Human Tissue (Authorisation) (Excepted Body Parts) (Scotland) Regulations 2020: consultation
Consultation seeking views on which parts of the body should be listed as exempt from deemed authorisation.
Intention of proposed regulations
28. The purpose of the excepted body parts regulations is to make clear which parts of the body deemed authorisation does not apply to. This applies the same principle adopted in Wales and England, where it was considered preferable to list parts of the body to which deemed authorisation doesn’t apply, as opposed to creating a list for which authorisation could be deemed. Our aim is to ensure that organs and tissue which are commonly transplanted are able to be donated with deemed authorisation. The regulations don’t affect which organs/tissue may be donated under express authorisation, although in practice it will be rare for the ‘excepted body parts’ as listed in the regulations to be donated for transplantation under express authorisation, due to the low level of clinical need.
Definitions and terminology
29. The terminology used in the regulations reflects the legal framework in Scotland. Definitions of terms used in the regulations are provided below to assist in the understanding of this consultation:
- Body parts: The term used in the 2006 Act, as amended by the 2019 Act to encompass parts of the body which may be authorised to be removed and used for transplantation. This term includes organs and tissue.
- Excepted body parts: The term used in the 2019 Act to encompass parts of the body, to be listed in regulations, that deemed authorisation will not apply to.
The excepted body parts
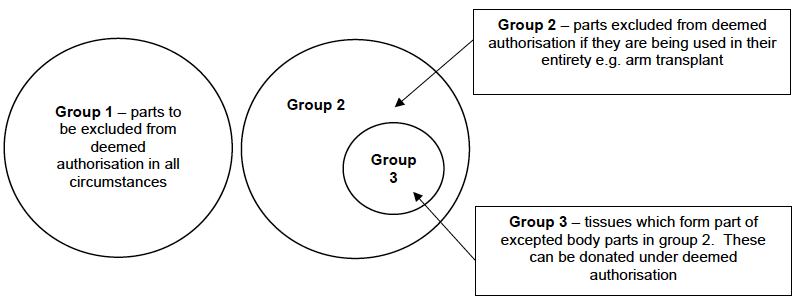
30. The proposed regulations list body parts in three different groups, depending on how they are treated in relation to deemed authorisation.
Group 1
31. It is proposed that the below list of body parts (group 1) are those which are excluded from deemed authorisation in all circumstances. These body parts can only be donated with explicit authorisation:
- Group 1 - body parts excluded from deemed authorisation in all circumstances
- brain
- cervix
- clitoris
- fallopian tube
- labia
- ovary
- penis
- perineum
- placenta
- prostate
- spinal cord
- testicle
- trachea
- umbilical cord
- uterus
- vagina
- vulva
Group 2
32. In order to ensure that current tissue donation practice is not inadvertently adversely impacted by the regulations, they set out that tissue from certain excepted body parts (those listed in group 2) may be donated under deemed authorisation. For example an area of skin taken from the leg to be used in treatment for burns victims could be donated with deemed authorisation, but the leg in its entirety couldn’t be removed with deemed authorisation. Without this measure, tissue donation in Scotland could be significantly adversely impacted which would limit the chances of those waiting for tissue transplants to receive the transplant they need.
33. Reflecting this, it is proposed that body parts listed below (group 2) would require explicit authorisation when they are being donated in their entirety. However from this list only those parts making an arm transplant (upper arm; fore arm; hand; finger) are currently transplanted from deceased donors in the UK. Where only tissue (listed in group 3) which forms part of the body part is being removed, it would be able to be removed under deemed authorisation:
- Group 2 - body parts excluded from deemed authorisation if being donated in their entirety
- face (meaning the front part of the head, from the forehead to the chin and containing the nose and mouth but excluding the eyes)
- finger
- foot
- forearm
- hand
- lower leg
- thigh
- toe
- upper arm
Group 3
34. It is proposed that the following tissue which forms part of an excepted body part included in group 2 could be donated with deemed authorisation:
- Group 3 – tissue which can be donated from a body part in group 2 with deemed authorisation
- blood vessel (meaning arteries, arterioles, capillaries, venules and veins)
- bone
- muscle
- nervous tissue
- skin
- tendon
Figure 1: How the excepted body parts regulations function
35. The procurement, storage and use of gametes or ‘reproductive cells’ is dealt with under the Human Fertilisation and Embryology Act 2008 (“the 2008 Act”) and requires a Human Fertilisation and Embryology Authority licence. It is dealt with separately from the 2006 Act and the 2019 Act and the introduction of these proposed regulations won’t change the requirements set out in the 2008 Act.
Parts of the body which would be included in deemed authorisation
36. The effect of the proposed regulations would be that all body parts which are not listed in either group 1 or 2, as well as the tissue listed in group 3, would be able to be donated under deemed authorisation. This means that deemed authorisation for transplantation will apply to all of the following parts of the body:
- heart
- lungs
- kidneys
- liver (including for liver cell transplantation)
- pancreas (including for islet transplantation)
- intestinal organs
- heart tissue (valves, conduits and patches)
- eyes (including for both cornea and sclera transplantation)
unless from a body part listed in group 1
- nervous tissue
- blood vessels
- bone
- muscle
- tendon
- skin
Contact
Email: ODlegislation@gov.scot