Queen Elizabeth University Hospital: case note review - overview report
This overview report examines the incidence and impact of qualifying episodes of infection in paediatric haemato-oncology patients cared for at the Queen Elizabeth University Hospital and the Royal Hospital for Children from 2015 to 2019 and the potential link to the hospital environment.
8. Areas of Concern
In this chapter we bring together issues encountered in the course of our Review that have caused us concern or otherwise wish to comment. We have separately identified examples of the good practice we observed which we discuss in chapter 9.
We address concerns about data availability and its quality in section 8.1 and offer a detailed analysis of our observations about the management, investigation and reporting of infection outbreaks in section 8.2: this is the longest section in this chapter and, we believe, provides a context against which the previous recognition and investigation of GNE bacteraemia within NHS GGC can be viewed. Section 8.3 looks at microbiology and IPC information systems and includes some important observations about how data relating bacterial typing were collated and stored. Section 8.4 addresses issues about clinical records and section 8.5 looks in more detail at Adverse Event reporting. Sections 8.6–8.8 address selected aspects of clinical practice.
Some of these observations create opportunities for changes to policy and practice, and all, we believe, offer learning for the future.
8.1 Data Availability and Data Quality
The concurrence of the COVID-19 pandemic with the period of the Review created additional challenges both for NHS GGC and for the Review Team, with pressures on staff resource and the necessity to work remotely. There were, nevertheless, areas in which NHS GGC’s response to the Panel’s need for access to data was unsatisfactory and where we encountered difficulties in its presentation.
8.1.1 Access to NHS GGC information systems
In March 2020, an Information Sharing Agreement (ISA) was approved between Scottish Government and NHS GGC as the designated data controllers for the project. This provided permissions for individuals named on the agreement to access specified NHS GGC IT systems, and set out the principles governing the use of the information obtained from those systems. A process was established whereby any amendments required to the ISA would be raised with Scottish Government and with NHS GGC Information Governance by email and subsequently submitted to the NHS GGC Caldicott Guardian for approval.
As resource assigned to the Review, particularly in relation to IPC expertise, had been re-directed to COVID-19 related work, changes to those contributing to the Review Team became inevitable, requiring amendment of the ISA. The response time from making such requests to NHS GGC Information Governance to receipt of approval was often slow. It became common that repeated emails were required to generate a response. One example was a request to add an individual to the ISA on 23 October 2020 which, despite twice chasing for a response and escalating the matter to a member of the NHS GGC Senior Executive Team, was not approved until 9 November 2020. Given the time constraint under which the Review Team was working, this caused delay to planned work.
Finally, there were several instances when the access of all members of the Review Team who had access to NHS GGC IT systems was unexpectedly suspended. For example, on 4 September 2020 the Review Team requested account extensions beyond the existing agreement to the end of that month. Despite this, all accounts were still suspended on 30 September 2020. This caused delays in retrieving the information required for us to carry out the review.
8.1.2 Environmental Microbiology and Facilities Maintenance Work data
By April 2020, the Review Team had identified the need for additional data which were not available from the access already granted to the clinical records. We needed to be able to consider environmental data for the QEUH/RHC buildings; NHS GGC was asked to supply results relating to environmental microbiology sampling and the records of facilities department maintenance work for the duration of our review. We have discussed the significance of such records in Chapter 5, specifically, sections 5.2, 5.4 and 5.5; notably, we needed data that could be related in time and place to the locations of care of the patients within the review. We initially requested ‘all environmental microbiology sampling results that are available’ in an email on 7 April 2020. Thereafter difficulties were encountered over the supply and quality of these data.
The first merged data were received on 11 May 2020 were for water samples only; we later discovered drain samples were not included. We found that the data appeared to be incomplete and inconsistent. For example, a large number of samples listed in the database provided were recorded to have ‘No DMA[73] Record’ and, for samples that had such a record, either no sample location was provided or it was identified only at the level of the ward and not by the patient room or other designated location.
In line with our initial request, the facilities maintenance data provided to us first came as HAI-SCRIBE[74] records. We subsequently recognised that our requirements would be better addressed by focusing on the work actually carried out in the Paediatric Haematology Oncology wards, and not on the HAI-SCRIBE risk assessments. Further communication with staff in NHS GGC Estates and Facilities provided these data on 1 June 2020 but with similarly limited location information which did not permit us to relate, for example, the visit of a plumber to Ward 2A to deal with a blocked drain, to any specific room or drain.
During June-September 2020, further attempts were made to communicate with NHS GGC to discuss the data received, its incompleteness and the lack of location identification, as well as to clarify additional data the Panel would require for review.
At a meeting on 1 October 2020 we began to understand for the first time that NHS GGC did not have data available in the form we needed or, it seemed in one place. Consequently, NHS GGC had to undertake significant work to generate an appropriate data set from source records. At the beginning of December 2020, as we were coming to the end of our initial review process, we received what we now believe to be the complete records available for water samples, environmental ‘hard surface’ samples (which included the drain samples) and maintenance data.
This delay, and others regarding our access to the laboratory information systems (section 8.1.3), necessitated us to undertake a second complete review of the entire series of infection episodes so as to incorporate this additional information.
8.1.3 Laboratory information systems
In Chapter 3, section 3.5, we have discussed the relevance of our access to both the Telepath and ICNet systems. Although access to ICNet was agreed in the ISA in March 2020, by August 2020 it had become evident that we had no access to the system. When exploring this with the NHS GGC IPC Team, we were initially advised that we would not require direct access and that any information could be requested from them as required. We rejected this suggestion and escalated the matter to senior members of the NHS GGC Executive Team who rapidly resolved the issue. In the meantime, however, the IPC Team provided us with extracts from ICNet for five patients scheduled for imminent Panel review. Our review of these extracts suggested that, in four of the five cases, potentially relevant information was lacking but we were unable to ascertain if this was because it was not available or had not been included – emphasising our need for direct access to the system itself.
It was only at this time that we became aware of the Telepath system. Initially, we received copies of entries in its Patient Note Pad function from NHS GGC. This part of the system records information relevant to our review as it documents the dialogue between microbiology and clinical staff and provides information about, for example, the identification and antibiotic sensitivity profile of the organism concerned; the advice given about the type and duration of treatment; and the necessity (or otherwise) to remove a central line. Our initial request to be provided with material recorded in Telepath within a period of 1 month either side of the date of an infection episode proved unsatisfactory as a wider perspective seemed likely to be helpful. We also recognised that we would benefit from independent IPC expertise to interrogate both this and ICNet.
By late September 2020, we had identified the IPC resources required to support our work and made arrangements via NHS GGC Information Governance for access to both systems. However, the access level set in Telepath provided limited functionality; this meant that it was not possible to copy or download information from the system, requiring data to be transcribed into a separate Word document for us to use in our review.
These issues contributed to impede and delay our ability to assess and integrate relevant information into our case note reviews.
8.1.4 IMT and PAG meeting records
In assessing causation in relation to specific infection episodes, we began to look for data utilised in NHS GGC’s internal processes for investigating and responding to infections in real time. We requested, and from September 2020 began to receive, minutes from PAG and IMT meetings. The relevance and agreed process for implementing such meetings is defined in NHS GGC’s SOP for Outbreaks/Incidents in hospitals.
We initially noted that, for some of the 2018 IMT minutes, environmental microbiology sample results were given with details of sample location. This prompted us to undertake an exercise to cross check some of the sample results found in IMT minutes against the data we had received. An example of the inconsistency we encountered is shown as Example 8.1:
Example 8.1
“Subsequently, the colonised patient and one of the cases were nursed sequentially in Room 12, which is the only room with water results positive for Stenotrophomonas.” (IMT Minute, 23 March 2018)
Our investigation of the environmental water sample results received from NHS GGC in May 2020, showed that a water sample positive for Stenotrophomonas could be identified, but no sample location was recorded.
This experience challenged our confidence about how records of data utilised in IMT meetings were located and stored. We began to reflect whether, on the basis of what we had seen, NHS GGC had systems in place to ensure comprehensive reporting and recording of data relevant to the IMT process.
We reassured ourselves that the quality of the environmental microbiology sampling data received in December 2020 had improved by undertaking a further cross checking exercise from which, for example, the sample results highlighted in Example 8.1, shown above, could now be identified. Unfortunately, inconsistent coding characterised this final data set and made it difficult for Data Managers to present the data in a usable and searchable format for the Panel to review. Substantial further manual checking and data cleaning were required before this could be achieved, resulting in an additional delay at a point when we were under considerable pressure to complete our second round of reviews.
8.2 Managing, investigating and reporting infection outbreaks
We examined the notes of investigations into outbreaks of infection undertaken by NHS GGC, to help our consideration of the likelihood of a hospital environmental source for the Gram-negative environmental infections under our review.
The process used for investigating a possible outbreak of infection is outlined in NHS GGC’s SOP for Outbreaks of Communicable or Alert Organisms in Healthcare Premises. This advises on the safe systems and processes required to identify and manage a potential outbreak/cluster of infections, and for convening a formal investigation into an increase in infections that can be linked by time, place and person: we make some observations about the SOP in section 8.2.1. In section 8.2.2. we look at evidence for compliance with the process.
In section 8.2.3 we provide our own critique of the HPS 2019 report. This is relevant as it addressed the recognition of outbreaks and provided guidance for future monitoring.
8.2.1 Recognising and Investigating an Outbreak: the NHS GGC Standard Operating Procedure
Our understanding of the process is that, once the possibility of an infection incident has been raised, a member of the IPCT should make an initial assessment; criteria are given in the SOP to guide the calling of a PAG[75] to further assess the situation. The framework mandated for use in the initial risk assessment is the Healthcare Infection Incident Assessment Tool (HIIAT). Whether or not the HIIAT is formally recorded at the earlier stage, the practice we have seen at NHS GGC has been for it to be completed (or confirmed) and documented once a PAG meeting has been convened.
The SOP also provides guidance for the institution of an IMT (Incident Management Team) which serves to further assess and manage the situation. Once the process is complete, a final report (in the form of a ‘Hot Debrief’ or a full IMT report) should be prepared by the IMT chair, agreed by the members and escalated up the organisation by a defined reporting pathway. Once the IMT process is complete and the report approved by its members, the SOP states that the incident should be reported on Datix.
We have reviewed the sequential SOPs during the era of our review (versions 2015, 2017, 2019) as well as that released in 2020. The SOP appears to be commensurate with the guidance published in the NIPCM[76].
The main changes to the 2017 version of the SOP included an update to organisational roles and responsibilities, as described in recommendation 16 of the Vale of Leven Hospital Enquiry[77], and the addition of a recommended agenda template from Chapter 3 of the NIPCM.
The SOP was further updated in October 2019. This update occurred at the end of the period of our review but incorporated reference to an Acting Chief Nursing Officer publication earlier in 2019[78] which reiterated guidance on ensuring robust communication with patients and their families during infection incidents and outbreaks. This revision also expanded the documentation required, stating that each meeting ‘will have an action log and a data collection tool presented at each meeting. Each agenda item will be listed in the action log and must document the discussion and rationale for each decision made. It is not enough to record actions; the relative risks and options and why the final decision was made must also be part of the documentation of the event (Civil Contingencies Act 2004)’. Whilst we saw this as welcome step to strengthen future responses to outbreaks of infection, the previous versions of the SOP had nevertheless indicated a requirement for minutes to be kept and actions to be recorded and justified.
8.2.2 Compliance with the process
We reviewed PAG and IMT documentation between 2016 and 2019 to assess the recognition, analysis, and action taken in relation to the GNE infection episodes included in our Review; and for evidence of compliance with the SOP.
8.2.2.1 Triggering an investigation. PAG and IMT reports covering incidents between 2016 and 2019 (no such documentation was available for 2015) identified the investigation of infections (relevant to our Review) caused by Stenotrophomonas, Cupriavidus and Enterobacter, whilst some IMTs were convened to address a more general increase in Gram-negative bacteraemia. Not all outbreaks which may appear relevant retrospectively were investigated at the time, and not all incidents/outbreaks progressed to IMT status.
The NHS GGC SOP defines outbreaks/incidents in line with the NIPCM. This defines a healthcare associated infection outbreak as:
- two or more linked cases with the same infectious agent associated with the same healthcare setting over a specified time period; or
- a higher than expected number of cases of HAI in a given healthcare area over a specified time period.
We note that the NHS GGC SOP does not define the term HAI which we have seen used both to mean Hospital Acquired Infection and Healthcare Associated Infection. This may be important as distinctions between the two[79] sometimes appear in the PAG/IMT records in the discussion of the significance of a reported bacteraemia. It is clear to us that the utility of the distinction offered by these two definitions is less informative in a clinical setting where, in addition to inpatient episodes, patients are attending for day care or outpatient appointments at the very high frequency seen in this patient group.
We also read accounts of discussions at IMTs where analyses prepared by different individuals were used to confirm or refute the reality of an increase in GNE infection over the period of our review.
We have reservations about the reliability of SPC charts used in this setting (although GGC followed a process as recommended by HPS). First because it is necessary to establish a prior baseline and it can be argued that the use of data for the incidence of GNE infections when the hospital was located at Yorkhill merely swaps one environmental baseline for another. Neither are we sure this is the most reliable approach when dealing with small numbers of incidents – and we note that the HPS 2019 report also advises caution in using this methodology with small numbers. We therefore also found it helpful to look at simple timelines to identify possible clusters of individual GNE bacteraemias, particularly those reported to be of the same genus/species (section 4.3). Example 8.2 provides a context for this point.
Example 8.2
There was no investigation into an increasing number of Klebsiella bacteraemias encountered between 2016 and 2018. Whilst Klebsiella bacteraemia is not infrequently seen in this patient population, and may be endogenously as well as environmentally acquired, we would have expected the evidence apparent to us for an increasing number of infections, to have triggered a formal investigative process.
Section 4.3.3 of our Report points out that, of 22[80] Klebsiella infections identified in the Review, 9 episodes (affecting 8 patients) were noted from June to November 2016; 9 (7 patients) between July and December 2017; and 5 episodes (5 patients) between January and May 2018.
We would have expected the number of infections to have attracted greater attention within NHS GGC at the time. We were informed by NHS GGC that Klebsiella spp. had been added to the list of alert organisms in 2018 but neither of the 2 Klebsiella infections seen in 2019 had had a case created on ICNet, raising concern that the alert was not active.
We perceive that part of the problem confronting NHS GGC was a relatively small number (small in relation to the overall IPC workload) of patients presented with unusual infections and our concern is that opportunities to instigate early investigation may have been missed because of too great an emphasis on ‘standard’ definitions for an outbreak.
8.2.2.2 Appropriate investigation and recording of action taken. Retrospective review of the records we received for the period within our Review did not always provide clarity that the governance and assurance required to establish an outbreak had been appropriately investigated and subsequently managed appropriately.
Root Cause Analysis (RCA) methodology was only agreed as the basis for future IMT investigation in late 2019 and applied prospectively in two patients in our Review. We have seen the template subsequently created to support RCA for bacteraemias in Haematology Oncology patients. This includes many of the data items we had identified as necessary for our own investigation. The template (appropriately, we believe) goes beyond the HPS Outbreak/Incident Data Collection Tool provided as an appendix to the NHS GGC outbreak SOP.
We found it surprising that a requirement (or even a recommendation) for the use of a structured process in line with the RCA approach does not feature in the 2019 SOP. It is difficult to understand why, given the experience of repeated GNE infection over a period of five years, this would not have been introduced earlier or more generally. We are, however, also aware that recommendations for use of a more detailed approach to the investigation of infection using RCA methodology do not feature in the NICPM[81].
We identified a consistent concern that action logs from individual IMT meetings were either not systematically created, or if they had been, were only rarely apparent to us. These were not routinely referenced within the minutes of the IMT meetings or provided to us separately. We could not identify a clear and contemporaneous record of all outbreak management actions to span the entire timeline of an IMT investigation.
We found several examples, particularly in earlier IMT meetings, where actions were not assigned, reviewed or recorded as completed or, if completed, there was any form of assurance that the actions had been sustained. Where logs were provided, we saw that some had outstanding actions for which we could find no closure. Overall, however, this improved in the incidents we reviewed from later in 2018 and 2019.
Example 8.3 illustrates a range of our concerns about: delay in escalating concerns identified at a PAG meeting to a full IMT; underestimation of HIIAT score; lack of documentation about follow through of actions agreed; failure to link to the wider context i.e. that two PAG meetings on the same day were addressing fundamentally the same problem of increased GNE infection; and the premature discontinuation of planned IMT meetings.
Example 8.3
Between 28.4.2018 and 20.8.2018, 8 isolates of Enterobacter cloacae were identified in 7 patients (one was infected twice) and including 2 isolates in separate patients on the same day.
A PAG was convened on 18.5.2018 (at this stage there had been 4 cases in 16 days). Only one patient had symptoms consistent with gut translocation and the minutes of the PAG meeting record concerns about cleanliness, ‘clutter’ in patient rooms and too many people on the ward. The latest hand hygiene combined compliance score was 85%. Surprisingly, the HIIAT only scored minor/moderate (Amber).
A separate PAG was held on the same day to discuss simultaneous concern about an increased incidence of Stenotrophomonas spp. isolates but no cross reference was made in the records of the two meetings and a separate HIIAT also scored Amber.
An IMT was not held until 29.5.2018 after a 5th isolate of Enterobacter spp. bacteraemia. The minutes record that various actions in relation to cleaning were to be implemented and a plan was made to sample drains. The HIATT score remained Amber and no further meetings were planned 'unless further isolates'.
There are further examples in 2018 where the IMT was closed or stood down despite continuing outstanding actions and with no clear process in place to continue to monitor the situation or measure the impact of interventions made.
We could find no evaluation in the IMT minutes of recommendations implemented that impacted the risk of infection to patients; for example, the installation of point of use water filters to taps and linking these to the results of water testing. This is illustrated in Example 8.4 which follows the continuing evolution of the Enterobacter cloacae outbreak already identified in Example 8.3 above.
Example 8.4
Despite the suspension of the IMT on 29.5.18, it was appropriately reinstated on 4.6.18 after swabs from drains on Ward 2A were shown to have grown a range of Gram-negative bacteria including Enterobacter cloacae, Pseudomonas aeruginosa, Sphingomonas spp., Cupriavidus pauculus, Acinetobacter ursingii and Klebsiella oxytoca. It was concluded that the recent Enterobacter spp. bacteraemias were associated with the contaminated drains.
The actions taken at this time included drain cleaning and Actichlor (chlorine containing disinfectant) treatment; filters on taps; and antibiotic prophylaxis for all children/young people with a central venous line.
Surprisingly, however, the IMT was discontinued again after meeting on 21.6.18 despite ongoing actions and did not meet again until 5.9.18 notwithstanding two further isolates of Enterobacter cloacae in July and August.
Although the parallel water review group continued to meet during this time, these meetings did not summarise the clinical situation, directly address patient management or record evaluation of the impact of interventions.
8.2.2.3 Adequacy of IMT meeting records. In reviewing IMT meeting minutes, we did not receive any supplementary appendices or microbiology reports that would have been necessary to have influenced critical recommendations. For example, we would have expected water and other environmental microbiology results to have been shared at IMT meetings in a format that allowed link to patient location, and for IPC audit reports to have been referenced and utilised in the decision-making process and risk assessments.
We pursued the issue of documentation for the IMT process in discussions with NHS GGC. Ultimately, we concluded that the records relating to each IMT meeting do not consist of a comprehensive written collation of all the information that may have been considered and/or shared at the meeting. It seems that certain pieces of information (for example, data relating to environmental microbiology results or bacterial typing) may be brought to the IMT by different individuals and are not stored centrally in the Infection Control Shared Drive as we had envisaged. We acknowledge that whilst such data may have been both shared and discussed, there is a limited audit trail of the evidence used to support conclusions made or action taken.
IMT minutes were not always easy to understand in retrospect: patients may not have been identified in a way that allowed them to be tracked across a series of meetings; staff were not always identified by their role, making it difficult to see if the attendance was appropriate in terms of relevant expertise; the structure of the documents varied and the style was sometimes informal. A short, written assessment of an IMT record dating from July 2019 and taken from our own records of a Panel meeting, is shown in Example 8.5.
Example 8.5
(This case was) Not specifically identified at IMT meetings, and minutes are not precise; for example (IMT) on 3.7.2019 identifies 6 Gram-negative bacteraemias in Ward 6A but these isolates are not dated or named and minutes go on to state “All Gram-negative bacteraemia have unique strains. This rules out cross transmission between staff/patients but not from water/drains which has tested positive for the organisms”. No detail of samples/results from water or drains is given.
These IMT minutes include a statement about the implications of typing results which does not seem correct and our comments illustrate the difficulty we had in linking IMT records to individual patients and to investigations undertaken.
In respect of investigations, we found it difficult to understand how requests for environmental samples were consistently agreed, implemented and reported to inform IMT discussions.
Example 8.5 (above) suggests that the IMT did not record results of environmental samples taken yet we know that this meeting referred to at least one of a series of 8 isolates of Enterobacter cloacae that occurred in 7 patients from 15.1.19 to 31.12.19. We have also ascertained from the data we received that there are no records to show any ‘hard surface’ (including drain) samples were positive for Enterobacter spp. in 2019. Water samples positive for Enterobacter spp. in the same period were identified from an anaesthetic kitchen and basement water tank on 27.3.2019 and from toilets in 3 patient rooms in Ward 6A on 24.6.2019. The ward samples were obtained within 12 days of two patients with Enterobacter cloacae bacteraemia although there was no co-location with the rooms in which these patients had been nursed. This possible connection was not documented in the IMT minutes.
8.2.2.4 Upward reporting from IMT meetings. We have seen no ‘Hot Debrief’ or full reports at the close of a series of IMT meetings relating to cases included in the review despite this being mandated in the GGC outbreak SOP. Examples of such documents have however been provided to us from IMTs in other clinical areas within NHS GGC, raising questions about consistency in practice across the organisation.
The SOP also indicates that these reports should be signed off by members of the IMT and sent to the Acute Infection Control Committee from which upward reporting to the NHS GGC Board is expected. There is little or no documented evidence that IMT members were asked to approve such reports.
Whilst it is evident from NHS GGC Board papers that reports about the problems encountered within Wards 2A/B, and subsequently 6A, were provided at Executive level, we are concerned that the significance and scale of what was happening may not have been adequately expressed. Example 8.6, describes a HAIRT (Healthcare Associated Infection Reporting Template) report made to the NHS GGC Board at the first meeting held following the death of a child after GNE bacteraemia[82].
Example 8.6
“Two cases of …..bacteraemia were identified over an 8- day period….. A Problem Assessment Group (PAG) was held ….. HPS were notified and a Healthcare Incident Infection and Outbreak Reporting Template (HIIORT) was completed. No further cases were identified and the two cases were later confirmed to be different types”.
We do not understand why it was important for the Board to hear that there had been two infections, that they had been appropriately reported and that they were considered to be of different types but not to be told that one of the children had died. We have since been told by NHS GGC that these infections and the death were reported as far as the Board Infection Control Committee but that, as the Board is a public meeting, there was a need to ensure awareness of infections but no requirement to discuss individual patient details (for patient confidentiality and Data Protection reasons). However, we note that the occurrence of another bacteraemia, caused by the same organism, earlier in the same year, following which the child also died, was not reported to the Board. It is not clear to us what was or was not expected to be reported to the Board. We conclude this shows an inconsistency in the process and purpose of reporting and may represent an organisational culture which promotes a focus on process (i.e. that a report was received) rather than being clear what the cause or consequences were.
8.2.2.5 Clinician concern. We noted that the there are occasions when the minutes record that clinicians present at an IMT meeting directly questioned if the environmental risks had been reported to senior management within NHS GGC (this was mainly in 2018 and 2019 and while there is an unsubstantiated suggestion that this could also have been in 2017, the Panel have not seen written evidence for this). It was interesting for us to hear, at a meeting with RHC clinicians in February 2020, the IMT process described as ‘lacking integration and fails to recognise patterns’. This simple statement reflects the overall impression of the Panel.
8.2.3 Review of NHS GGC Paediatric Haemato-oncology data (HPS October 2019)[83]
The context for the report is that, having supported NHS GGC in dealing with cases of blood stream infection in patients in Wards 2A and 2B, associated with concerns about the contaminated water supply in 2018, HPS were asked to assist when concerns emerged about a suspected increase in Gram-negative environmental (GNE) bacteraemias in patients on Ward 6A during the summer of 2019.
We had not intended to provide a critique of this report as we saw it as one of a number of previous investigations, the results of which should not influence our own. However, its significance loomed large in our discussions with NHS GGC and we have therefore added this short section summarising our view of the reports findings.
The aims of the report were to describe any differences in the datasets being used to explore the situation; to review the GNE infections; and to identify if there had been a change. The principal methodology used was the creation of Statistical Process Control (SPC) charts which were used to explore the data collected from July 2013, before the move of patients to the new site at QEUH/RHC, until September 2019. Changes in hospital activity data for the Paediatric Haematology Oncology service were explored in parallel and, finally, comparisons were made between data for the whole of RHC, for the period June 2015 to September 2019, with similar data for the Royal Hospital for Sick Children, Edinburgh and Royal Aberdeen Children’s Hospital.
In summary, the report identified periods at which there were upward shifts, trigger points (above the Upper Warning Limit) and outliers (above the Upper Control Limit) in the SPC plots of bacteraemia identified since the move to the new hospital. Overall, however, patterns showed no consistent trend. There were also differences between NHS GGC and the data from Edinburgh and Aberdeen. This showed higher rates for environmental with enteric bacteria over the whole time period at NHS GGC, but lower rates for Gram-positves and no difference for Gram-negatives and environmentals alone. Various subgroup analyses showed no consistent message.
As far as we are able to ascertain from our own assessment of the data presented in the report, we agree: a) that the dataset used was providing an accurate reflection of the situation at NHS GGC; b) that there were episodes of variation in the SPC data (the latest occurring in September 2019) but that this alone did not provide clarity about its cause or significance; and c) that the caution expressed about small numbers in the analysis of some subsets of the data, is justified.
We do not see that this report would have provided any clear message of either reassurance or concern about past events. Nor do we see that it offered a clearly interpretable and favourable comparison with other Scottish children’s hospitals (not least because the size of the paediatric haematology oncology services in these three hospitals varies very substantially – NHS GGC being easily the largest).
From our perspective, the most useful output of the HPS report lies in the clarity of its recommendations for the future, some of which align with our own. We would particularly emphasise the points made that, going forward, interpretation of these data requires the systematic collection of clinical data; must be set in an environmental context; and requires continual monitoring. NHS GGC accepted the need for the ongoing monitoring.
8.3 Microbiology and IPC information systems
We have already discussed issues over our access to the Telepath and ICNet systems (section 8.1.3). In this section we discuss the constraints encountered in using the systems and focus on two particular issues: the challenges we experienced in accessing and interpreting data on bacterial typing; and the concerns we identified about the alert system used for ICNet.
8.3.1 Telepath and Bacterial Typing
The Telepath LIMS is used across all laboratories within NHS GGC. The system provides listings of all microbiological samples, detailing the laboratory processing and results for these, in addition to a Patient Note Pad (PNP) option for a given patient, which allows microbiologists to record free text information related to any positive isolates/infection episodes of key interest (which typically includes isolates from sterile sites, including blood cultures). The PNP is also used to record information obtained from communications with ward based clinical teams, and any advice provided to these.
We found that the PNP generally provided very good evidence of frequent engagement and information sharing between the microbiology and ward based clinical teams, including recommendations for choice and duration of antibiotic treatment, based on laboratory derived susceptibility testing, and associated infection management (for example, removal of sites of infection such as intravascular catheters), and follow on diagnostic sampling/testing. This information was helpful to us in understanding more about the nature of the infections we reviewed and their management.
Notably, however, the Telepath system did not systematically offer the basis for recording the results of typing bacterial isolates (mainly derived from reports provided by the Public Health England reference laboratory at Colindale, London but some data also from the Scottish Microbiology Reference Laboratories), either by annotating the original specimen results page or within a patient’s results at a later date (when the typing information was received).
We found that typing results were also not routinely entered into the PNP, although some results were referenced and, where so, the results were most frequently reported as ‘unique’. Some were also referenced with the statement that a full report could be found on the Clinical Portal (the electronic clinical patient records system). We were able to access the typing results on the Clinical Portal, but these reports were similarly vague, reporting isolates as ‘unique’ but without any crucial context of which bacterial strains it had been compared with (what strains, their origin and how many other strains?).
Discussion with NHS GGC about bacterial typing revealed that, hitherto, there had been no electronic database of typing results. Generally, results from PHE Colindale had been received as pdf documents which were filed as such, either in paper form or, more recently, electronically. Consequently, the organisation had no ability to search a database in order to relate potentially linked bacteria whether these came from a patient or the environment. Useful linkage searches would involve several items of data about the bacterial isolate: the date it was obtained, the patient sample or environmental site from which it derived, and the physical location within the hospital environment from which it was obtained.
This is precisely what we had hoped we might have been able to achieve to support our Review and we were surprised that, despite over five years of experience with outbreaks of GNE bacteraemia and concerns about the hospital environment, a database with this functionality had not been created by the time the Case Note Review had been commissioned. It appeared from our discussions with NHS GGC that work had commenced but a considerable amount of work was needed for staff to collate information held in different systems in order to provide us with the data we requested.
Most of these data were not received by us until December 2020. Databases identifying bacterial typing by year from 2015-2019 were supplemented by additional data relating to more sophisticated analyses using Whole Genome Sequencing (WGS) methodology in specific types of bacteria. The year-related databases were very large and appeared to include all typing done within NHS GGC for that year, i.e. for patients of all ages, at all clinical sites and involving many different clinical samples other than blood cultures. It was not clear, even in 2019, that all isolates from patients within our Review had been typed but, in general terms, we were not able to ascertain evidence of a direct links between bacterial isolates obtained from children in our review and other specimens.
However, the number of environmental samples in these databases were limited. For example, the 2019 database (being the database we assumed would be most likely to be complete) listed almost 550 samples but included only 6 water samples, of which 3 could not be typed. There were approximately 140 other samples from environmental sites but none had complete location information rendering it impossible to relate to sites of patient care.
WGS is the state of the art fingerprinting method for the comparison of microorganisms. Its strength lies in its ability to help determine how closely microorganisms are linked. However, the interpretation of WGS derived data in linking bacterial isolates has significant challenges given the way the genetic code evolves/mutates. Differences between microorganisms can be measured as SNPs (Single Nucleotide Polymorphisms) each representing an individual DNA building block. There is a risk that defining difference by an absolute number of SNPs (for example, by saying anything more than a 25 SNPs difference is not significant when comparing two samples of the same bacteria isolated from different patients/places) may result in an oversimplification. It is likely that bacteria found in environmental locations may exist as multiple types and it may best to say that whilst the demonstration of a close relationship between a patient specimen and an environmental isolate of the same bacteria is strongly indicative of a relationship, the reverse does not necessarily apply.
The WGS was carried on three groups of isolates: Enterobacter spp., Stenotrophomonas spp. and Cupriavidus spp..
The Enterobacter spp. (n=42) comprised 36 clinical/patient isolates and 6 environmental isolates. However, isolates from 5 of the children with Enterobacter spp. bacteraemia were not included. Similarly, the records of water and surface sampling show a total of 25 Enterobacter spp. isolates during the review period, and thus approximately three-quarters of these were not included in the WGS exercise.
The Stenotrophomonas spp. (n=84) included n=15 isolates from patients in our Review, 10 from other patients and 59 environmental strains, 11 of which were from 2020. Five children in our series with Stenotrophomonas spp. bacteraemia were not included.
There were 263 isolates of Cupriavidus spp. from water or surface sampling in the review period but only 18 samples were included in this exercise. As far as being informative for the Case Note Review, this included one patient from Ward 2A with a sample dated 25.2.18 (which doesn’t match the date of infection for either of the patients with Cupriavidus in the CNR) and 7 environmental samples from ward 6A taken on three dates 18.11.2019, 7.1.2020, 14.1.2020. This is far from an adequate sample to exclude an environmental source.
NHS GGC told us that it was possible to definitively link the environment to infection in only two patients; firstly, in 2016 when Cupriavidus spp. was identified in the Aseptic Dispensing Unit[84], and secondly a case of Mycobacterium chelonae included in our Review. However, we concluded from our investigations (above) that there are too many gaps in terms of which isolates were included (alongside the inconsistent environmental sampling – Chapter 5) to be able to interpret the true extent of relatedness between patient and environmental isolates from these WGS results.
8.4.2 ICNet and IPC Alerts
The ICNet system relies on data being exported from Telepath at regular intervals. If a microorganism is identified as one of a pre-defined list of ‘alert’ microorganisms, it will automatically create a ‘case’. This case will alert the IPC Nurse/Team responsible for that hospital site, who will then review the situation, ascertain if there is an infection risk to the clinical area or patient population and advise on the appropriate care for that patient.
The IPCN is then required to complete a question set, which will determine if the infection is hospital acquired for the purposes of local surveillance. The questions also confirm what written information should be provided for the alert microorganism such as care plans, care bundles or patient information leaflets. Following the initial assessment, the IPCN has the opportunity to close the case if no infection risk is identified, or to keep the case open to monitor the patient’s condition until they are discharged or no longer an infection transmission risk. There is a patient notes function within the ICNet system, which allows IPCNs to record any communication with the clinical team or microbiologists. NHS GGC/IPCT policy is that patients with open ICNet cases should be reviewed weekly as a minimum.
The NIPCM provides a nationally agreed minimum list of alert organisms/conditions; this informs NHS Boards of those alert organisms/conditions that may require further investigation. The guidance states ‘The list is not exhaustive and specialist units, for example those managing patients with cystic fibrosis, will also be guided by local policy regarding other alert microorganisms not included within these lists.’
As part of our review, we assessed information provided to us from ICNet and identified whether cases were created or not. We found little evidence, even as late as summer 2019, that the GGC alert list had been modified in light of the evolving experience with bacteraemias caused by Gram-negative environmental infections. This resulted in frequent absence of alerts being triggered within ICNet and the subsequent absence of IPCN input into cases under our review. Example 8.7 provides brief details of two different situations.
Example 8.7
a) In late July 2019, a patient presented with an Enterobacter cloacae bacteraemia.
This was the seventh isolate of this organism in the Paediatric Haematology Oncology population in 6 months. An IMT had been initiated in May because of concerns about the frequency of this type of bacteraemia (see also Example 8.2) but no alert was raised in ICNet for this next case despite the previous experience. Why?
Although NHS GGC told us that Enterobacter had been added to the alert list in 2018. However, we reviewed 8 episodes of Enterobacter spp. bacteraemia in 2019 and none had an alert created in ICNet. This suggests that the alert was not active.
b) In late September 2019, a patient presented with bacteraemia associated with Achromobacter spp. which is a particularly unusual bacterium.
In this case, however, an alert was triggered on ICNet, not because of the specific nature of the bacterium, but because the system had by then been adjusted to trigger an alert should two or more positive blood cultures be reported on Ward 6A within 14 days.
This coincided with a period of great concern about the safety of Ward 6A and limitations being placed on admissions. Why was this change not implemented previously?
We have heard that requests from some microbiologists for the list of microorganisms on the ICNet alert list to be augmented were not heeded.
We understand, however, that when cases are not identified by alerts in ICNet, there is still capacity within the system for IPCNs to manually create a case for any patient - if they are alerted to the identification of a microorganism of concern. We have been told that some microbiologists did make direct contact with IPCNs to alert them in this way and under certain circumstances, but we have also seen evidence that Infection Control management within NHS GGC sought to discourage this. This seems entirely inappropriate as it would have excluded the IPC nurses from the management of some GNE infections at NHS GGC, which, at the very least, would have limited wider awareness of the problem.
Overall, however, our observations suggest to us that the communication between microbiologists, the infection control doctor and IPC nursing team is not as robust or systematic as it should be. The teams often appear to work independently and communication between these staff groups appears to occur on an adhoc basis: referral of patients with alert organisms on the basis of an automated electronic process (where it happens) is not direct communication.
8.4 Clinical records
This commentary is based on the experience of reviewing the health care records for 83 patients with 117 episodes of infection[85]. It highlights the challenges we experienced in extracting relevant information from the case records, focusing particularly on inpatient medical records.
8.4.1 The Clinical Portal
The Clinical Portal is the web-based application that presents patient clinical data from various NHS clinical systems. It is widely accessed by a range of medical, nursing, AHP and administration staff, as well as by GPs and other Health Professionals, and has largely replaced paper-based case records at most NHS Scotland locations.
In general, the review team found that the medical and nursing care for each patient was identifiable in the Clinical Portal, and was recorded routinely and reliably on a day to day basis. The challenge, however, was locating the specific information required as there are wide variations in the way that parts of the clinical record are scanned into and filed within the Clinical Portal.
Daily recordings of In-patient medical care were found in 3 different areas in the Portal:
- written and scanned in a sub section tab of Clinical Notes detailed as “In-patient Medical Notes”
- embedded in the “Nursing Assessment” tabs on a generic continuation sheets continuous with the nursing records and not necessarily recorded as a medical record of care
- digitally recorded in the “Clinical Notes”
Nursing care is reliably recorded and stored in the nursing assessment tabs. These records are exemplary with dated, signed entries of the elements of care recorded. In particular, standardised elements of care (for example CEWS[86] and CVC/PVC bundle[87] care) are reliably recorded in the dedicated record segments.
Medications are recorded in a wide range of documents/places within the medical and nursing records in narrative form when administered or considered for change as instructed by medical staff.
All laboratory results are reliably entered into the associated test carried out under the separate laboratory headings.
When a procedure was undertaken, such as the insertion or removal of a central line, the information was usually recorded in dedicated records for “Interventions” under the sub tabs of “Anaesthetics” and “Operation Notes”. In some cases, the records for the same procedure were not dated correctly or signed. Mentions of the procedures/interventions are also recorded in the medical and nursing records.
Admission and Transfer information was embedded in the nursing and medical records and in the “Patient Notes” sections. Transfers of care within the hospital system are difficult to identify, as Medical PICU admission and discharge summaries were often scanned and embedded within nursing notes within the Nursing Assessment section; and not all patients had an immediate discharge or final discharge letter prepared and stored.
It was challenging to find all components of the records, although knowledge and frequent use of the system enabled easier navigation of the anomalies. Some records were scanned in long sections, representing one document with a variety of records within. Some records were scanned in with dates many months or years after discharge. Scanned records for each episode did not necessarily have the correct care episode date. Scanned pages within the records, particularly for patients with extended in-patient stays and/or multiple episodes of care were often the most problematic. We found that many cases had pages of the records scanned in reverse order and had multiple admission episodes within the same scanned document, and not necessarily in date/time order.
8.4.2 Inpatient Medical Records
We focused on an analysis of in-patient medical records - both the scanned hand written records and the digital notes - as these related directly to the management of the bacteraemia.
8.4.2.1 Scanned Hand Written Notes. For the 117 infection episodes, we found completed written notes for 76 (65%), incomplete notes for 22 (19%) and no written notes for 19 (16%). Only 60% of the written notes were filed under the date of discharge; others were filed up to 14 months after the date of discharge.
Standards varied to a considerable extent. One patient, who experienced multiple episodes of GNE infection, had 906 pages of hand written notes covering 418 days of admission, which were complete, in order and with no irrelevant information. In contrast, another patient had 139 pages of hand written notes covering care after a GNE bacteraemia, but many of the pages were undated or were not filed in chronological order; the notes commenced one week after the bacteraemia and contained very few details regarding the clinical management of the bacteraemia itself. However, further hand written medical notes with critical information about bacteraemia management, including discussions with parents, were found filed in the nursing records.
8.4.2.2 Digital Notes. Digital medical records may be filed in three separate areas within Clinical Notes - Generic Continuation, Patient Notes and Pharma Care Plan. When filed under Generic Continuation, notes were not linked to specific admissions and contained diverse inpatient and outpatient records from a range of clinical disciplines and specialties. When Generic Continuation records were labelled Paediatrics, we found those to contain digital inpatient medical notes. These were detailed and fully electronic, which enabled word searching but might cover several admissions.
Patient Notes were labelled by medical (e.g. Haematology) or AHP (e.g. Dietetics) specialty. Most Patient Notes were outpatient contacts covering a clinic appointment, home visit or telephone call. However, there were some notes about inpatient contacts.
Pharma Care Plan notes are stored within a standardised care plan exclusively recording information about medicines.
We found digital notes (to any degree) for a minority (37%) of episodes. There was no trend to show the increasing use of digital records over time suggesting that there was no planned evolution to full digital record keeping over the period of the review.
8.4.3 Completeness of Inpatient Medical Records
Overall, we were able to locate complete inpatient medical records for 111 (95%) of all episodes. However, only 46 (39%) of all episodes had complete medical records filed by the date of discharge for the episode concerned. Finding medical records for 61% episodes required searching through written records for up to 14 months and digital records for up to 35 months after the date of discharge for the episode.
Both written and digital notes were found for 28 (24%) of 117 episodes, but these were not duplicate records and sometimes included separate, important information about the same day of the episode. For example, inpatient medical notes for one patient were correctly filed under the date of discharge but in fact only contained records for 2 of the 18 days of the admission: records were ultimately identified for every day of this admission but were filed within different areas of the Clinical Oncology, Haematology and Paediatrics Patient Notes sections of the record. For another patient, we found inpatient records relating to a single 24 hour period in three different locations in the clinical portal system.
We found no written or digital medical notes for three episodes
8.5 Patient location records
The locations of patients during hospital attendance and inpatient stays were obtained from TrakCare, the Patient Management System used by NHS GGC. All patient episodes (Outpatient, Inpatient and Emergency) are recorded and managed on TrakCare. In the course of our Review, we found that a specific bed was identified for almost all inpatient stays, but the system did not provide location (to the level of a specific bed space) when patients were receiving day care in ward 2B or, subsequently, in ward 6A. This limited the sensitivity by which we could assess location of care as a risk factor for infection.
One singularly unexpected issue was the coding of Haematology Oncology Day Care patients as attending Ward 2B after the date on which both wards 2A & 2B had been closed in September 2018.
This occurred inconsistently within individual records; although we were made aware that ward 2B was used for the RHC pre-assessment service from 29.4.2019 to 15.11.2019, we have been assured that no Haematology Oncology patients attended that area during this period. It seems self-evident for the benefit of tracking purposes that patients should never be coded to an area other than that to which they physically attended.
It was also often difficult to identify from the clinical records in which operating theatre surgical procedures took place. It also seems likely that procedures (e.g. bone marrow sampling and lumbar puncture procedures) were undertaken in anaesthetic rooms, also without a record of the location.
Attention needs to be paid to the accuracy with which patient location is defined, should a review of this kind be required again, or if support to an internal investigation of linked episodes of infection is required.
8.6 Adverse Event Reporting
We have already discussed data derived from AE reporting, whether from the PTT or Datix notifications, in Chapter 6, section 6.4. In this section, we compare AE rates in the Haematology Oncology patients we have reviewed at NHS GGC with data available from the literature from other paediatric hospitals, and offer some further reflections about issues we have identified.
8.6.1 Comparison of AE rates at NHS GGC with other paediatric hospitals
A literature search identified 11 studies involving 15,153 paediatric inpatients from 104 hospitals in five countries (Argentina, Canada, Norway, the UK[88] and the USA). This is summarised in Figure 8.1. These studies only included data from randomly selected patients but, for comparison, the event rate at NHS GGC was calculated using the PTT data from the 45 inpatient episodes with one or more AE that were not related to the management of infection in these 117 admissions. The proportion of patients receiving intensive care was calculated using all PICU admissions.
The reported AE rate ranged from 7% to 74%, but much of this variation can be explained by the different settings in which data were collected (shown in Figure 8.1) which confirms that: the highest AE rates were in the two studies that only included patients from ICUs; studies that only included tertiary care hospitals had higher AE rates; and in tertiary care hospitals, a higher proportion of ICU patients was associated with higher AE rates.
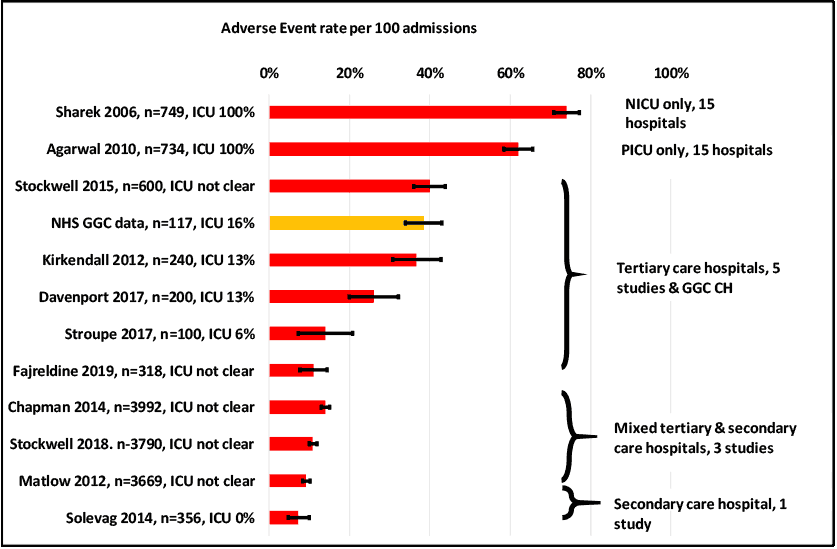
Bars show 95% CI of event rates. The numbers for each study are the total number of admissions and the % of admissions that included admission to the ICU.
Eight of these studies used the NCC-MERP classification of harm[89] to assign severity to AEs; this is also the classification used in the UK PTT. The median proportion of Category I events from those studies was 11%, range 2-22%; in comparison, 5% of AEs at NHS GGC were category I.
Appendix C shows the PTT score sheet used in our Review. For ease of analysis, the adverse events that derive from searching for these triggers can be grouped as shown in Figure 8.2.
| Biochem | Biochemistry | Intervention for increased creatinine; high/low potassium, sodium, sugar |
|---|---|---|
| Comps | Care complications | Intervention for tissue damage, thrombosis, other complication (e.g. adverse drug reaction, central line infection) or pain |
| Deter | Deteriorating patient | Delayed response to Early Warning Score; intervention for cardiac/respiratory arrest, hypoxia or hypovolaemia |
| Haem | Haematology | Intervention for anticoagulation, anaemia, thrombocytopenia or neutropenia |
| Infection | Infection | Intervention for infection causing admission or occurring >48h after admission, bacterial or fungal |
| PICU | PICU | Unplanned transfer to PICU |
| Meds | Medication | Intervention with naloxone, chlorpheniramine, glucagon; unplanned anti-emetic; interruption of planned treatment |
| Surgery | Surgery | Returned to theatre for unplanned procedure |
| Transfer | Transfer to/from hospital | Readmission, unplanned admission, delayed discharge |
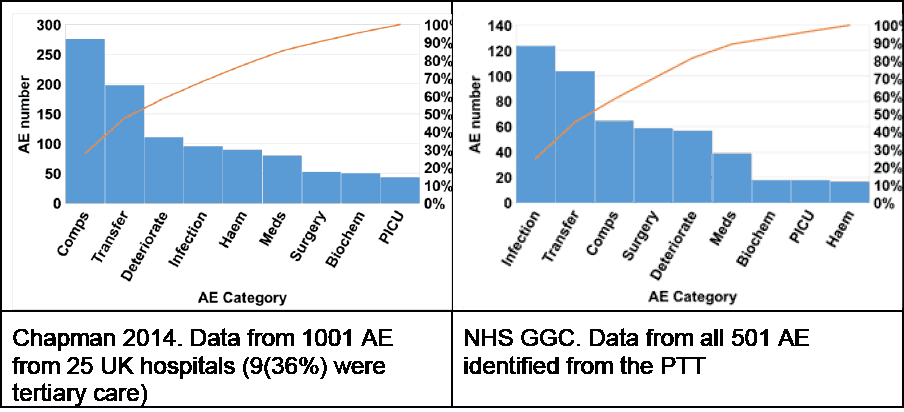
Chapman 2014. Data from 1001 AE from 25 UK hospitals (9(36%) were tertiary care)
NHS GGC. Data from all 501 AE identified from the PTT
The NHS GGC data are dominated by AE classified as Infection and Transfer, which is much as would be expected from the nature of the group selected. It is reassuring, however, that although nearly one third of ‘Deteriorating patient’ AE in the Chapman study were caused by failure to do or to respond to Early Warning Scores, this was not identified in any of the NHS GGC episodes. In contrast, 74% of these AEs in the NHS GGC data were in patients who were given fluid resuscitation on the ward in response to symptoms of their bacteraemia – in itself, this is indicative of the serious nature of such infections.
8.6.2 Learning for the future
Using the data derived from the PTT to identify AE, we saw that most were related to the appropriate management of the serious infections under our Review. The analysis illustrated in Figure 8.1 suggests that overall AE rate in this population of patients at NHS GGC is comparable with reports from other tertiary care hospitals. It was clear to us, however, that Datix reporting significantly underestimated AE rates and that individual AE were sometimes incorrectly classified and under scored for their significance (section 6.4.2). We also have concerns about their identification and suggestions for learning from incidents.
Only one of the Category I events identified via the PTT was reported as risk level 4/5 on Datix; and there was only one other risk level 4/5 incident reported on the entire patient cohort from 2015-2019. The NHS GGC Incident Management Policy is clear that these events should be reported on Datix and “will be considered potential Significant Clinical Incidents and subject to screening using the appropriate tool to support decision making as to whether the incident should be confirmed as an SCI. Our data suggest that this was not done.
Of the 17 episodes with one or more Category I events, 14 included a PICU admission. The only exception was a patient who was resuscitated for sepsis on the ward but did not require PICU admission. Therefore, 94% of the patients with Category I events could have been identified from routine data (PICU admission within 28 days of their bacteraemia). This illustrates a way to use routine data to identify patients for review. Other opportunities to use routine data to identify Category I events might include, for example, deaths within 7 days of stem cell transplant or within 30 days of chemotherapy.
Our analysis suggests that Category II events will occur in 20-40% of children in tertiary care, but we are not clear how incidents are selected for reporting, review and audit within NHS GGC. An advantage of looking for AE in random samples of patients is that it provides a systematic approach to the identification and classification of events in an unselected setting. In addition, reviewing a random sample of patients rather than starting with an incident provides a better opportunity to identify and feedback on good practice. We recognise, however, that many of the episodes we have reviewed are of very long duration, and so consideration could be given to focusing such reviews on a limited period within an admission (for example, within 28 days of admission or 28 days of a bacteraemia).
8.7 Morbidity and Mortality Reports
In Chapter 6, we have looked at the characteristics of the 22 children and young people included in our review who had died by the time of the publication of this Report (section 6.3). Cause of death was assessed from the clinical records in all cases, and validated from death certificates in the 19 cases for whom these were available.
We were provided with 17 Morbidity and Mortality (M&M) reviews (also known as Paediatric Review and Assessment Meetings (PRAM)), all from amongst the patients who had died. Two of these reviews were about patients who died and where the infection was attributed, at least in part, as the cause of death and two others were deaths within 28 days after discharge following an infection episode.
In one of these four patients, both the PICU M&M review and the cardiac death audit referred to the infection which was also recorded as a contributory factor on the death certificate. At the request of NHS GGC management, an additional review was undertaken of this patient by a paediatric intensive care consultant, over two years after the child died. The death had been correctly reported on Datix as an Extreme incident.
The other three M&M reviews of deaths that related in time to infection episodes were all initiated by clinical staff. None of these reviews included a discussion of the Gram-negative environmental infection but they did identify other significant discussion points, including death within 30 days of chemotherapy and the very large resource implications of transferring a ventilated patient from PICU to another hospital for other treatment. None of these issues were reported on Datix and the M&M reports do not include action plans.
Much of the content of the other M&M reports related to the chronology of the patient’s underlying disease, its treatment, and to aspects of end of life care. There was no reference to Gram-negative environmental infection. The M&M reports we have seen were limited to patients who died, but the Scottish Mortality and Morbidity Programme[90] clearly states that such reports should include review of care complications in addition to patient deaths.
Some of the M&M reviews were presented by Specialist Trainees. Audit and quality improvement are Outcome 8 in the RCPCH Paediatric Training Curriculum, and this is one of nine areas for assessment of applicants for Specialist Training, with clearly described indicators of involvement in audit/quality improvement and learning from this[91],[92]. We could not identify a systematic approach to how the use of incident reporting or M&M review was used either to improve patient care or to provide professional learning. Some of the M&M reviews clearly identified important issues. If these were cross referenced to, or entered as reports on Datix, this would create an opportunity to engage more widely with the organisational response and in creating action plans and auditing improvement.
8.8 Central Venous Line Care
We have looked at aspects of central venous line (CVL) care in the patients in our review. CVLs, like other indwelling medical devices, present a clear risk for infection but are intrinsic to the delivery of many aspects of the complex care required by children and young people undergoing chemotherapy or treatment for other serious blood diseases.
We assessed central line care in 81[93] patients who had 115 episodes of central line associated infection that were treated as an inpatient in the GGC Paediatric Oncology Unit. We collected information from written and digital inpatient medical and nursing records, and from the Patient Note Pad section in Telepath.
8.8.1 NHS GGC policies
The antibiotic policy for Paediatric Haematology Oncology patients with febrile neutropenia incorporates detailed recommendations about antibiotic treatment and addresses aspects of CVL usage in the context of presumed line related infection. This has been regularly updated from v1.0 dated 2010, to v4.0 dated March 2020. The policy includes a recommendation to document the central line insertion site in febrile patients and cautions that if a child deteriorates with flush or continuing use of the line, consideration should be given to siting a peripheral cannula and discontinuing use of the line, with further consideration given to adjusting the antibiotic regimen. There are otherwise no specific recommendations for resting, removing or challenging lines.
The Patient Note Pad notes in Telepath frequently state that microbiology advice is based on evidence from the IDSA (Infectious Diseases Society of America) guidelines on management of intravascular catheter related infection[94]. Overall recommendations for GNE bacteraemia in patients with long term catheters are:
- If the line is removed, treat with 7-14 days antibiotics.
- For line salvage, use systemic and antibiotic lock therapy for 10-14 days.
Specific recommendations from the IDSA guidelines provide more detailed advice about criteria for line removal, treatment without line removal and about management of infection in paediatric patients. The IDSA guidelines do not mention line challenge.
8.8.2 Observed CVL management
The appearance of the line site was recorded when the patient became symptomatic in 94 (82%) episodes and was documented as clean in 84 (89%) of these records.
The line was rested in 51 (45%) episodes and subsequently challenged in 21 (18%) episodes. Signs and symptoms of sepsis occurred after 9 (43%) of those line challenges and, in one case, resulted in a patient who experienced rigors, became cyanosed, tachycardic and had limited response to bolus fluid infusion, being admitted to PICU.
The Chief Nurse for Paediatric and Neonatal Services at NHS GGC provided us with this information about central line challenges: “Challenging the lines was a rather historic practice where if a child had a pyrexia they would stop using the line, insert a cannula and use that, then a few days later ‘challenge’ the line by taking more blood cultures and flushing, gradually using for fluids and medications. This practice was discussed at the QI group (set up in May 2017) and we worked from there towards a change. Microbiology and other representatives within the group agreed to continue to use a line or remove a line depending on the clinical and microbiological status of the child”.
However, the frequency of line challenges did not appear to reduce with time and was identified in 5 (14%) of 36 episodes occurring up to May 2017 versus 16 (20%) of 79 episodes from June 2017 onwards. The latest line challenge we noted in our Review was for a bacteraemia diagnosed in March 2019.
Patient Note Pad notes do not document any microbiology concerns about plans to challenge the line where this is explicitly mentioned.
Data in section 6.2.3 looks at the removal of a CVL in response to GNE infection. This occurred in 78 (68%) of episodes. We found that the PNP notes consistently recorded when line removal was considered to be the optimal microbiology advice but, recognising this was not always clinically optimal for continuing patient management, when line salvage was attempted, there was regular advice from microbiology about systemic and antibiotic lock therapy, with frequent reference to IDSA guidelines. We were, however, concerned to see that when a decision was reached to remove a line, there were delays in its implementation. We were not able to investigate this in detail but recognise that this may be a consequence of competing priorities for operating theatre and anaesthetic time. Nevertheless, we believe that delay in removal of an infected line carries risk and so that removal should be prioritised accordingly.
8.8.3 Conclusions
We have seen that CVL care was well documented by the nursing staff and that good advice was provided by the microbiologists in the context of bacteraemia. We acknowledge that considerable work was being undertaken within NHS GGC during the period of our Review to reduce the incidence of central line associated blood stream infections (CLABSI) through a Quality Improvement framework. We are, however, concerned both about the practice of ‘line challenge’ and the lack of documentation in the medical records when attempts to continue to salvage a line were preferred over advice from microbiology to remove it.
Episodes of central line associated bacteraemia present an opportunity as much to learn from its management as from the analysis of its causation.
8.9 Other aspects of clinical care
Two other issues have arisen in our review that we discuss briefly here.
8.9.1. Antimicrobial prophylaxis
The prophylactic (preventative) use of antibiotics, antifungal and antiviral drugs to reduce the risk of infection in patients who are at high risk by virtue of their disease and/or treatment is well established in Paediatric Haematology Oncology care. The evidence base varies according in relation to diagnosis, treatment and age. In practice, consistency is often addressed by guidance incorporated within established treatment schedules and clinical trial protocols.
Concern about the incidence of GNE bacteraemia at NHS GGC raised an understandable question for the clinical and microbiological teams about the use of antibiotic prophylaxis (i.e. whether its use should be extended beyond the settings in which it would normally have been considered). The use of fluoroquinolone antibiotics is a particular focus because of concern that this can contribute to selection of antibiotic resistance and to the risk of Clostridium difficile infection. Its use in the context of preventing neutropenic sepsis has recently been reviewed by the National Institute for Health and Care Excellence[95], but whether use of fluoroquinolone prophylaxis is useful in a setting where there is concern about a possible environmental focus for infection is unclear. Furthermore, once a policy of this kind has been initiated, it is understandably difficult to know when to de-escalate.
We are not critical of the use of fluoroquinolone prophylaxis in this context and recognise from what we have since been told that the matter was carefully considered at the time. We note that the continuation of its use was reviewed in an SBAR written by Dr Andrew Murray, Medical Director, NHS Forth Valley and Co-chair, Scottish Managed Service Network for Children and Young People with Cancer in December 2019 (on behalf of the Oversight Board).This concluded that the continuing use of fluoroquinolone prophylaxis should be on the basis of individual patient assessment; no indication was given for criteria against which such individual assessment should be effected but consensus guidelines for the use of antibiotic prophylaxis in Paediatric Haematology Oncology practice have recently been published and should be reviewed for their use in NHS GGC[96]. We have not sought information about audit of ongoing use of antibiotic prophylaxis but best practice would anticipate this is being undertaken.
8.9.2 The impact of the organisational response on the delivery of clinical care
In Chapter 6 we have tried to share data we obtained or derived from our Review in order to demonstrate the impact of GNE bacteraemia on individual patients. We were less able to form a view of the overall effect on the clinical service although it was obvious that disruption was substantial, particularly in relation to the decisions to close Ward 2A and 2B in September 2018, to move patients out of Ward 6A for a short period at the beginning of 2019, and to limit admissions to Ward 6A in the summer and early autumn of that year.
Throughout our Review we had not seen any document prepared by the clinical team, by NHS GGC management or by the Managed Service Network that set out an analysis of how these decisions affected the overall delivery of Paediatric Haematology Oncology care. Measures that would have been of interest are, for example, timeliness in delivering planned chemotherapy; deferral of planned treatment (e.g. surgery, radiotherapy, stem cell transplantation); use of shared care; and transfers to other units.
We questioned the availability of evidence of this kind at a meeting with the Haematology Oncology clinicians in December 2020 and have since seen two documents. One is an audit of admissions with bacteraemia from 1.7.2017 to 31.8.2018. This looked at characteristics of patients affected by age, gender, diagnosis and the profile of the microorganisms causing infection and their antibiotic sensitivities (this was not restricted to Gram-negative environmentals). The main focus of the audit seemed to be on defining the optimal choice of empirical antibiotics. It did not attempt to look at the observed frequency of bactaeraemia against that which might have been expected, but it is possible to see that 7 out the 8 most frequent bacteria identified in the series fell into the Gram-negative environmental group. We do not know where these data were presented within the organisation or what response was made.
The second document presents an analysis of episodes of care transferred to other Wards/Hospitals/Health Boards for delivery of chemotherapy and relates to data collected from 29.7.2019 to 4.11.2019, during the period when there were restrictions on admission to Ward 6A. In summary, this showed that 8 children (9 episodes of treatment) were transferred to Edinburgh during this period; 4 children (5 episodes) to Aberdeen; 1 child (1 episode) to Newcastle; and 1 young person (2 episodes) to the Young Person’s Unit at the Beatson West of Scotland Cancer Centre. Internally, accommodation was found within Ward 4B for 11 children (17 episodes) in addition to the ongoing Paediatric Stem Cell Transplant activity planned to be delivered in that ward. We have also been informed that shared care[97] activity increased during this time and has since been maintained although we have seen no data.
Short term adjustment to patient flow is expedient under such circumstances and it was good that these transfers were able to take place to limit delay to treatment. It seems, however, that there may also have been some more permanent change to shared care activity as a result of the impact of these infections. The wider development of shared care with local hospitals may have been helpful to individual families in offering more care, closer to home but appropriate structures and processes are needed to ensure that a shared care network is both supported and safe. We have not seen evidence that the issues that arose at NHS GGC were supported by any action from the Managed Service Network[98].
Contact
Email: CNOD_Admin@gov.scot