Swimming depth of sea trout
Data on swimming depth of sea trout Salmo trutta (L.) in Icelandic waters was extracted and collated into a suitable format for use in marine renewables risk assessment. The data shows that the fish are close to the surface much of the time, with some time being spent at greater depths.
4. Results
One matter to consider when drawing conclusions is the characteristics of the fish within a particular stock. The main information on the monitored sea trout is put forward in Tables 2 and 3. The length distribution was 32-76 cm at tagging and 47-81 cm at recapture (Table 1). The sea trout age at tagging ranged from 4-10 years. The life history based on scale readings of the tagged fish reflected the typical pattern in sea trout life in the local area. The sea trout spent 2-5 years in the river before their first sea migration. They then migrated every year into the sea and instances of up to six sea migrations were observed.
Table 3. The sea trout monitored during 1996-2008 listed by their reference number and the year of sea migration. When available the sex of the fish is given and their size and condition factor both at tagging and recapture. Information from scale readings on their life history is also presented: age, maturation history, age at first sea migration (smolt age) and history of earlier sea migrations.
| Fish | Sea migr. | Sex | Length of Fish At tagging = L1 At recapture = L2 |
Weight of Fish At tagging = W1 At recapture = W2 |
Condition Factor At tagging = K1 At recapture = K2 |
Life history Information Prior to the recorded sea migration (information from scales) |
|||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Year | L1 | L2 | W1 | W2 | K1 | K2 | Age | Maturity | Prior Spawning | Smolt Age | Prior Sea Migration | ||
| (no.) | Male= 1 Female=2 |
(cm) | (cm) | (g) | (g) | (K) | (K) | (year) | Immature=0 Mature=1 |
(number) | (year) | (number) | |
| 1 | 1996 | 42.4 | 47.1 | 830 | 1020 | 1.09 | 0.98 | 6 | 0 | 5 | 1 | ||
| 2 | 1996 | 44.9 | 1060 | 1.17 | 5 | 0 | 3 | 2 | |||||
| 3 | 1996 | 46.2 | 50.0 | 1150 | 1300 | 1.17 | 1.04 | 5 | 0 | 3 | 2 | ||
| 4 | 1996 | 2 | 46.8 | 52.0 | 1260 | 1500 | 1.23 | 1.07 | 5 | 0 | 3 | 2 | |
| 5 | 1996 | 1 | 47.4 | 54.0 | 1020 | 1704 | 0.96 | 1.08 | 5 | 0 | 3 | 2 | |
| 6 | 1996 | 2 | 49.6 | 54.8 | 1340 | 1870 | 1.10 | 1.14 | 5 | 0 | 3 | 2 | |
| 7 | 1996 | 1 | 51.7 | 62.2 | 1520 | 2590 | 1.10 | 1.08 | 0 | ||||
| 8 | 1996 | 1 | 53.5 | 62.6 | 1570 | 2550 | 1.03 | 1.04 | 6 | 1 | 3 | 3 | |
| 9 | 1996 | 55.5 | 1550 | 1700 | 0.91 | 1 | 1 | ||||||
| 10 | 1996 | 1 | 56.4 | 60.0 | 1720 | 2800 | 0.96 | 1.30 | 6 | 1 | 1 | 3 | 3 |
| 11 | 1996 | 1 | 56.1 | 64.0 | 1940 | 2750 | 1.10 | 1.05 | 5 | 1 | 1 | 2 | 3 |
| 12 | 1996 | 1 | 56.3 | 62.0 | 1910 | 2400 | 1.07 | 1.01 | 1 | ||||
| 13 | 1996 | 2 | 60.4 | 62.9 | 1950 | 2753 | 0.88 | 1.11 | 8 | 1 | 3 | 3 | 5 |
| 14 | 1996 | 1 | 67.5 | 71.6 | 3190 | 3780 | 1.04 | 1.03 | 8 | 1 | 1 | 4 | 4 |
| 15 | 1997 | 1 | 44.5 | 48.1 | 960 | 1100 | 1.09 | 0.99 | 4 | 0 | 0 | 3 | 1 |
| 16 | 1997 | 2 | 51.8 | 56.9 | 1620 | 2020 | 1.17 | 1.10 | 5 | 0 | 0 | 3 | 2 |
| 17 | 1997 | 1 | 53.7 | 60.0 | 1820 | 2110 | 1.18 | 0.98 | 5 | 0 | 0 | 3 | 2 |
| 18 | 1997 | 2 | 55.6 | 60.0 | 1830 | 2500 | 1.06 | 1.16 | 6 | 0 | 0 | 3 | 3 |
| 19 | 1997 | 1 | 57.5 | 62.1 | 2340 | 2700 | 1.23 | 1.13 | 7 | 0 | 0 | 5 | 2 |
| 20 | 1997 | 2 | 54.4 | 58.0 | 1390 | 2100 | 0.86 | 1.08 | 6 | 1 | 1 | 3 | 3 |
| 21 | 1997 | 2 | 57.3 | 64.0 | 1650 | 3000 | 0.88 | 1.14 | 7 | 1 | 1 | 3 | 4 |
| 22 | 1997 | 2 | 59.0 | 61.3 | 1990 | 2240 | 0.97 | 0.97 | 8 | 1 | 2 | 4 | 4 |
| 23 | 1997 | 1 | 64.5 | 69.5 | 2780 | 3250 | 1.04 | 0.97 | 7 | 1 | 1 | 4 | 3 |
| 24 | 1997 | 2 | 68.8 | 70.0 | 2910 | 4000 | 0.89 | 1.17 | 10 | 1 | 3 | 4 | 6 |
| 25 | 1998 | 2 | 32.0 | 50.0 | 340 | 1550 | 1.04 | 1.24 | 4 | 0 | 3 | 1 | |
| 26 | 1998 | 2 | 49.6 | 66.0 | 1400 | 3000 | 1.15 | 1.04 | 5 | 0 | 3 | 2 | |
| 27 | 1998 | 1 | 66.3 | 69.0 | 2860 | 3900 | 0.98 | 1.19 | 7 | 1 | 2 | 3 | 4 |
| 28 | 1998 | 2 | 69.5 | 74.0 | 2840 | 4200 | 0.85 | 1.04 | 1 | ||||
| 29 | 1998 | 1 | 71.9 | 76.0 | 4050 | 5000 | 1.09 | 1.14 | 7 | 1 | 2 | 3 | 4 |
| 30 | 1999 | 2 | 49.0 | 54.0 | 1500 | 1995 | 1.27 | 1.27 | 0 | ||||
| 31 | 1999 | 1 | 53.6 | 58.0 | 1600 | 2200 | 1.04 | 1.13 | 7 | 0 | 4 | 3 | |
| 32 | 1999 | 1 | 54.4 | 56.5 | 1890 | 2000 | 1.17 | 1.11 | 0 | ||||
| 33 | 1999 | 1 | 57.6 | 65.0 | 2140 | 3050 | 1.12 | 1.11 | 7 | 1 | 1 | 4 | 3 |
| 34 | 1999 | 1 | 60.6 | 66.0 | 2210 | 3000 | 0.99 | 1.04 | 1 | ||||
| 35 | 1999 | 62.3 | 64.7 | 2600 | 3520 | 1.08 | 1.30 | 1 | |||||
| 36 | 1999 | 1 | 73.8 | 78.0 | 4100 | 6100 | 1.02 | 1.29 | 7 | 1 | 2 | 3 | 4 |
| 37 | 2004 | 1 | 66.0 | 70.0 | 2911 | 5000 | 1.01 | 1.46 | 6 | 1 | 1 | 3 | 3 |
| 38a) | 2006 | 2 | 58.8 | 68.0 | 2495 | 3165 | 1.23 | 1.01 | 6 | 1 | 1 | 3 | 3 |
| 39 | 2006 | 1 | 59.0 | 64.8 | 2252 | 2600 | 1.10 | 0.96 | 7 | 1 | 1 | 4 | 3 |
| 38a) | 2007 | 2 | a) | 68.0 | a) | 3165 | 7 | 1 | 2 | 3 | 4 | ||
| 39b) | 2007 | 1 | 72.7 | 76.5 | 3290 | 4390 | 0.86 | 0.98 | 7 | 1 | 2 | 3 | 4 |
| 39b) | 2008 | 1 | 76.5 | 77.8 | 3625 | 3840 | 0.81 | 0.82 | 8 | 1 | 3 | 3 | 5 |
| 38a) | 2008 | 2 | 68.0 | 69.0 | 2400 | 3130 | 0.76 | 0.95 | 8 | 1 | 3 | 3 | 5 |
| 40 | 2008 | 1 | 76.2 | 81.3 | 4005 | 4345 | 0.91 | 0.81 | 7 | 1 | 2 | 2 | 5 |
a) Sea migration of Fish no. 38 was monitored for 3 years: 2006 (not recaptured); 2007 (recaptured); 2008 (tagged again in spring, recaptured)
b) Sea migration of Fish no. 39 was monitored for 2 years: 2006 (recaptured); 2007 (tagged again in spring, recaptured)
Table 4. The sea trout monitored during 1996-2008 listed by their reference number and the year of sea migration together with time of tagging, sea entry, river entry and information on the duration of the sea migration. Recapture time is given and the number of measurements derived from the densest uniform measuring interval covering the sea migration of all fish in the given year.
| Fish | Sea Migration | Tagging Time | Sea Migration Duration | Recapture Time | ||||
|---|---|---|---|---|---|---|---|---|
| Number | Year | Date | Sea Entry | River Entry | Duration | Fish Recaptured/ Tag Retrieved |
Uniform Measurements throughout Sea Migration |
|
| (no.) | (year day.month) | (day.month) | (day.month) | (hrs) | (days) | (year-season) | (no.) | |
| 1 | 1996 | 1996 5.5 | 7/8 | 2/9 | 632 | 26 | 1997-Spring | 632 |
| 2 | 1996 | 1996 20.4 | 26/5 | 16/8 | 1983 | 83 | 1996-Summer | 1,983 |
| 3 | 1996 | 1996 5.5 | 24/6 | 17/7 | 545 | 23 | 1996-Summer | 545 |
| 4 | 1996 | 1996 20.4 | 3/6 | 20/7 | 1127 | 47 | 1996-Autumn | 1,127 |
| 5 | 1996 | 1996 5.5 | 6/7 | 2/9 | 1400 | 58 | 1997-Spring | 1,400 |
| 6 | 1996 | 1996 5.5 | 1/6 | 17/7 | 1109 | 46 | 1996-Summer | 1,109 |
| 7 | 1996 | 1996 5.5 | 1/6 | 21/7 | 1193 | 50 | 1996-Autumn | 1,193 |
| 8 | 1996 | 1996 4.5 | 4/6 | 17/8 | 1787 | 74 | 1997-Spring | 1,787 |
| 9 | 1996 | 1996 4.5 | 24/5 | 22/7 | 1421 | 59 | 1997-Spring | 1,421 |
| 10 | 1996 | 1996 20.4 | 22/5 | 23/7 | 1483 | 62 | 1996-Summer | 1,483 |
| 11 | 1996 | 1996 20.4 | 31/5 | 18/7 | 1148 | 48 | 1996-Autumn | 1,148 |
| 12 | 1996 | 1996 4.5 | 4/6 | 17/7 | 1045 | 44 | 1996-Autumn | 1,045 |
| 13 | 1996 | 1996 20.4 | 1/6 | 12/7 | 999 | 42 | 1996-Summer | 999 |
| 14 | 1996 | 1996 4.5 | 26/5 | 1/9 | 2356 | 98 | 1997-Summer | 2,356 |
| 15 | 1997 | 1997 27.4 | 19/6 | 9/8 | 619 | 52 | 1998-Spring | 619 |
| 16 | 1997 | 1997 26.4 | 29/5 | 19/7 | 610 | 51 | 1997-Autumn | 610 |
| 17 | 1997 | 1997 26.4 | 31/5 | 21/7 | 606 | 51 | 1998-Spring | 606 |
| 18 | 1997 | 1997 26.4 | 10/6 | 19/7 | 478 | 40 | 1997-Summer | 478 |
| 19 | 1997 | 1997 27.4 | 10/6 | 21/7 | 499 | 42 | 1997-Summer | 499 |
| 20 | 1997 | 1997 26.4 | 31/5 | 30/7 | 727 | 61 | 1997-Autumn | 727 |
| 21 | 1997 | 1997 26.4 | 17/5 | 29/7 | 877 | 73 | 1997-Summer | 877 |
| 22 | 1997 | 1997 26.4 | 17/5 | 20/7 | 772 | 64 | 1997-Autumn | 772 |
| 23 | 1997 | 1997 26.4 | 17/5 | 11/8 | 1034 | 86 | 1998-Summer | 1,034 |
| 24 | 1997 | 1997 26.4 | 31/5 | 24/7 | 656 | 55 | 1997-Summer | 656 |
| 25 | 1998 | 1998 2.5 | 6/6 | 22/8 | 926 | 77 | 2000-Spring | 926 |
| 26 | 1998 | 1998 3.5 | 5/6 | 26/7 | 613 | 51 | 2000-Summer | 613 |
| 27 | 1998 | 1998 2.5 | 24/6 | 11/8 | 576 | 48 | 1998-Autumn | 576 |
| 28 | 1998 | 1998 2.5 | 24/6 | 11/8 | 575 | 48 | 1998-Summer | 575 |
| 29 | 1998 | 1998 3.5 | 24/6 | 30/7 | 433 | 36 | 2000-Summer | 433 |
| 30 | 1999 | 1999 1.5 | 29/5 | 11/8 | 3586 | 75 | 1999-Summer | 35,869 |
| 31 | 1999 | 1999 1.5 | 12/6 | 29/7 | 2251 | 47 | 1999-Summer | 2,251 |
| 32 | 1999 | 1999 1.5 | 12/6 | 30/7 | 2276 | 47 | 1999-Summer | 2,276 |
| 33 | 1999 | 1999 1.5 | 12/6 | 13/8 | 2976 | 62 | 1999-Summer | 2,976 |
| 34 | 1999 | 1999 1.5 | 23/5 | 6/8 | 3633 | 76 | 1999-Summer | 3,633 |
| 35 | 1999 | 1999 1.5 | 1/6 | 24/7 | 2581 | 54 | 2000-Spring | 2,581 |
| 36 | 1999 | 1999 2.5 | 12/6 | 31/7 | 2373 | 49 | 1999-Summer | 2,373 |
| 37 | 2004 | 2004 24.4 | 28/5 | 3/8 | 19262 | 67 | 2004-Autumn | 19,262 |
| 38a) | 2006 | 2005 2.10 | 1/6 | 1/8 | 1460 | 61 | 2007-Autumn | 1,460 |
| 39 | 2006 | 2005 2.10 | 5/6 | 5/8 | 1465 | 61 | 2006-Autumn | 1,465 |
| 38a) | 2007 | 2005 2.10 | 30/5 | 25/7 | 674 | 56 | 2007-Autumn | 674 |
| 39b) | 2007 | 2007 2.4 | 5/6 | 11/7 | 433 | 36 | 2008-Winter | 433 |
| 39b) | 2008 | 2008 13.5 | 1/6 | 13/7 | 2013 | 42 | 2008-Winter | 2,013 |
| 38a) | 2008 | 2008 13.5 | 23/5 | 29/6 | 1776 | 37 | 2008-Summer | 1,776 |
| 40 | 2008 | 2008 13.5 | 4/6 | 20/8 | 3687 | 77 | 2009-Winter | 3,687 |
a) Sea migration of Fish no. 38 was monitored for 3 years: 2006 (not recaptured); 2007 (recaptured); 2008 (tagged again in Spring, recaptured)
b) Sea migration of Fish no. 39 was monitored for 2 years: 2006 (recaptured); 2007 (tagged again in Spring, recaptured)
Based on the life history data determined from scale reading, it was shown that the sea trout first spawned after the second or, more commonly, the third sea migration and then each year following sea migration. Back calculation of length from scales showed that most of the sea trout were 20-30 cm long when starting their first sea migration.
The period in which sea migrations of sea trout took place in 1996-2008 (Figure 3) shows that the main foraging of sea trout takes place in June and July although the first ones are already feeding at sea in May and the last ones are finishing their feeding migration in September. The main sea migration period corresponds closely with the non-darkness period from May 20 to July 23.
Fig. 3. The time sea trout spent at sea by research week as a proportion of the overall stay of all sea trout monitored throughout their sea migration with DSTs in each year. The mean proportional stay for each year is also given.
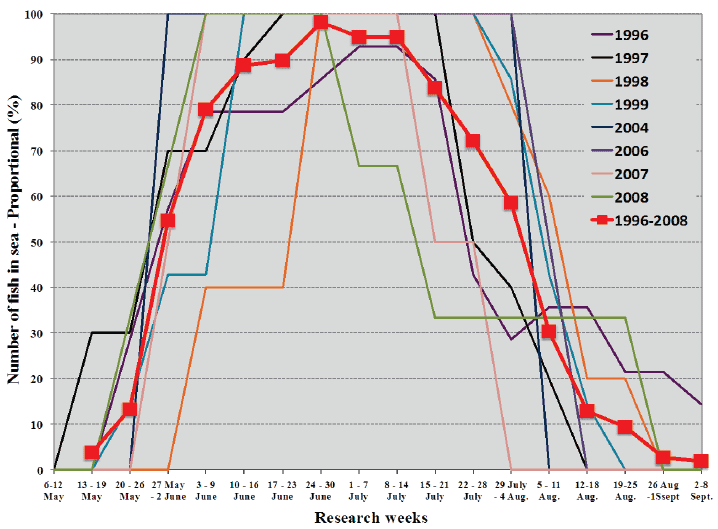
The similar sea migration duration of the same fish monitored for 2 and 3 sea migrations (Table 4) shows how uniform the sea migration of individuals can be between years.
In Figs. 4-7, examples of depth profiles from recordings throughout sea migration of four sea trout are shown, together with data from their migration in fresh water before and following their sea journey. The data demonstrate the pelagic behaviour of the sea trout during their sea migration. The temperature and salinity recordings show that the feeding migrations are shoreline orientated. This is shown by the rather frequent and brief deviations in temperature and salinity. These reflect the movements into low salinity warmer sea water close to the shore where the large inflow of fresh water from glacial rivers creates these conditions.
Fig. 4. Migratory pattern of sea trout during sea (feeding) migration and river migration. The depth distribution of the fish and corresponding ambient temperature and salinity in relation to time are shown.
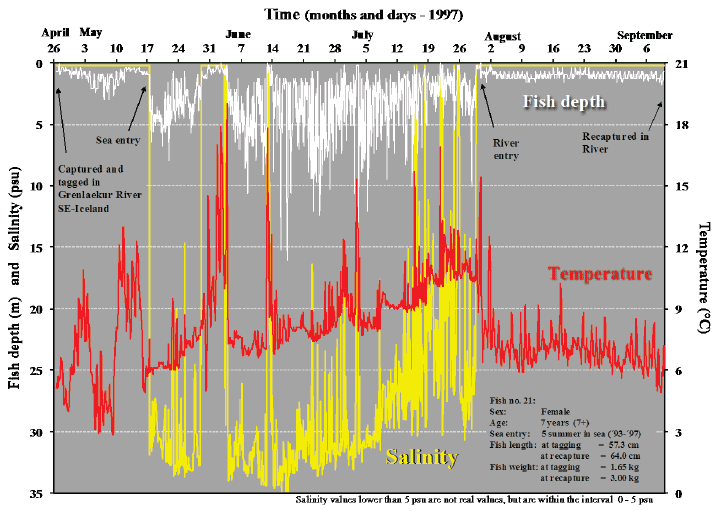
Fig. 5. Migratory pattern of sea trout during sea (feeding) migration and river migration. The depth distribution of the fish and corresponding ambient temperature and salinity in relation to time are shown.
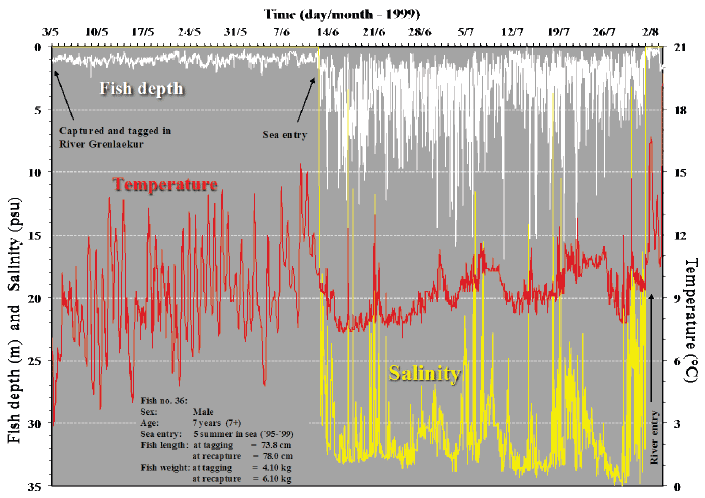
Fig. 6. Migratory pattern of a sea trout during sea (feeding) migration and river migration. Depth distribution of the fish and corresponding ambient temperature in relation to time are shown in A and the behaviour of the fish is examined in more detail in B.
A.

B.

Fig. 7. Migratory pattern of sea trout during sea (feeding) migration and river migration. The depth distribution of the fish and corresponding ambient temperature are shown in relation to time.
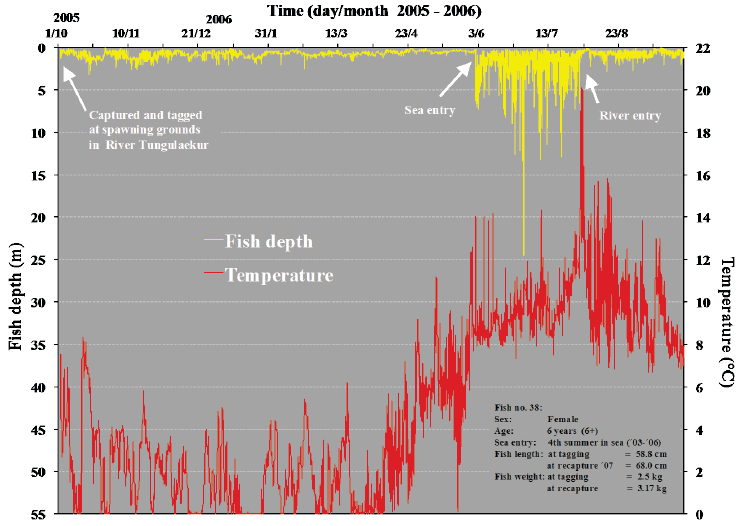
The overall mean depths occupied by all the sea trout in any given year were calculated (Table 5). The results show that sea trout mainly stay just below the sea surface. In shallow water, they could of course also be close to the sea bottom at the same time. Some depth profiles from rapid sampling indicate periods of feeding up from bottom but the opposite, feeding down from surface layers, is also observed.
In Figure 8, the mean values for the overall depth of sea trout in sea is shown for every sampling year during the period 1996-2008 with 95% confidence limits. The variation is significant between some of the years although the absolute difference is small. Table 6 (and Fig. 9) show, for each sampling year, the proportion of the overall time spent within given 5m depth intervals, with the means for all fish across all years included in Table 6. Table 7 shows the mean depth within the 5 m depth intervals, given along with standard deviation for the eight sampling years, and again includes the means for all fish across all years.
Fig. 8. Mean values for the overall mean fish depth with 95% confidence limits for each of the years sea trout were monitored with data storage tags during sea migration. The numerical values of the means and the corresponding lower and upper bounds of the 95% confidence intervals are shown.
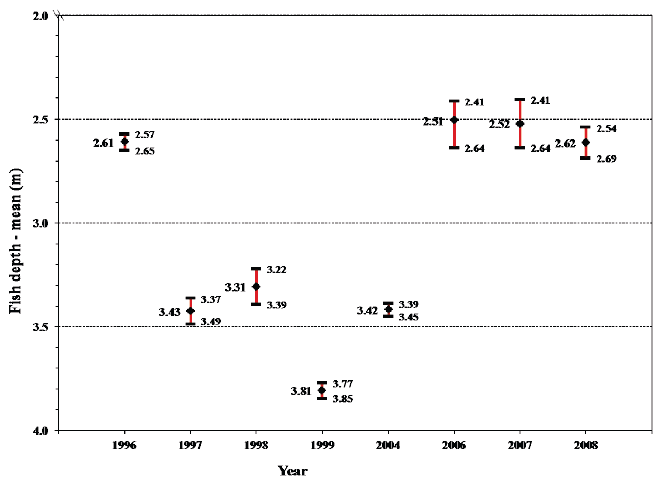
Table 5. Overall mean depth of sea trout for every year within the period 1996-2008. The mean standard deviation is shown and the maximum depth recorded. The number of fish behind the recordings in sea each is also given along with the recording interval that covered the total sea migration and the total number of recordings behind the mean depth.
| Year | Fish Depth (Based on Uniform Measurements throughout the Sea Migration) |
Fish Number Monitored during Sea Migration |
Recording Interval Covering the whole Sea Migration and Corresponding Number of Rec. |
|||
|---|---|---|---|---|---|---|
| Mean | SD | Max | (no.) | Intervals | (no.) | |
| 1996 | 2.6 | 2.73 | 46.5 | 14 | 1 hour | 18,228 |
| 1997 | 3.4 | 2.60 | 21.5 | 10 | 2 hours | 6,878 |
| 1998 | 3.3 | 2.41 | 17.0 | 5 | 2 hours | 3,123 |
| 1999 | 3.8 | 2.59 | 26.9 | 7 | 30 min | 19,676 |
| 2004 | 3.4 | 2.11 | 32.8 | 1 | 5 min | 19,262 |
| 2006 | 2.5 | 2.57 | 45.2 | 2 | 1 hour | 2,925 |
| 2007 | 2.5 | 1.97 | 13.4 | 2 | 2 hours | 1,107 |
| 2008 | 2.6 | 3.25 | 38.3 | 3 | 30 min | 7,476 |
| Sea migration monitored - Total = | 44 | |||||
Table 6. Overall proportional time spent within given depth intervals for each of the years
| Depth Interval | Dwelling within Depth Interval - Proportional (%) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1996 | 1997 | 1998 | 1999 | 2004 | 2006 | 2007 | 2008 | Overall mean | |
| 0-5 m | 85.44 | 78.22 | 80.37 | 72.21 | 81.57 | 88.58 | 88.98 | 87.68 | 81.49 |
| 5.1-10 m | 12.54 | 19.31 | 17.64 | 25.37 | 17.66 | 9.33 | 10.57 | 9.03 | 16.34 |
| 10.1-15 m | 1.45 | 2.22 | 1.83 | 2.19 | 0.56 | 1.64 | 0.45 | 1.67 | 1.75 |
| 15.1-20 m | 0.43 | 0.22 | 0.16 | 0.21 | 0.09 | 0.31 | 0.95 | 0.32 | |
| 20.1-25 m | 0.07 | 0.03 | 0.01 | 0.03 | 0.10 | 0.40 | 0.06 | ||
| 25.1-30 m | 0.02 | 0.01 | 0.07 | 0.17 | 0.02 | ||||
| 30.1-35 m | 0.02 | 0.02 | 0.07 | 0.01 | |||||
| 35.1-40 m | 0.03 | 0.00 | |||||||
| 40.1-45m | 0.02 | 0.01 | |||||||
| 45.1-50 m | 0.01 | 0.03 | 0.01 | ||||||
Table 7. Mean depth of sea trout within 5 m depth intervals, given along with standard deviation for the 8 sampling years.
| Depth Interval | Mean Depth of Fish within given Depth Interval (m) | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1996 | 1997 | 1998 | 1999 | 2004 | 2006 | 2007 | 2008 | No fish | Overall mean | |||||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |||
| 0-5 m | 1.7 | 1.21 | 2.3 | 1.26 | 2.4 | 1.31 | 2.5 | 1.26 | 2.7 | 1.05 | 1.8 | 1.12 | 2.0 | 1.22 | 1.7 | 1.05 | 44 | 2.1 |
| 5.1-10 m | 6.8 | 1.29 | 6.8 | 1.32 | 6.6 | 1.25 | 6.7 | 1.28 | 6.5 | 1.11 | 6.9 | 1.37 | 6.6 | 1.29 | 6.8 | 1.30 | 44 | 6.8 |
| 10.1-15 m | 11.7 | 1.36 | 11.8 | 1.26 | 11.5 | 1.20 | 11.6 | 1.24 | 11.5 | 1.25 | 11.6 | 1.24 | 11.3 | 1.18 | 12.1 | 1.39 | 44 | 11.7 |
| 15.1-20 m | 16.8 | 1.43 | 16.2 | 1.45 | 16.2 | 0.86 | 16.0 | 0.93 | 17.9 | 1.44 | 16.9 | 1.55 | 17.2 | 1.39 | 32 | 16.6 | ||
| 20.1-25 m | 22.0 | 1.69 | 21.3 | 0.28 | 23.5 | 22.5 | 1.33 | 22.2 | 2.04 | 21.9 | 1.33 | 14 | 22.1 | |||||
| 25.1-30 m | 26.5 | 0.70 | 26.9 | 27.3 | 1.55 | 27.5 | 1.23 | 7 | 26.9 | |||||||||
| 30.1-35 m | 34.3 | 0.70 | 31.8 | 1.20 | 31.9 | 1.63 | 4 | 32.5 | ||||||||||
| 35.1-40 m | 38.2 | 0.14 | 2 | 38.2 | ||||||||||||||
| 40.1-45 m | 41.5 | 0.98 | 1 | 41.5 | ||||||||||||||
| 45.1-50 m | 46.5 | 45.2 | 2 | 45.9 | ||||||||||||||
Fig. 9. The proportion of the time that the sea trout spent on average within 5 m depth intervals during their sea migration in each year. The time fish spent within the depth range is based on data for the total sea migration at the same measuring interval and is shown as the proportion of the total time at sea in the given year.

The depth distribution for the sampling years shows that the sea trout spent the majority of their time in the uppermost 5 m while migrating at sea (72-89%). The sea trout used the 5-10 m depth zone for 9-25% of their time with very little time spent in deeper sea layers. Although the sea trout were observed to move down to 47 m (Table 5) such movement into very deep layers was very rare. As given in Table 6, the time spent below 30 m was 0.1% or less for the sampling years. The time spent below 20 m acounted for 0.7% or less of their time at sea.
In Fig. 10, results based on recordings from subsampling are used to present the depth distribution in same way as in Fig. 9. The results shows the same main distribution pattern as presented in Fig. 9 although the subsampled recordings are potentially biased as the sampling was mainly very clustered in time.
Comparison of mean fish depths for a period of high frequency sampling in 2004 did not show significant difference in the mean depth observed between the 5sec-2hours sampling intervals, although the variation increased slightly for the coarser sampling periods (Fig. 11). The results partly reflected the uniformity of the depth the sea trout were utilizing.
The results on the sea trout swimming depth at sea (Figs. 8-10), as well as the results from comparison of mean depth of individuals (Figs. 12-14), results from comparison of light condition on the depth of sea trout (Figs. 15-16) and results from comparison of mean depth throughout the summer (Figs. 17-19) lead to the same conclusion, namely that the great majority of the sea trout are feeding very close to the sea surface and that seasonal and circumstantial environmental differences during their main sea migration period did not change this behaviour. The feeding grounds are mostly close to shore. Although the sea trout were observed to stay close to surface most of the time, this could at times encompass feeding close to sea bottom, if they are in shallow water.
Fig. 10. The time that the sea trout spent at average within 5 m depth intervals during their sea migration for given years. The time fish spent within depth range is based on data sampled with denser measurement during the subsampling period during the sea migration. For each year the measuring interval is the same. The time spent within the given depth range is shown as a proportion of the total time spent at sea in the given year.
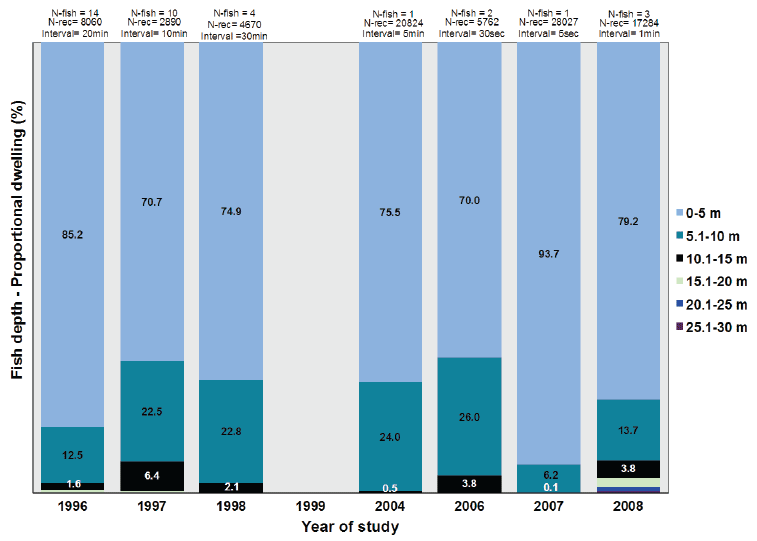
Fig. 11 . Mean values of fish depth of fish no. 37 with 95% confidence limits. The mean values are based on 7 different sampling rates in order to compare the effect that sampling rates have on the observed results.
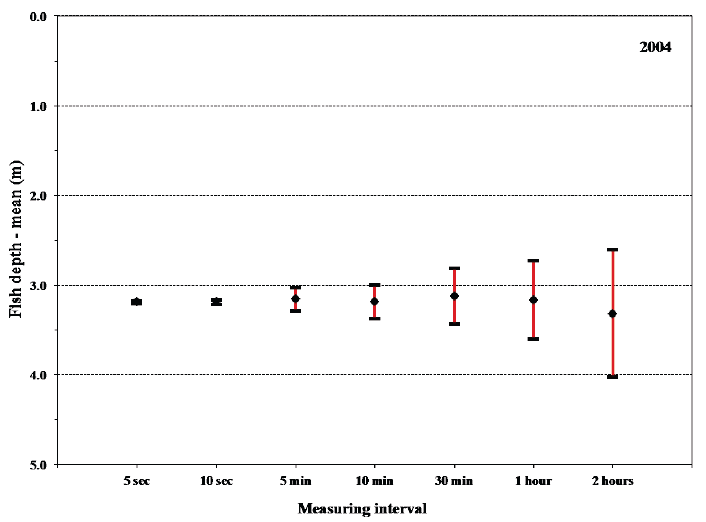
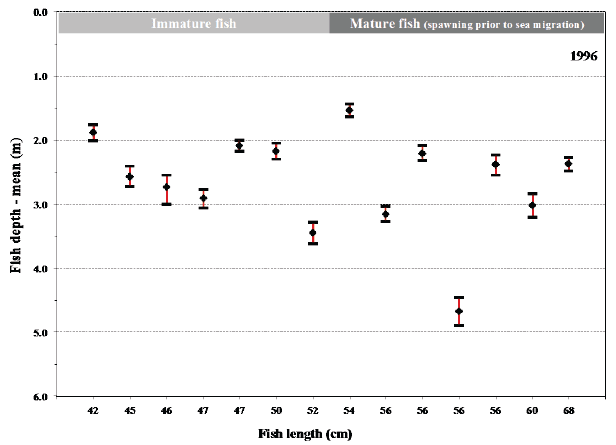
Fig. 12. Mean values with 95% confidence limits of the depth of individual fish (length at tagging given) during their sea migration in 1996 (A) and in 1997 (B). Maturation status of the fish at tagging before their sea migration is shown for comparison.
A.
B.
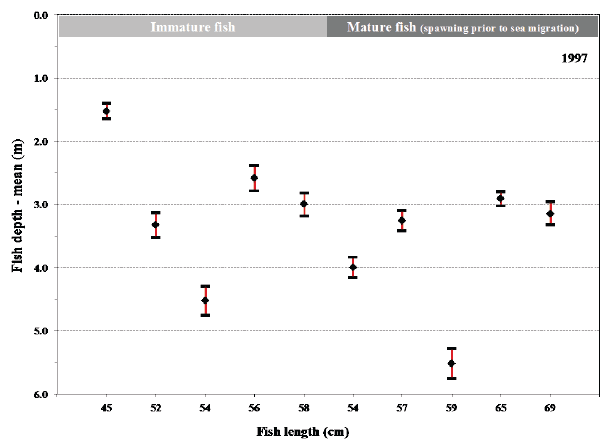
Fig. 13. Mean values with 95% confidence limits of the depth of individual fish (length at tagging given) during their sea migration in 1998 (A) and in 1999 (B). Maturation status of the fish at tagging before their sea migration is shown for comparison.
A.
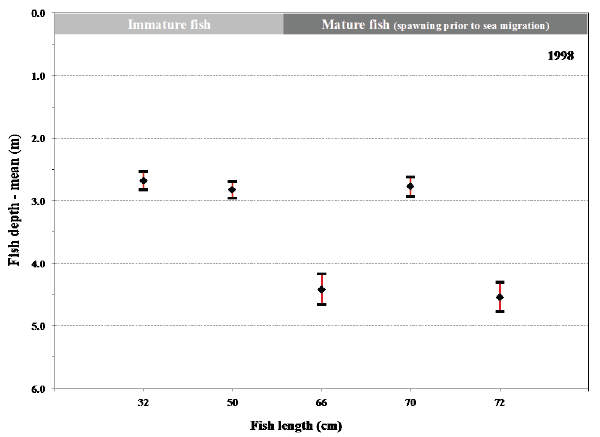
B.
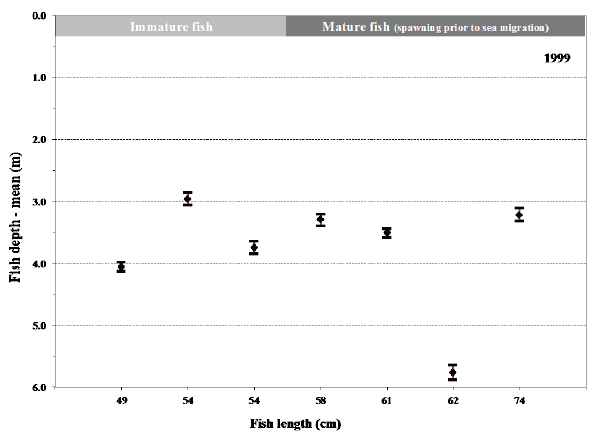
Fig. 14. Mean values with 95% confidence limits of the depth of individual fish (length at tagging given) during their sea migration in 2004-2008. Maturation status of the fish at tagging before their sea migration is shown for comparison.
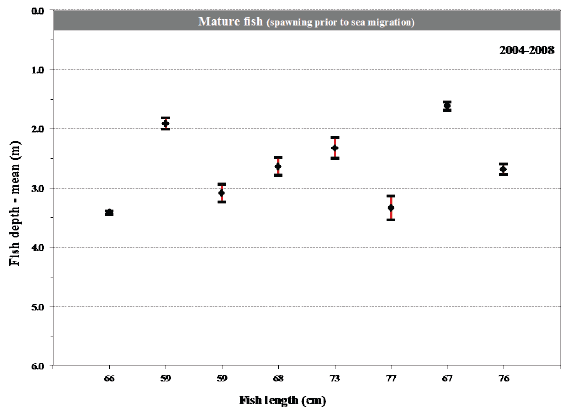
Fig. 15. Mean values with 95% confidence limits of the depth of sea trout during sea migration in 1996 during the summer period when darkness is not involved.
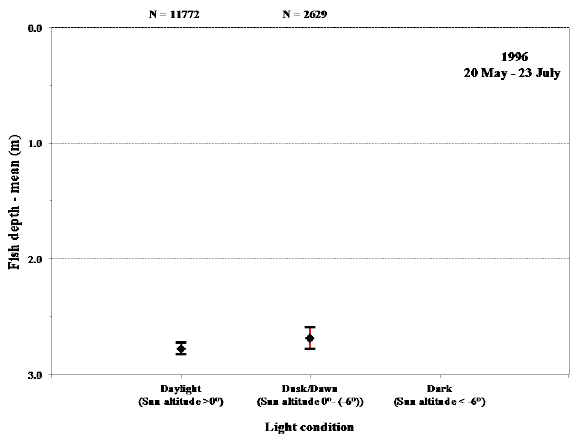
Fig. 16. Mean values with 95% confidence limits of the depth of sea trout during sea migration in 1996 during the late summer period when hours of darkness are involved.

Fig. 17. Mean values with 95% confidence limits of the overall fish depth recorded by sea trout during sea migration in 1997 in each research week.
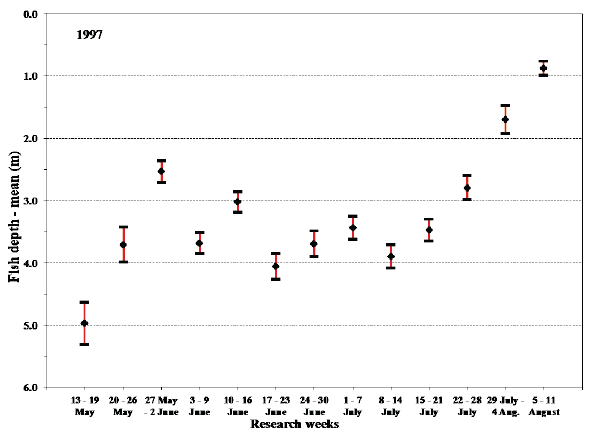
Fig. 18. Mean values with 95% confidence limits of the overall temperature experienced by sea trout during sea migration in 1997 in each research week.
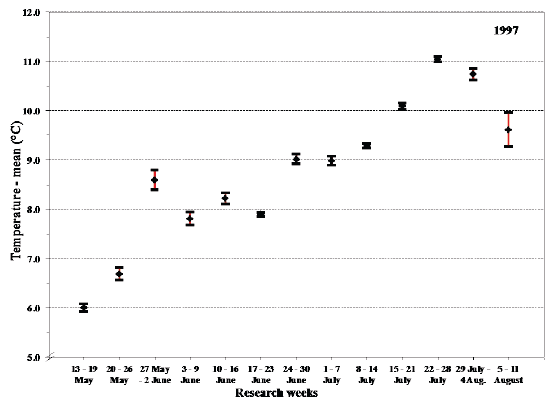
Fig. 19. Mean values with 95% confidence limits of the overall salinity experienced by sea trout during sea migration in 1997 in each research week.
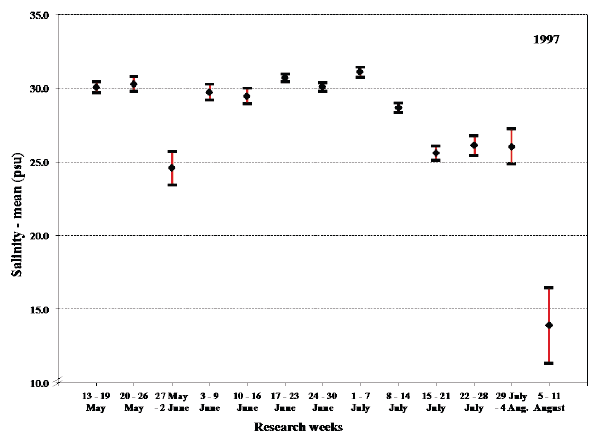
The deviation in the overall mean temperature and salinity experienced by the sea trout during 20-26 May 1997 represents a temporary stay in brackish water (Fig. 18 and 19). This could be very close to the shore or in estuarine areas but could also partly be explained by proportion of fish starting their sea migration in this period. The similar deviation during 5-11 August is presumably the result of fish in the estuary finishing their sea migration.
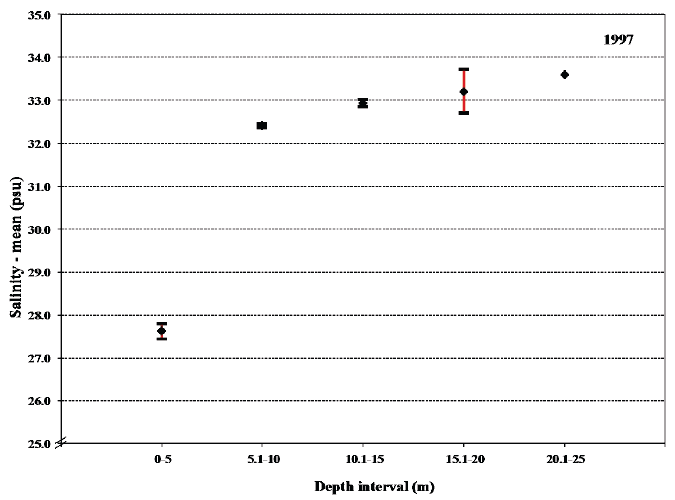
When the overall mean values for temperature and especially salinity within each of the 5 m depth intervals are compared it is apparent that these values for the uppermost 5 m differ significantly from the other depth intervals (Figs. 20 and 21). The low salinity shows that during sampling in 1997 the sea trout were experiencing relatively low salinity during the majority of their sea migration. This again points towards their shoreline orientation when feeding at sea.
Fig. 20. Mean values with 95% confidence limits of the temperature experienced during sea migration of sea trout in 1997 in the 5 m depth intervals.
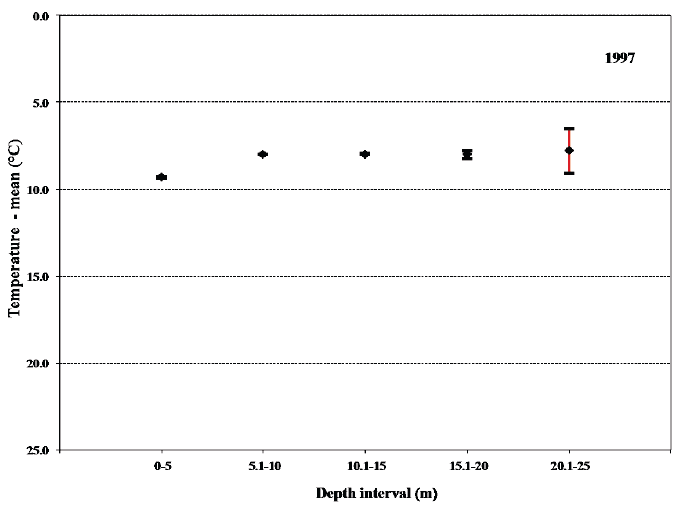
Fig. 21. Mean values with 95% confidence limits of the salinity experienced during sea migration of sea trout in 1997 in the 5 m depth intervals.
Contact
Email: Ross Gardiner