Pharmacists in Scotland - five-year integrated education programme: scoping report
Report on the integration of the existing four-year pharmacist degree and one-year training scheme into a single five-year programme.
Introduction
In response to the vision for pharmaceutical care in Scotland, the Programme for Government commitments and current and future workforce developments, the Cabinet Secretary for Health and Sport, through the auspices of the Chief Pharmaceutical Officer ( CPO), established an Advisory Group to explore the evolution of the existing pharmacist four-year undergraduate degree and the one-year pre-registration (4+1) training scheme into an integrated five-year programme to support the initial education of pharmacists in Scotland.
The initial education and training of a pharmacist comprises a four-year Master of Pharmacy (MPharm) degree and a one-year pre-registration training programme.
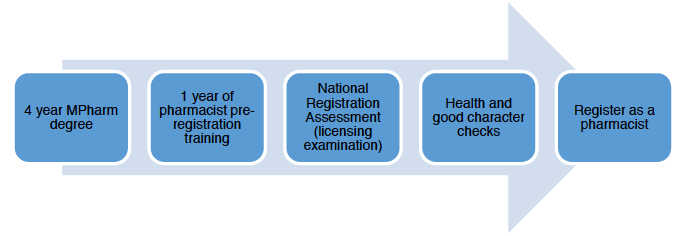
The two Higher Education Institutions ( HEIs) in Scotland who offer an MPharm are the School of Pharmacy and Life Sciences at Robert Gordon University ( RGU) in Aberdeen and Strathclyde Institute of Pharmacy and Biomedical Sciences ( SIPBS) at Strathclyde University in Glasgow. This is followed by 52-week pre-registration training programme which, in Scotland, is a national professional training year managed by NHS Education for Scotland ( NES). Pre-registration trainees then have to pass a national registration assessment and health and good character checks before registering as a pharmacist.
Pharmacy undergraduate and pre-registration education and training in Scotland is highly regarded across the UK and the Schools of Pharmacy, NES and the General Pharmaceutical Council ( GPhC) have continued to develop and improve aspects of both undergraduate and pre-registration training. However, there is an emerging view about the need for a stepped change to evolve to a more clinically focused integrated education programme in order to ensure the competencies and qualities of future graduates meet the workforce demands in Scotland. This includes delivering enhanced experiential learning in clinical practice and recognising the value of workplace learning for reinforcing confidence and competence. It also provides opportunities for earlier development and assessment of professionalism, holistic clinical decision-making and consultation and clinical assessment skills.
This report describes the outputs from the initial scoping exercise undertaken by the Advisory Group and provides a background context, as well as the drivers for change, to the current arrangements. The report outlines work undertaken during the scoping exercise and the outcome of engagement with a range of stakeholders including the NHS and staff and students at the two Scottish Schools of Pharmacy. The Advisory Group also considered evidence from both the medical and nursing professions as well as from the School of Pharmacy at the University of Nottingham in order to assimilate lessons that could be learnt from other professions and pilots.
Recommendation
The consensus view from the Advisory Group was to recommend progressing to a five-year integrated initial education programme for pharmacists in Scotland. It was the view of the Group that this recommendation provided opportunities to better prepare new pharmacists for practice in Scotland and permitted better management of pharmacy trainee numbers to meet workforce demands both in terms of initial recruitment and on-going progression. This, in turn, supported Ministerial priorities to strengthen the clinical workforce, especially in primary care. It also supports the development of the pharmacy profession towards achieving the aim that every GP practice will have access to a pharmacist with advanced clinical skills as detailed in the recently published Health and Social Care Delivery Plan, by ensuring the new generation of pharmacists are able to practice in the evolving NHS health and social care landscape.
The Advisory Group acknowledged that there were a number of different models that could be used to provide a platform to deliver the five-year integrated MPharm however they did not recommend how an integrated model would be delivered (for example a 12 month block in year five; two six month blocks, the later in the final six months of year five; or a set of dispersed blocks across the first four years and a final six month block at the end of the fifth year). They agreed that this aspect was better worked through as part of the implementation arrangements.
Advisory Group members also agreed that a Scotland-wide approach to introducing a new model was preferable in common with the Once for Scotland approach promoted by Scottish Government and the service. It could also support a regional accreditation approach going forward and this would help facilitate that from a Scotland perspective.
Implementation, transition and addressing any associated issues
In reaching its recommendation the Advisory Group identified a number of issues and logistical challenges with regards the journey towards a five-year integrated programme which led them to articulate the importance of ensuring a robust transition process which should include matters such as resourcing, culture change, internationalisation and adequate piloting in order to ensure it is introduced in a measured way. The group agreed that it was imperative that the work was taken forward building on the right founding principles, that the benefits of the changes were clearly articulated and that all essential preparatory activities were identified and translated into effective operational plans and that sufficient piloting was included prior to national roll-out.
In forming this decision, the following key issues and priorities were considered and will form the basis of transitional and implementation arrangements:
- Funding: deliver within the existing funding envelope and utilise the current funding arrangements more effectively.
- Numbers: agree and align student/trainee intake numbers including any flexibility in trainee numbers based on workforce needs to ensure NHS Scotland is self-sufficient and to provide for a progression based on merit.
- Attributes: ensure suitable attribute and qualities to meet workforce demands.
- Admissions: develop a standardised admissions process centred on values-based recruitment and including multiple mini-interviews.
- Experiential learning: increase experiential learning in the undergraduate programmes to support preparation for practice. Embed clinical decision-making and clinical assessment skills in the undergraduate MPharm. Increase interprofessional learning.
- Training providers: increase the range of training providers utilised for both MPharm experiential learning placements and pre-registration training, and provide a quality management framework to ensure effective support. Ensure the service supports experiential learning across hospital, community pharmacy and primary care.
- Quality management: develop the existing quality management processes to support the new arrangements as they are implemented.
- International students: ensure the model supports the two Schools of Pharmacy to maintain and manage international students.
Implementation outline
It is proposed that the new five-year integrated initial education programme for pharmacists in Scotland could commence implementation from 2020-2021. The aim would be for the programme to provide coterminus graduation and registration. The first students achieving this status could graduate and register in summer 2025. This will require an alignment of the reaccreditation of the two Schools of Pharmacy by the GPhC who are the pharmacy regulator in Great Britain so that they both occur within the same timescale (2019-2020). Between now and the introduction of the five-year integrated MPharm an enhanced 4+1 model will provide a transitional programme structure.
This proposal prepares for a managed integration of the 4-year MPharm and 1-year pre-registration training programmes, with progress to a fully integrated five-year programme determined by agreement with stakeholders and, in particular, students already enrolled in MPharm degrees in Scotland. The modular Pre-registration Pharmacist Scheme ( PRPS) pilot will be rolled out across the transitional period providing experience in the three main patient-facing sectors of practice.
A new funding model will be proposed for the transition period to support both trainees and training providers. Alongside this, improvements to the admissions process will mean that all students who successfully complete their MPharm during the transition period will automatically progress, based on merit, to a NES pre-registration position.
Scottish Government, in collaboration with NES, the service and the two Schools of Pharmacy, will advise on student intake numbers and a values-based admissions process including multiple mini-interviews will be developed that will include the involvement of training providers in the selection of the student intake. The plan is to pilot the admissions process in 2018-2019 and roll out from 2019-2020.
Next steps
- Agree proposal indicative timescales;
- Sign off transitional arrangements;
- Establish a series of working groups to consider: admissions and recruitment; funding; programme development (including experiential learning and interprofessional learning; and quality management and governance;
- Clarify position of international students including visa requirements; and
- Schools of Pharmacy to start planning towards accreditation of a five-year integrated MPharm.
Pharmacy and Medicines Division
Directorate of the Chief Medical Officer
April 2017
Contact
Email: Elaine Muirhead
Phone: 0300 244 4000 – Central Enquiry Unit
The Scottish Government
St Andrew's House
Regent Road
Edinburgh
EH1 3DG