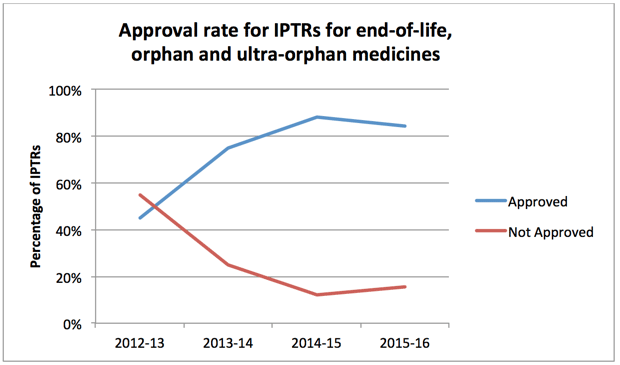
6.7.7 Using the same data subject to the same assumptions, it appears that in 2012-13 medicines that would now be classed as end-of-life, orphan and ultra-orphan had a lower approval rate at around 45% compared with other medicines at 80%. This difference appears to have narrowed with the figures for 2014-15 being in the region of 88% and 94% respectively and the same figures for 2015-16 being in the region of 85% and 91%. These figures are shown graphically in Chart 1.
6.7.8 Even given the limitations of the data it is clear that there has been an increase in the number of IPTRs submitted and an increase in the number of IPTRs being approved both in absolute and percentage terms. This is further evidence of increased access.
6.7.9 There was a strong concern expressed by several stakeholders that the system has evolved to the point where, in relation to access to end-of-life, orphan and ultra-orphan medicines the most likely outcome, regardless of SMC's decision, is that the patient will get access to new medicines. The question is not if access will be granted but when.
Chart 1
Recommendations
17 Review the data set and definitions for data relating to IPTRs collected by Boards with the aim of achieving consistency and comparability and also extending the dataset to include data on outcomes.